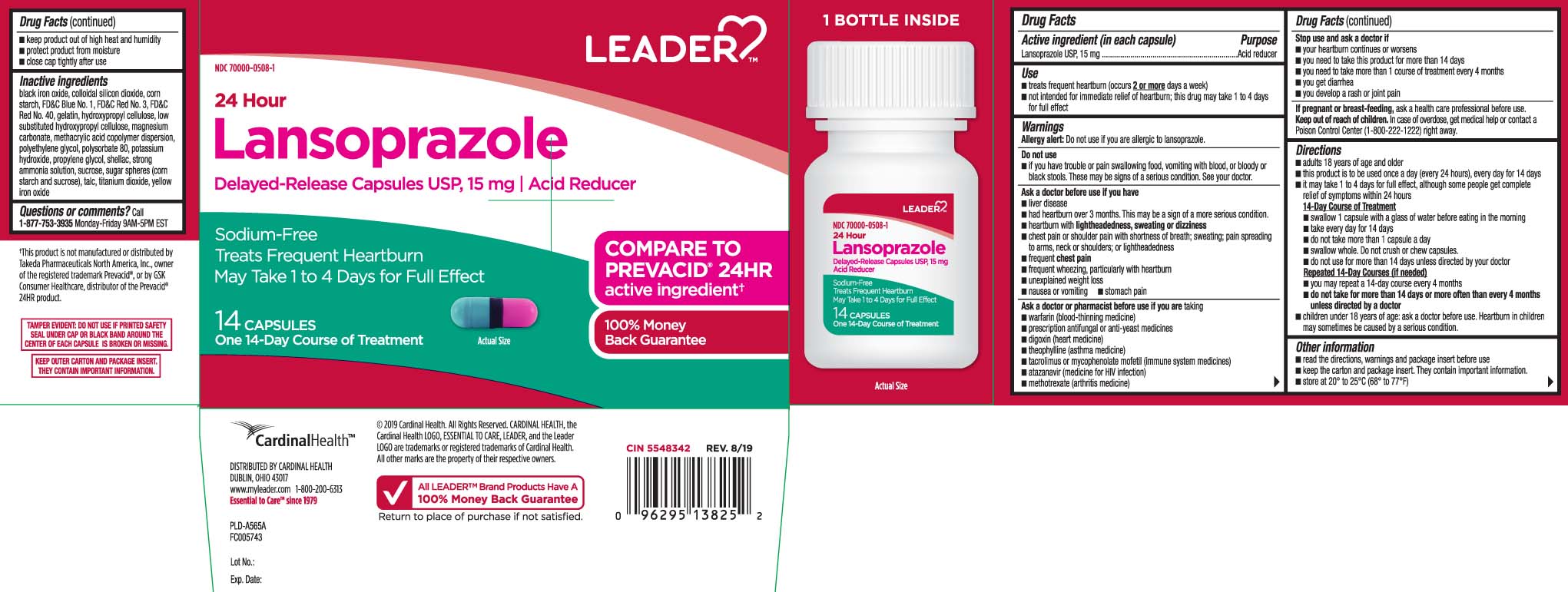
Use
- treats frequent heartburn (occurs 2 or more days a week)
- not intended for immediate relief of heartburn; this drug may take 1 to 4 days for full effect
WARNINGS
Allergy alert: Do not use if you are allergic to lansoprazole.
Do not use
- if you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools. These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have
- liver disease
- had heartburn over 3 months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Ask a doctor or pharmacist before use if you are
taking
- warfarin (blood-thinning medicine)
- prescription antifungal or anti-yeast medicines
- digoxin (heart medicine)
- theophylline (asthma medicine)
- tacrolimus or mycophenolate mofetil (immune system medicine)
- atazanavir (medicine for HIV infection)
- methotrexate (arthritis medicine)
Directions
- adults 18 years of age and older
- this product is to be used once a day (every 24 hours), every day for 14 days
- it may take 1 to 4 days for full effect, although some people get complete relief of symptoms within 24 hours
14-Day Course of Treatment
- swallow 1 capsule with a glass of water before eating in the morning
- take every day for 14 days
- do not take more than 1 capsule a day
- swallow whole. Do not crush or chew capsules.
- do not use for more than 14 days unless directed by your doctor
Repeated 14-Day Courses (if needed)
- you may repeat a 14-day course every 4 months
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- children under 18 years of age: ask a doctor before use. Heartburn in children may sometimes be caused by a serious condition.
Other information
- read the directions, warnings and package insert before use
- keep the carton and package insert. They contain important information.
- store at 20°-25°C (68°-77°F)
- keep product out of high heat and humidity
- protect product from moisture
- close cap tightly after use
Inactive ingredients
black iron oxide,colloidal silicon dioxide, corn starch, FD&C Blue #1, FD&C red #3, FD&C red #40, gelatin, hydroxypropyl cellulose, low substituted hydroxypropyl cellulose, magnesium carbonate, methacrylic acid copolymer dispersion, polyethylene glycol, polysorbate 80, potassium hydroxide, propylene glycol, shellac, strong ammonia solution, sucrose, sugar spheres (corn starch and sucrose), talc, titanium dioxide, yellow iron oxide
Principal Display Panel
COMPARE TO PREVACID® 24 HR active ingredient†
24 Hour
Lansoprazole
Delayed-Release Capsules USP, 15 mg | Acid Reducer
Sodium-Free
Treats Frequent Heartburn
May Take 1 to 4 Days for Full Effect
Capsules
†This product is not manufactured or distributed by Takeda Pharmaceuticals North America, Inc., owner of the registered trademark Prevacid®, or by GSK Consumer Healthcare, distributor of the Prevacid® 24HR product.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP OR BLACK BAND AROUND THE CENTER OF EACH CAPSULES IS BROKEN OR MISSING.
KEEP OUTER CARTON AND PACKAGE INSERT. THEY CONTAIN IMPORTANT INFORMATION
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com 1-800-200-6313