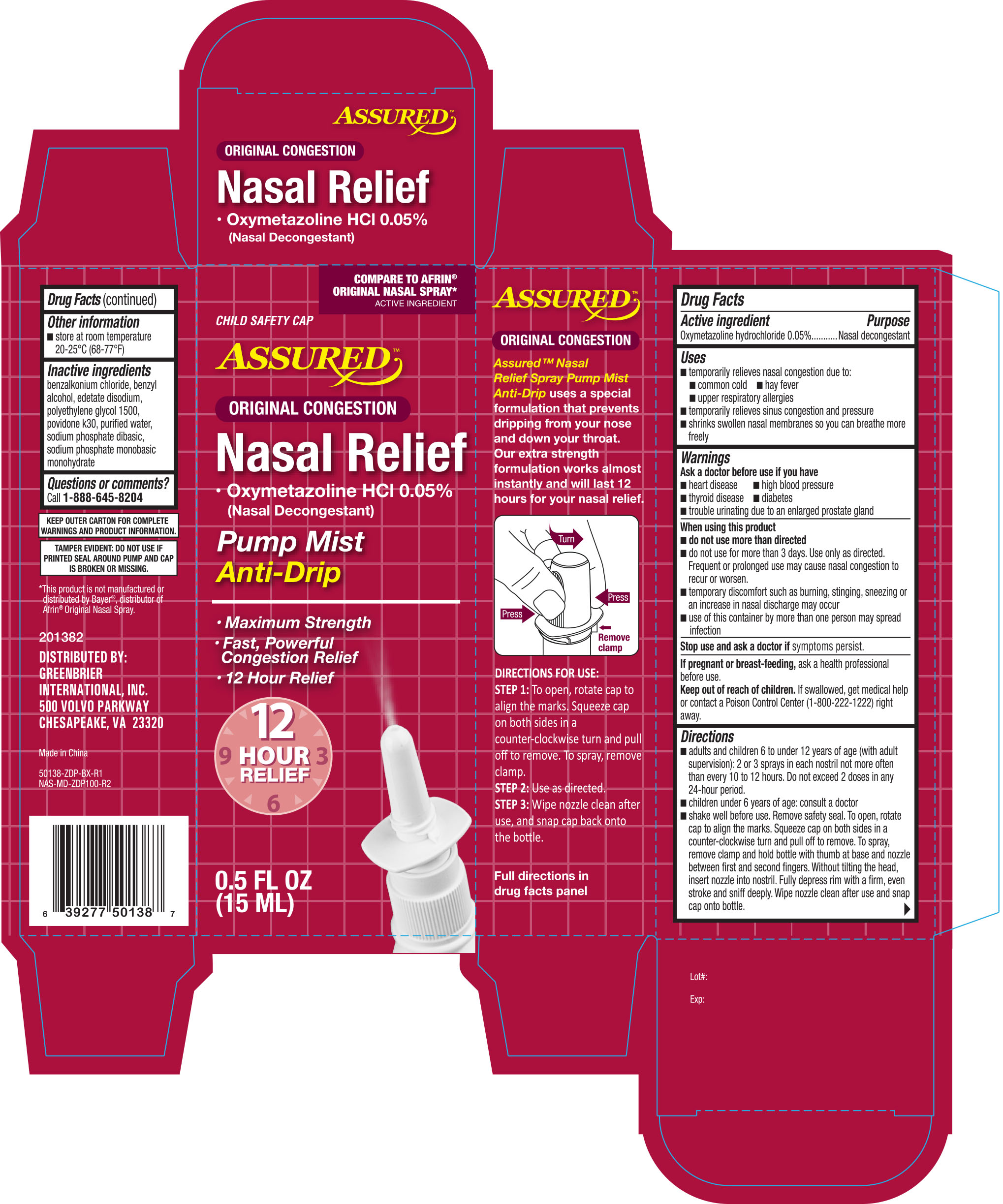
Active Ingredient Purpose
Oxymetazoline hydrochloride 0.05% ...........................................Nasal decongestant
Uses
- temporarily relieved nasal congestion due to:
- common cold
- hay fever
- upper respiratory allergies
- sinusitis
- shrinks swollen nasal membranes so you can breathe more freely
Ask a doctor before use if you have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
When using this product
- do not use more than directed
- do not use more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen
- temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
- use of this container by more than one person may spread infection
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- adults and children 6 to 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period.
- children under 6 years of age: ask a doctor. To open, rotate cap to align the marks. Squeeze cap on both sides in a counter-clockwise turn and pull off to remove. To srapy, remove clamp and hold bottle with thumb at base and nozzle between first and second fingers. Without tiliting the head, insert nozzle into nostril. Fully depress rim with a firm, even stroke and sniff deeply. Wipe nozzle clean after use and snap cap back onto the bottle.
Other information
- store between 20 o and 25 oC (68 o and 77 oF)
- retain carton for future reference on full labeling