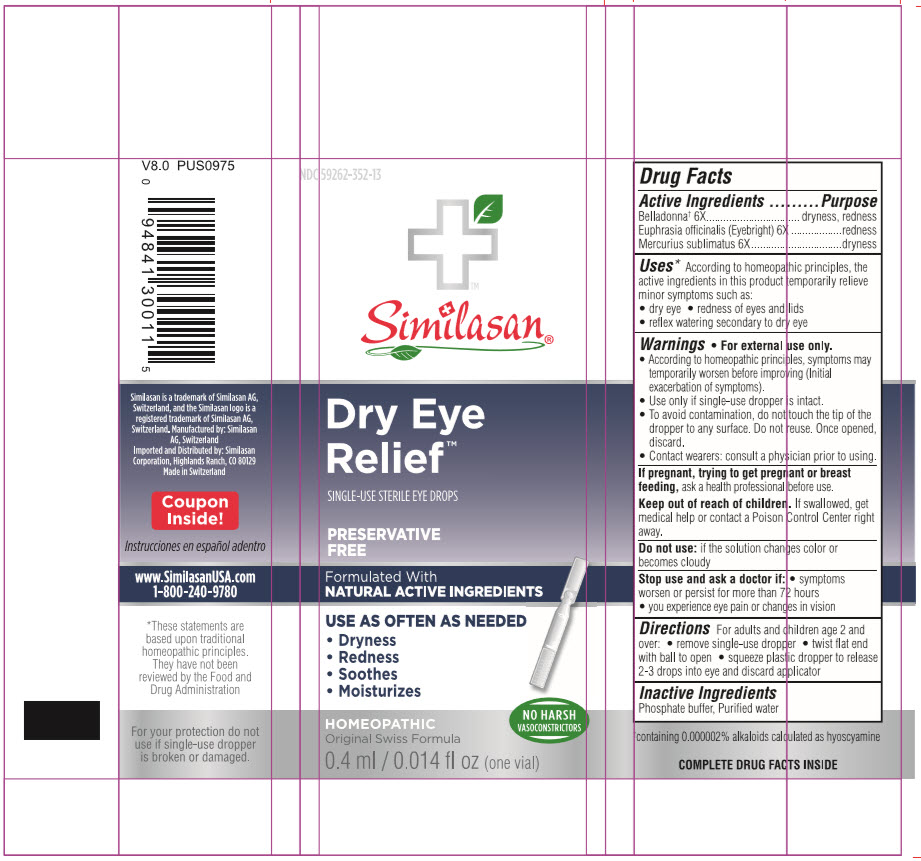
DRY EYE RELIEF- atropa belladonna and euphrasia stricta and mercuric chloride solution/ drops
Similasan Corporation
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
Active Ingredient
Belladonna* 6X
*containing 0.000002% alkaloids calculated as hyoscyamine.
Active Ingredient
Euphrasia 6X
Active Ingredient
Mercurius sublimatus 6X
Uses
According to homeopathic principles, the active ingredients in this medication temporarily relieve minor symptoms such as:
- dry eye
- redness of eyes and lids
- reflex watering secondary to dry eye
- sensation of grittiness
- sensitivity to light
Warnings
-
For external use only.
- Initial exacerbation of symptoms may occur.
- Use only if single-use dropper is intact.
- To avoid contamination, do not touch the tip of the dropper to any surface. Do not reuse. Once opened, discard.
- Contact wearers: consult a physician prior to using.
Do not use:
if the solution changes color or becomes cloudy
Stop use and ask a doctor if:
- symptoms worsen or persist for more than 72 hours.
- changes in vision occur.
- you experience eye pain.
If pregnant or breast feeding,
ask a health professional before use.
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
For adults and children age 2 and over:
- remove a single-use dropper
- twist flat end with ball to remove
- squeeze plastic dropper to release 2-3 drops into eye and discard applicator
Inactive Ingredients
Phosphate buffer, Purified water
Principal Display Panel
NDC 59262-352-13
Similasan
Dry Eye
Relief
SINGLE-USE STERILE EYE DROPS
Preservative Free
0.4 ml/ 0.014 fl oz each
Principal Display Panel
SIMILASAN / USA
30011
Dry Eye Relief, eye drops
270 pieces(Stk)

