FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Durezol (difluprednate ophthalmic emulsion) 0.05%, a topical corticosteroid, is indicated for the treatment of inflammation and pain associated with ocular surgery.
2 DOSAGE AND ADMINISTRATION
Instill one drop into the conjunctival sac of the affected eye(s) 4 times daily beginning 24 hours after surgery and continuing throughout the first 2 weeks of the postoperative period, followed by 2 times daily for a week and then a taper based on the response.
3 DOSAGE STRENGTHS
Durezol contains 0.05% difluprednate as a sterile preserved emulsion for topical ophthalmic administration.
4 CONTRAINDICATIONS
The use of Durezol, as with other ophthalmic corticosteroids, is contraindicated in most active viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal disease of ocular structures.
5 WARNINGS AND PRECAUTIONS
5.1 IOP Increase
Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. Steroids should be used with caution in the presence of glaucoma. If this product is used for 10 days or longer, intraocular pressure should be monitored.
5.3 Delayed Healing
The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical steroids. The initial prescription and renewal of the medication order beyond 28 days should be made by a physician only after examination of the patient with the aid of magnification such as slit lamp biomicroscopy and, where appropriate, fluorescein staining.
5.4 Bacterial Infections
Prolonged use of corticosteroids may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions, steroids may mask infection or enhance existing infection. If signs and symptoms fail to improve after 2 days, the patient should be re-evaluated.
5.5 Viral Infections
Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution. Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex).
5.6 Fungal Infections
Fungal infections of the cornea are particularly prone to develop coincidentally with long-term local steroid application. Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use. Fungal culture should be taken when appropriate.
6 ADVERSE REACTIONS
Adverse reactions associated with ophthalmic steroids include elevated intraocular pressure, which may be associated with optic nerve damage, visual acuity and field defects, posterior subcapsular cataract formation, secondary ocular infection from pathogens including herpes simplex, and perforation of the globe where there is thinning of the cornea or sclera.
Ocular adverse reactions occurring in 5-15% of subjects in clinical studies with Durezol included corneal edema, ciliary and conjunctival hyperemia, eye pain, photophobia, posterior capsule opacification, anterior chamber cells, anterior chamber _are, conjunctival edema, and blepharitis. Other ocular adverse reactions occurring in 1-5% of subjects included reduced visual acuity, punctate keratitis, eye inflammation, and iritis. Ocular adverse events occurring in < 1% of subjects included application site discomfort or irritation, corneal pigmentation and striae, episcleritis, eye pruritis, eyelid irritation and crusting, foreign body sensation, increased lacrimation, macular edema, scleral hyperemia, and uveitis. Most of these events may have been the consequence of the surgical procedure.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects
Pregnancy Category C. Difluprednate has been shown to be embryotoxic (decrease in embryonic body weight and a delay in embryonic ossification) and teratogenic (cleft palate and skeletal) anomalies when administered subcutaneously to rabbits during organogenesis at a dose of 1-10 μg/kg/day. The no-observed-effect-level (NOEL) for these effects was 1 μg/kg/day, and 10 μg/kg/day was considered to be a teratogenic dose that was concurrently found in the toxic dose range for fetuses and pregnant females. Treatment of rats with 10 μg/kg/day subcutaneously during organogenesis did not result in any reproductive toxicity, nor was it maternally toxic. At 100 μg/kg/day after subcutaneous administration in rats, there was a decrease in fetal weights and delay in ossification, and effects on weight gain in the pregnant females. It is difficult to extrapolate these doses of difluprednate to maximum daily human doses of Durezol, since Durezol is administered topically with minimal systemic absorption, and difluprednate blood levels were not measured in the reproductive animal studies. However, since use of difluprednate during human pregnancy has not been evaluated and cannot rule out the possibility of harm, Durezol should be used during pregnancy only if the potential benefit justifies the potential risk to the embryo or fetus.
8.3 Nursing Mothers
It is not known whether topical ophthalmic administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Caution should be exercised when Durezol is administered to a nursing woman.
11 DESCRIPTION
Durezol (difluprednate ophthalmic emulsion) 0.05% is a sterile, topical anti-inflammatory corticosteroid for ophthalmic use. The chemical name is 6α,9difluoro-11β,17,21-trihydroxypregna-1,4-diene-3,20-dione 21-acetate 17-butyrate (CAS number 23674-86-4). Difluprednate is represented by the following structural formula:
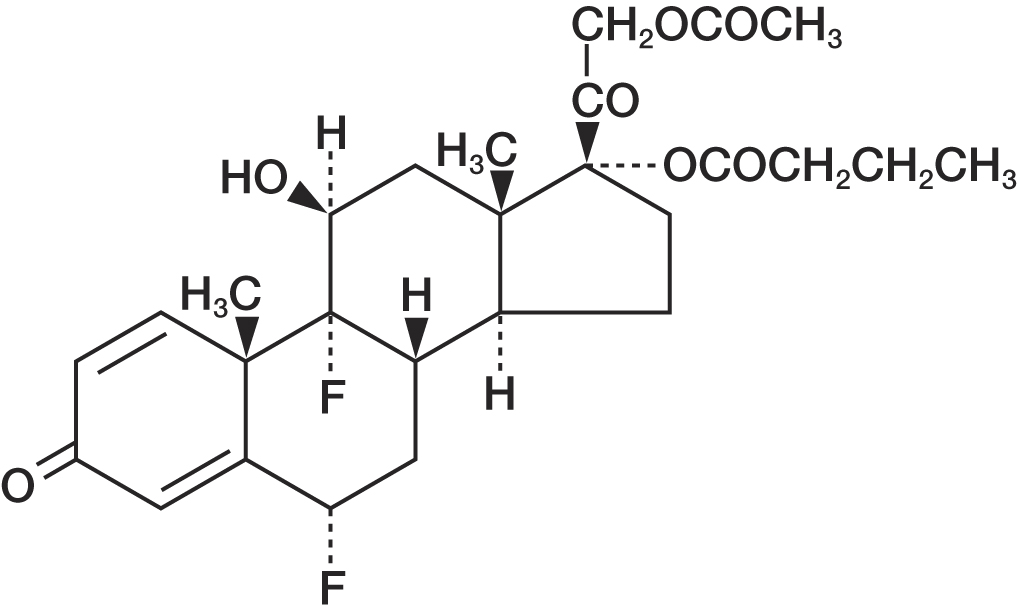
Difluprednate has a molecular weight of 508.56, and the empirical formula is C27H34F2O7.
Each mL contains: ACTIVE: difluprednate 0.5 mg (0.05%); INACTIVE: boric acid, castor oil, glycerin, polysorbate 80, water for injection, sodium acetate, sodium EDTA, sodium hydroxide (to adjust the pH to 5.2 to 5.8). The emulsion is essentially isotonic with a tonicity of 304 to 411 mOsm/kg. PRESERVATIVE: sorbic acid 0.1%.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids inhibit the inflammatory response to a variety of inciting agents that may delay or slow healing. They inhibit edema, fibrin deposition, capillary dilation, leukocyte migration, capillary proliferation, fibroblast proliferation, deposition of collagen, and scar formation associated with inflammation. There is no generally accepted explanation for the mechanism of action of ocular corticosteroids. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of in_ammation such as prostaglandins and leukotreines by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
Difluprednate is structurally similar to other corticosteroids.
12.3 Pharmacokinetics
Difluprednate undergoes deacetylation in vivo to 6α, 9-difluoroprednisolone 17-butyrate (DFB), an active metabolite of difluprednate.
Clinical pharmacokinetic studies of difluprednate after repeat ocular instillation of 2 drops of difluprednate (0.01% or 0.05%) QID for 7 days showed that DFB levels in blood were below the quantification limit (50 ng/mL) at all time points for all subjects, indicating the systemic absorption of difluprednate after ocular instillation of Durezol is limited.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
Difluprednate was not genotoxic in vitro in the Ames test, and in cultured mammalian cells CHL/IU (a fibroblastic cell line derived from the lungs of newborn female Chinese hamsters). An in vivo micronucleus test of difluprednate in mice was also negative. Treatment of male and female rats with subcutaneous difluprednate up to 10 μg/kg/day prior to and during mating did not impair fertility in either gender. Long term studies have not been conducted to evaluate the carcinogenic potential of difluprednate.
13.2 Animal Toxicology and/or Pharmacology
In multiple studies performed in rodents and non-rodents, subchronic and chronic toxicity tests of difluprednate showed systemic effects such as suppression of body weight gain; a decrease in lymphocyte count; atrophy of the lymphatic glands and adrenal gland; and for local effects, thinning of the skin; all of which were due to the pharmacologic action of the molecule and are well known glucocorticosteroid effects. Most, if not all of these effects were reversible after drug withdrawal. The NOEL for the subchronic and chronic toxicity tests were consistent between species and ranged from 1-1.25 μg/kg per day.
14 CLINICAL STUDIES
14.1 Postoperative Ocular Inflammation and Pain
Clinical efficacy was evaluated in 2 randomized, double-masked, placebo-controlled trials in which subjects with an anterior chamber cell grade ≥ "2" (a cell count of 11 or higher) after cataract surgery were assigned to Durezol or placebo (vehicle) following surgery. One drop of Durezol or vehicle was self instilled either 2 (BID) or 4 (QID) times per day for 14 days, beginning the day after surgery. The presence of complete clearing (a cell count of 0) was assessed 8 and 15 days post-surgery using a slit lamp binocular microscope. In the intent-to-treat analyses of both studies, a signifocant benefit was seen in the QID Durezol-treated group in ocular inflammation and reduction of pain when compared with placebo. The consolidated clinical trial results are provided below.
| Ocular Inflammation and Pain Endpoints (Studies Pooled) | ||||
|---|---|---|---|---|
| Durezol QID N = 107 | Vehicle
N = 220 |
|||
| Day | 8 | 15 | 8 | 15 |
| Anterior Chamber cell clearing (% subjects) | 24 (22%)* | 44 (41%)* | 17 (7%) | 25 (11%) |
| Pain free (% subjects) | 62 (58%)* | 67 (63%)* | 59 (27%) | 76 (35%) |
| * Statistically significantly better than vehicle, p<0.01 | ||||
16 HOW SUPPLIED/STORAGE AND HANDLING
Durezol (difluprednate ophthalmic emulsion) 0.05% is a sterile, aqueous topical ophthalmic emulsion supplied in an opaque plastic bottle with a controlled drop tip and a pink cap in the following size:
5 mL in a 5 mL bottle NDC 54868-6296-0.
Storage
Store at 15-25°C (59-77°F). Do not freeze. Protect from light. When not in use keep the bottles in the protective carton.
17 PATIENT COUNSELING INFORMATION
This product is sterile when packaged. Patients should be advised not to allow the dropper tip to touch any surface, as this may contaminate the emulsion. If pain develops or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult a physician. As with all ophthalmic preparations containing a preservative, patients should be advised not to wear contact lenses when using Durezol.
Revised: August 2010
U.S. Patent: 6,114,319
©2010 Alcon , Inc. ®
Manufactured for:
ALCON®
Alcon Laboratories, Inc.
6201 South Freeway
Fort Worth, Texas 76134 USA
1-800-757-9195
MedInfo@AlconLabs.com
Manufactured By:
Catalent Pharma Solutions
Woodstock, IL 60098
9005602-0810
