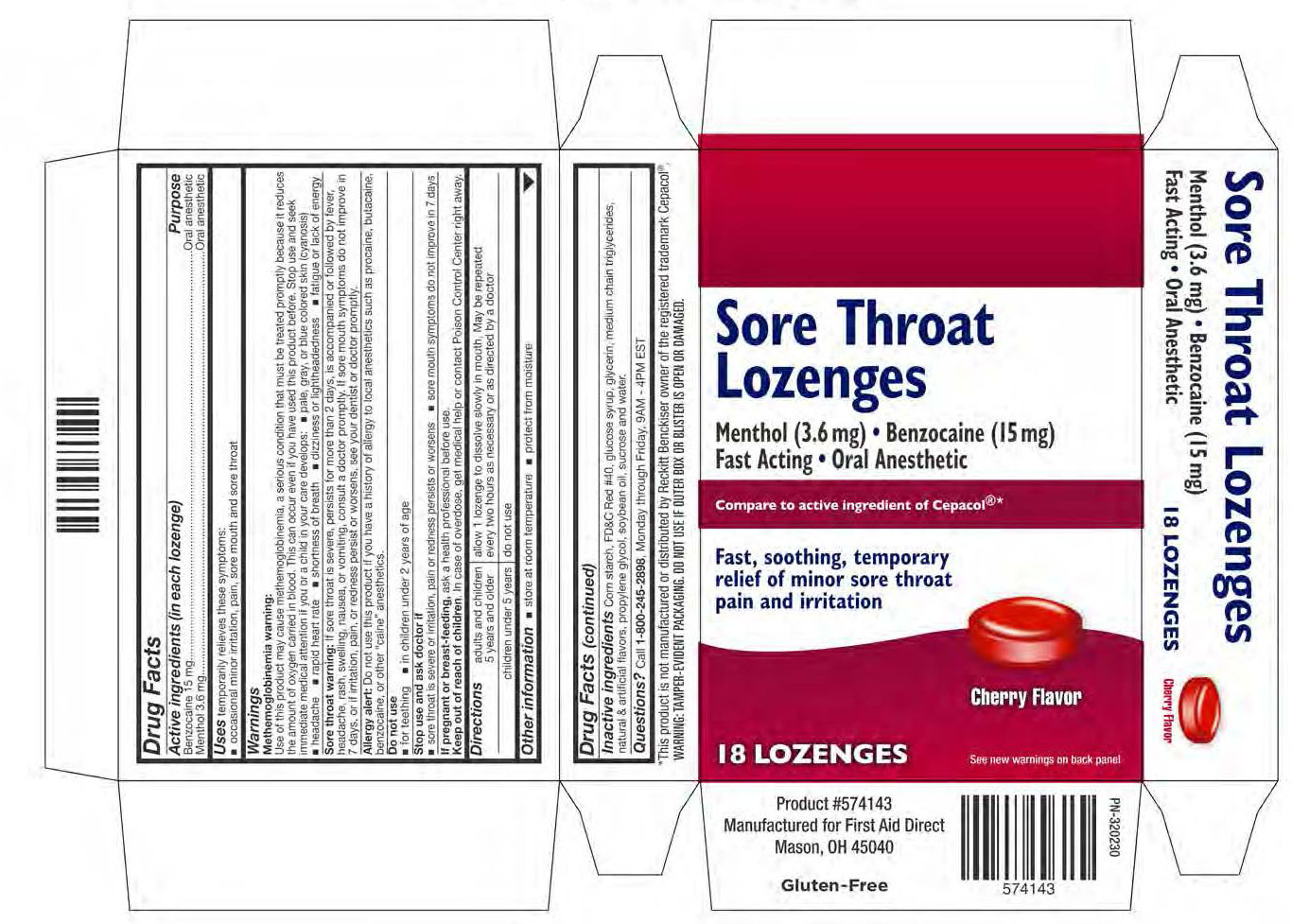
Uses temporarily relieves these symptoms:
occasional minor irritation, pain, sore mouth and sore throat
Warnings
Methemoglobinemia warning:
Use of this product may cause methemoglobinemia, a serious conditin that must be treated promptly because it reduces the amount of oxygen carried in blood. This can occur even if you have used this product before. Stop use and seek immediate medical attention if you or a child in your care develops:
- pale, gray, or blue colored skin (cyanosis)
- headache
- rapid heart rate
- shortness of breath
- dizziness or lightheadedness
- fatigue or lack of energy
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, swelling, nausea, or vomiting, consult a doctor promptly . If sore mouth symptoms do not improve in 7 days, or if irritation, pain, or redness persists or worsens, see your dentist or doctor promptly.
Allergy alert: Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "caine" anesthetics.
Stop use and ask doctor if
- sore throat is severe or irritation, pain or redness persists or worsens
- sore mouth symptoms do not improve in 7 days
Directions
adults and children 5 years and older - allow 1 lozenge to dissolve slowly in mouth. May be repeated every 2 hours as necessary or as directed by a doctor.
children under 5 years - do not use