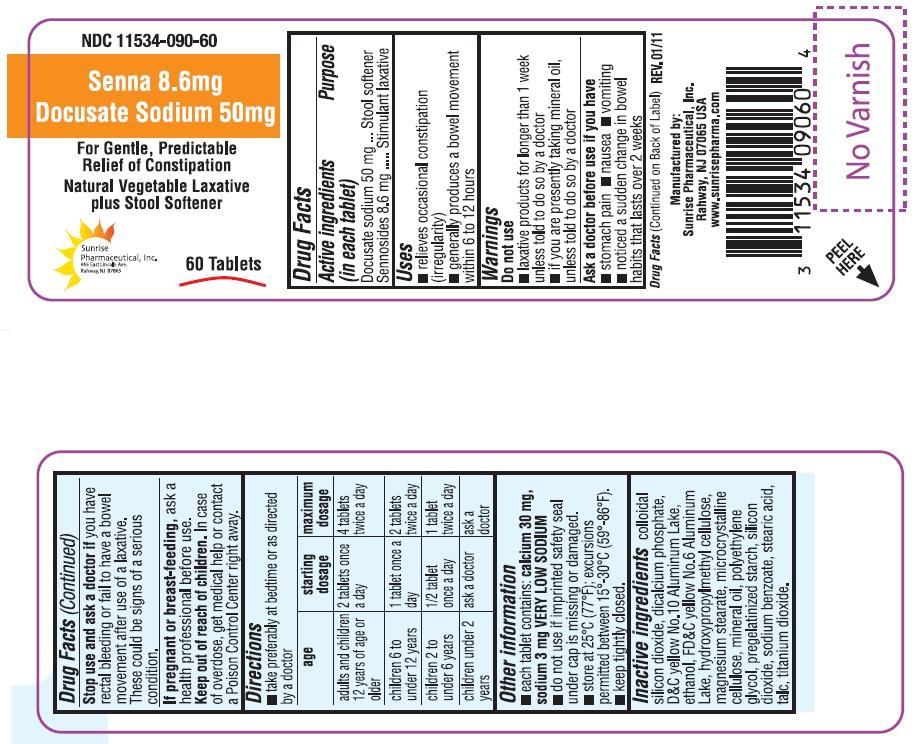
INDICATIONS AND USAGE
- relieves occasional constipation (irregularity)
- generally produces a bowel movement within 6 to 12 hours
WARNINGS
Do not use:
- laxative products for longer than 1 week unless told to do so by a doctor
- if you are presently taking mineral oil, unless told to do so by a doctor
OTC - ASK A DOCTOR BEFORE USE IF YOU HAVE
- Stomach pain
- Nausea
- Vomiting
- Noticed a sudden change in bowel habits that lasts over 2 weeks
OTC - STOP USE AND ASK A DOCTOR IF
You have rectal bleeding or fail to have a bowel movement after use of a laxative. These could be signs of a serious condition.
OTC - KEEP OUT OF REACH OF CHILDREN
In case of overdose, get medical help or contact a Poison Control Center right away.
DOSAGE AND ADMINISTRATION
- Take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
| adults and children 12 years of age or older | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years | ½ tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
OTHER INFORMATION
- Each tablet contains: calcium 30 mg, sodium 3 mg VERY LOW SODIUM
- Do not use if imprinted safety seal under cap is missing or damaged.
- Store at 25° C (77° F); excursions permitted between 15° C-30° C (59° -86° F).
- Keep tightly closed.
INACTIVE INGREDIENT
Colloidal silicon dioxide, dicalcium phosphate, D&C yellow No 10 Aluminum Lake, ethanol, FD&C yellow No 6 Aluminum Lake, hydroxypropyl methyl cellulose, magnesium stearate, microcrystalline cellulose, mineral oil, polyethylene glycol, pregelatinized starch, silicon dioxide, sodium benzoate, stearic acid, talc, titanium dioxide