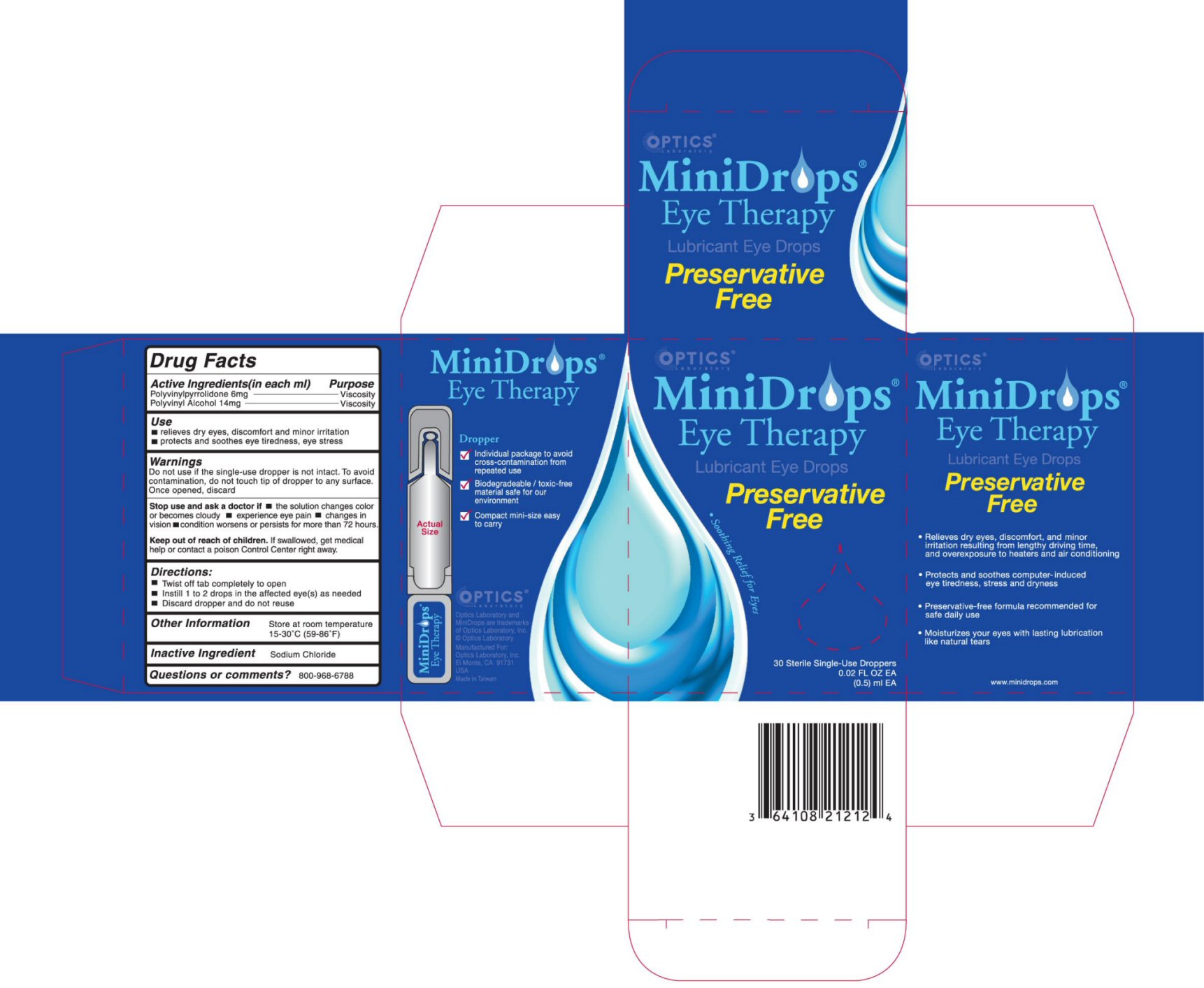
MINIDROPS - lubricant eye drops liquid
Taiwan Biotech Co, Ltd
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
ACTIVE INGREDIENTS (in each ml)
Polyvinylpyrrolidone 6 mg
Polyvinyl Alcohol 14mg
PURPOSE
PURPOSE VISCOSITY
PURPOSE VISCOSITY
USE
RELIEVES DRY EYES, DISCOMFORT AND MINOR IRRITATION
PROTECTS AND SOOTHES EYE TIREDNESS, EYE STRESS
WARNINGS
DO NOT USE IF THE SINGLE-USE DROPPER IS NOT INTACT. TO AVOID CONTAMINATION, DO NOT TOUCH TIP OF DROPPER TO ANY SURFACE. ONCE OPENED, DISCARD.
STOP USE AND ASK A DOCTOR IF
- THE SOLUTION CHANGES COLOR OR BECOMES CLOUDY
- EXPERIENCE EYE PAIN
- CHANGES IN VISION
- CONDITION WORSENS OR PERSISTS FOR MORE THAN 72 HOURS.
KEEP OUT OF REACH OF CHILDREN
IF SWALLOWED GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
DIRECTIONS
- TWIST OFF TAB COMPLETELY TO OPEN
- INSTILL 1 TO 2 DROPS IN THE AFFECTED EYE(S) AS NEEDED
- DISCARD DROPPER AND DO NOT REUSE
OTHER INFORMATION
STORE AT ROOM TEMPERATURE 15-300 C (59-860 F)
INACTIVE INGREDIENT
SODIUM CHLORIDE
QUESTIONS OR COMMENTS?
800-968-6788