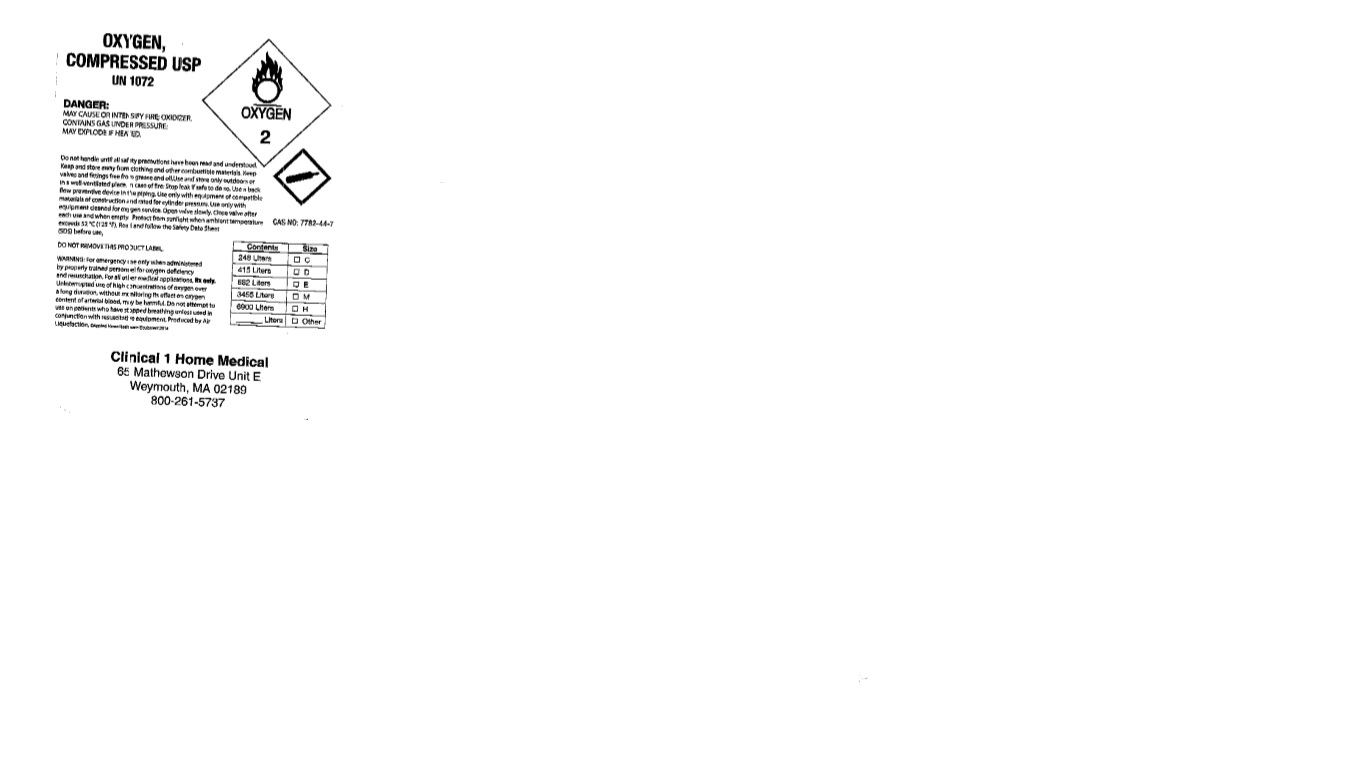
OXYGEN, COMPRESSED USP
UN 1072
DANGER: MAY CAUSE OR INTENSIFY FIRE; OXIDIZER. CONTAINS GAS UNDER PRESSURE; MAY EXPLODE IF HEATED.
Do not handle until all safety precautions have been read and understood. Keep and store away from clothing and other combustible materials. Keep valves and fittings free from grease and oil. Stop leak if safe to do so. Use a back flow preventive device in the piping. Use only with equipment of compatible materials of construction and rated for cylinder pressure. Use only with equipment cleaned for oxygen service. Open valve slowly. Close valve after each use and when empty. Protect from sunlight when ambient temperature exceeds 52 C (125 F). Read and follow the Safety Data Sheet (SDS) before use.
DO NOT REMOVE THIS PRODUCT LABEL.
WARNING: For emergency use only when administered by properly trained personnel for oxygen deficiency and resuscitation. For all other medical applications, Rx only. Uninterrupted use of high concentrations of oxygen over a long duration, without monitoring its effect on oxygen content of arterial blood, may be harmful. Do not attempt to use on patients who have stopped breathing unless used in conjunction with resuscitative equipment. Produced by Air Liquefaction.
Clinical 1 Home Medical
65 Mathewson Drive Unit E
Weymouth, MA 02189
800-261-5737
CAS NO: 7782-44-7
OXYGEN, REFRIGERATED LIQUID USP
UN 1073
CONTENTS: LITERS
CAS NO: 7782-44-7
DANGER:
MAY CAUSE OR INTENSIFY FIRE; OXIDIZER. CONTAINS REFRIGERATED GAS; MAY CAUSE CRYOGENIC BURNS OR INJURY. COMBUSTIBLES IN CONTACT WITH LIQUID OXYGEN MAY EXPLODE ON IGNITION OR IMPACT.
Do not handle until all safety precautions have been read and understood. Keep and store away from clothing and other combustible materials. Keep valves and fittings free from grease and oil. Use and store only outdoors or in a well-ventilated place. Wear cold insulating gloves, face shield, and eye protection. In case of fire: Stop leak if safe to do so. Use a back flow preventive device in the piping. Use only with equipment of compatible materials of construction and rated for cylinder pressure. Use only with equipment cleaned for oxygen service. DO NOT change or force fit connections. Avoid spills. Do not walk on or roll equipment over spills. Close valve after each use and when empty. Always keep container in upright position. Read and follow the Safety Data Sheet (SDS) before use.
FIRST AID:
IF ON SKIN: Thaw frosted parts with lukewarm water. Do not rub affected area. Get immediate medical advice/attention.
DO NOT REMOVE THIS PRODUCT LABEL.
Warning: For emergency use only when administered by properly trained personnel for oxygen deficiency and resuscitation. For all other medical applications, Rx only. Uniterrupted use of high concentrations of oxygen over a long duration, without monitoring its effect on oxygen content of arterial blood, may be harmful. Do not attempt to use on patients who have stopped breathing unless used in conjunction with resuscitative equipment. Produced by Air Liquefaction.
Clinical 1 Home Medical
65 Mathewson Drive
Unit E
Weymouth, MA 02189
800-261-5737
