FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
DENAVIR is a deoxynucleoside analog HSV DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in adults and children 12 years of age or older.
2 DOSAGE AND ADMINISTRATION
Apply DENAVIR every 2 hours during waking hours for a period of 4 days. Start treatment as early as possible (i.e., during the prodrome or when lesions appear).
3 DOSAGE FORMS AND STRENGTHS
DENAVIR is supplied in a 5 gram tube containing 10 mg of penciclovir per gram in a cream base, which is equivalent to 1% (w/w).
4 CONTRAINDICATIONS
DENAVIR is contraindicated in patients with known hypersensitivity to the product or any of its components.
5 WARNINGS AND PRECAUTIONS
5.1 General
DENAVIR should only be used on herpes labialis on the lips and face. Because no data are available, application to human mucous membranes is not recommended. Particular care should be taken to avoid application in or near the eyes since it may cause irritation. Lesions that do not improve or that worsen on therapy should be evaluated for secondary bacterial infection. The effect of DENAVIR has not been established in immunocompromised patients.
6 ADVERSE REACTIONS
6.1 Clinical Studies
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In two double-blind, placebo-controlled trials, 1,516 patients were treated with DENAVIR (penciclovir cream) and 1,541 with placebo. One or more local adverse reactions were reported by 3% of the patients treated with DENAVIR and 4% of placebo-treated patients. The rates of reported local adverse reactions are shown in Table 1.
|
Penciclovir N=1,516 % |
Placebo N=1,541 % |
|
|
Application site reaction |
1 |
2 |
|
Hypesthesia/Local anesthesia |
<1 |
<1 |
|
Taste perversion |
<1 |
<1 |
|
Rash (erythematous) |
<1 |
<1 |
Two studies, enrolling 108 healthy subjects, were conducted to evaluate the dermal tolerance of 5% penciclovir cream (a 5-fold higher concentration than the commercial formulation) compared to vehicle using repeated occluded patch testing methodology. The 5% penciclovir cream induced mild erythema in approximately one-half of the subjects exposed, an irritancy profile similar to the vehicle control in terms of severity and proportion of subjects with a response. No evidence of sensitization was observed.
6.2 Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of DENAVIR. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following events have been identified from worldwide post-marketing use of DENAVIR in treatment of recurrent herpes labialis (cold sores) in adults. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to DENAVIR.
General: Headache, oral/pharyngeal edema, parosmia.
Skin: Aggravated condition, decreased therapeutic response, local edema, pain, paresthesia, pruritus, skin discoloration, and urticaria.
7 DRUG INTERACTIONS
No drug interaction studies have been performed with DENAVIR. Due to minimal systemic absorption of DENAVIR, systemic drug interactions are unlikely.
8 USE IN SPECIFIC POPULATIONS
8.4 Pediatric Use
An open-label, uncontrolled trial with penciclovir cream 1% was conducted in 102 subjects, ages 12-17 years, with recurrent herpes labialis. The frequency of adverse events was generally similar to the frequency previously reported for adult patients. Safety and effectiveness in pediatric patients less than 12 years of age have not been established.
10 OVERDOSAGE
Since penciclovir is poorly absorbed following oral administration, adverse reactions related to penciclovir ingestion are unlikely. There is no information on overdose.
11 DESCRIPTION
DENAVIR (penciclovir) cream 1% contains penciclovir, an antiviral agent active against herpes viruses. DENAVIR is available for topical administration as a 1% white cream. Each gram of DENAVIR contains 10 mg of penciclovir and the following inactive ingredients: cetostearyl alcohol, mineral oil, polyoxyl 20 cetostearyl ether, propylene glycol, purified water and white petrolatum.
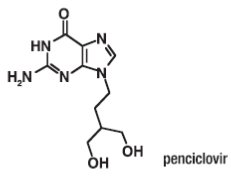
Chemically, penciclovir is known as 9-[4-hydroxy-3-(hydroxymethyl)butyl] guanine. Its molecular formula is C10H15N5O3; its molecular weight is 253.26. It is a synthetic acyclic guanine derivative and has the following structure:
Penciclovir is a white to pale yellow solid. At 20°C it has a solubility of 0.2 mg/mL in methanol, 1.3 mg/mL in propylene glycol, and 1.7 mg/mL in water. In aqueous buffer (pH 2) the solubility is 10.0 mg/mL. Penciclovir is not hygroscopic. Its partition coefficient in n-octanol/water at pH 7.5 is 0.024 (logP = -1.62).
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Penciclovir is an antiviral agent active against alpha herpes viruses [see Microbiology (12.4)].
12.3 Pharmacokinetics
Measurable penciclovir concentrations were not detected in plasma or urine of healthy male volunteers (n=12) following single or repeat application of the 1% cream at a dose of 180 mg penciclovir daily (approximately 67 times the estimated usual clinical dose).
12.4 Microbiology
Mechanism of Action
Penciclovir has inhibitory activity against herpes simplex virus types 1 (HSV-1) and 2 (HSV-2). In cells infected with HSV-1 or HSV-2, the viral thymidine kinase phosphorylates penciclovir to a monophosphate form that, in turn, is converted by cellular kinases to the active form penciclovir triphosphate. Biochemical studies demonstrate that penciclovir triphosphate inhibits HSV DNA polymerase competitively with deoxyguanosine triphosphate. Consequently, α-herpes viral DNA synthesis and, therefore, replication are selectively inhibited. Penciclovir triphosphate has an intracellular half-life of 10 hours in HSV-1 and 20 hours in HSV-2 infected cells grown in culture. However, the clinical significance of the intracellular half-life is unknown.
Antiviral Activity
In cell culture studies, penciclovir has antiviral activity against the following herpes viruses: HSV-1 and HSV-2. The antiviral activity of penciclovir against wild type strains grown on human foreskin fibroblasts was assessed with a plaque reduction assay and staining with crystal violet 3 days post-infection for HSV. The median EC50 values of penciclovir against laboratory and clinical isolates of HSV-1 and HSV-2 were 2 μM (range 1.2 to 2.4 μM, n=7) and 2.6 μM (range 1.6 to 11 μM, n=6), respectively.
Resistance
In Cell Culture
Penciclovir-resistant HSV-1 and HSV-2 strains were isolated in cell culture. Penciclovir-resistant mutants of HSV resulted from mutations in the viral thymidine kinase (TK) and DNA polymerase (POL) genes. Frameshifts were commonly isolated and result in premature truncation of the HSV TK product with decreased enzymatic activity and consequent decreased susceptibility to penciclovir. Mutations in the viral TK gene may lead to complete loss of TK activity (TK negative), reduced levels of TK activity (TK partial), or alteration in the ability of viral TK to phosphorylate the drug without an equivalent loss in the ability to phosphorylate thymidine (TK altered). In cell culture, the following resistance-associated substitutions in TK of HSV-1 and HSV-2 were observed: HSV-1 TK G6C, F13L, H142Y, G200D, L205S, S254Stop, V267G, and T287M; HSV-2 TK G39E, F191L, E226K, and T288M. The median EC50 values observed in a plaque reduction assay with penciclovir resistant HSV-1 and HSV-2 were 69 μM (range 14 to 115 μM, n=6) and 46 μM (range 4 to > 395 μM, n=9), respectively.
Resistance and Cross-resistance in Clinical Isolates from HSV-Infected Patients
Clinical HSV-1 and HSV-2 isolates obtained from patients who failed treatment with acyclovir for their α-herpesvirus infections were evaluated for genotypic changes in the TK and POL genes. These HSV isolates had frameshift mutations leading to loss of thymidine kinase or had substitutions in the viral thymidine kinase and viral DNA polymerase. Phenotypic analysis of these clinical isolates confirmed resistance to penciclovir and acyclovir. These and other resistance-associated substitutions reported in the literature, or observed in clinical trials, are listed in Table 2. The list is not all inclusive and additional changes will likely be identified in HSV variants isolated from patients who fail penciclovir containing regimens. The possibility of viral resistance to penciclovir should be considered in patients who fail to respond or experience recurrent viral shedding during therapy.
| Note: Many additional pathways to penciclovir resistance likely exist. | ||
|
HSV-1 |
TK |
G6C, R32H, R51W, Y53C/H, H58N, G59W, G61A, S74Stop, E83K, P84L, T103P, Q104Stop, D116N, M121R, I143V, R163H, L170P, Y172C, A174P, R176Q/W, Q185R, A189V, G200D, G206R, L208H, R216C, R220H, R222C/H, FS 224, Y239S, T245M, Q250Stop, S254Stop, R256W, Q261Stop, R281Stop, T287M, L315S, M322K, C336Y |
|
HSV-2 |
TK |
G39E, R51W, Y53N, R221H, T288M |
|
HSV-1 |
POL |
A657T, D672N, V715G, A719V, S724N, E798K, V813M, N815S, Y818C, G841S, R842S, F891C, V958L |
|
HSV-2 |
POL |
- |
Cross-resistance has been observed among HSV isolates carrying foscarnet resistance-associated substitutions (Table 3).
|
HSV-1 |
POL |
D672N, S724N, E798K, V813M, Y818C, F891C, V958L |
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In clinical trials, systemic drug exposure following topical administration of penciclovir cream was negligible, as the penciclovir content of all plasma and urine samples was below the limit of assay detection (0.1 mcg/mL and 10 mcg/mL, respectively). However, for the purpose of inter-species dose comparisons presented in the following sections, an assumption of 100% absorption of penciclovir from the topically applied product has been used. Based on the use of the maximal recommended topical dose of penciclovir of 0.05 mg/kg/day and an assumption of 100% absorption, the maximum theoretical plasma AUC0-24 hrs for penciclovir is approximately 0.129 mcg.hr/mL.
Carcinogenesis
Two-year carcinogenicity studies were conducted with famciclovir (the oral prodrug of penciclovir) in rats and mice. An increase in the incidence of mammary adenocarcinoma (a common tumor in female rats of the strain used) was seen in female rats receiving 600 mg/kg/day (approximately 395x the maximum theoretical human exposure to penciclovir following application of the topical product, based on area under the plasma concentration curve comparisons [24 hr. AUC]). No increases in tumor incidence were seen among male rats treated at doses up to 240 mg/kg/day (approximately 190x the maximum theoretical human AUC for penciclovir), or in male and female mice at doses up to 600 mg/kg/day (approximately 100x the maximum theoretical human AUC for penciclovir).
Mutagenesis
When tested in vitro, penciclovir did not cause an increase in gene mutation in the Ames assay using multiple strains of S. typhimurium or E. coli (at up to 20,000 mcg/plate), nor did it cause an increase in unscheduled DNA repair in mammalian HeLa S3 cells (at up to 5,000 mcg/mL). However, an increase in clastogenic responses was seen with penciclovir in the L5178Y mouse lymphoma cell assay (at doses ≥1000 mcg/mL) and, in human lymphocytes incubated in vitro at doses ≥250 mcg/mL. When tested in vivo, penciclovir caused an increase in micronuclei in mouse bone marrow following the intravenous administration of doses ≥500 mg/kg (≥810x the maximum human dose, based on body surface area conversion).
Impairment of Fertility
Testicular toxicity was observed in multiple animal species (rats and dogs) following repeated intravenous administration of penciclovir (160 mg/kg/day and 100 mg/kg/day, respectively, approximately 1155 and 3255x the maximum theoretical human AUC). Testicular changes seen in both species included atrophy of the seminiferous tubules and reductions in epididymal sperm counts and/or an increased incidence of sperm with abnormal morphology or reduced motility. Adverse testicular effects were related to an increasing dose or duration of exposure to penciclovir. No adverse testicular or reproductive effects (fertility and reproductive function) were observed in rats after 10 to 13 weeks dosing at 80 mg/kg/day, or testicular effects in dogs after 13 weeks dosing at 30 mg/kg/day (575 and 845x the maximum theoretical human AUC, respectively). Intravenously administered penciclovir had no effect on fertility or reproductive performance in female rats at doses of up to 80 mg/kg/day (260x the maximum human dose [BSA]).
There was no evidence of any clinically significant effects on sperm count, motility or morphology in 2 placebo-controlled clinical trials of Famvir® (famciclovir [the oral prodrug of penciclovir], 250 mg twice daily; n=66) in immunocompetent men with recurrent genital herpes, when dosing and follow-up were maintained for 18 and 8 weeks, respectively (approximately 2 and 1 spermatogenic cycles in the human).
14 CLINICAL STUDIES
DENAVIR was studied in two double-blind, placebo (vehicle)-controlled trials for the treatment of recurrent herpes labialis in which otherwise healthy adults were randomized to either DENAVIR or placebo. Therapy was to be initiated by the subjects within 1 hour of noticing signs or symptoms and continued for 4 days, with application of study medication every 2 hours while awake. In both studies, the mean duration of lesions was approximately one-half-day shorter in the subjects treated with DENAVIR (N=1,516) as compared to subjects treated with placebo (N=1,541) (approximately 4.5 days versus 5 days, respectively). The mean duration of lesion pain was also approximately one half-day shorter in the DENAVIR group compared to the placebo group.
16 HOW SUPPLIED/STORAGE AND HANDLING
DENAVIR is supplied in a 5 gram tube containing 10 mg of penciclovir per gram in a cream base, which is equivalent to 1% (w/w). The white cream is available as follows:
NDC 0378-9720-55
carton containing one 5 gram tube
Store at 20º to 25ºC (68º to 77ºF). [See USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
Administration Instructions
Advise the patient that treatment should be started at the earliest sign of a cold sore (i.e. tingling, redness, itching, or bump). Inform the patient to apply the cream every 2 hours during waking hours for 4 days and to wash their hands with soap and water after using DENAVIR.
Denavir is a registered trademark of Denco Asset, LLC, a Mylan Company.
The brands listed are trademarks of their respective owners.
©2018 Mylan Pharmaceuticals Inc.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Manufactured by:
Confab Laboratories Inc.
St-Hubert, Canada J3Y 3X3
Revised: 11/2018
301459-03
CON:PENCCR:R2
PRINCIPAL DISPLAY PANEL – 1%
NDC 0378-9720-55
Rx only
Denavir®
(penciclovir cream) 1%
For Topical Use Only
5 g (Net Wt.)
Each gram contains 10 mg penciclovir; inactive ingredients consist of cetomacrogol 1000
BP, cetostearyl alcohol, mineral oil, propylene glycol, purified water and white petrolatum.
Usual Dosage: See accompanying prescribing information.
To Open Tube: Unscrew cap, puncture aluminum seal with pointed end of cap.
Store at controlled room temperature 20° to 25°C (68° to 77°F).
[See USP Controlled Room Temperature.]
Denavir® is a registered trademark of Denco Asset, LLC, a Mylan Company
© 2018 Mylan Pharmaceuticals Inc.
Comments or Questions? Call 1-877-446-3679 (1-877-4-INFO-RX)
Manufactured in Canada for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
CON:9720:55:1C:R1