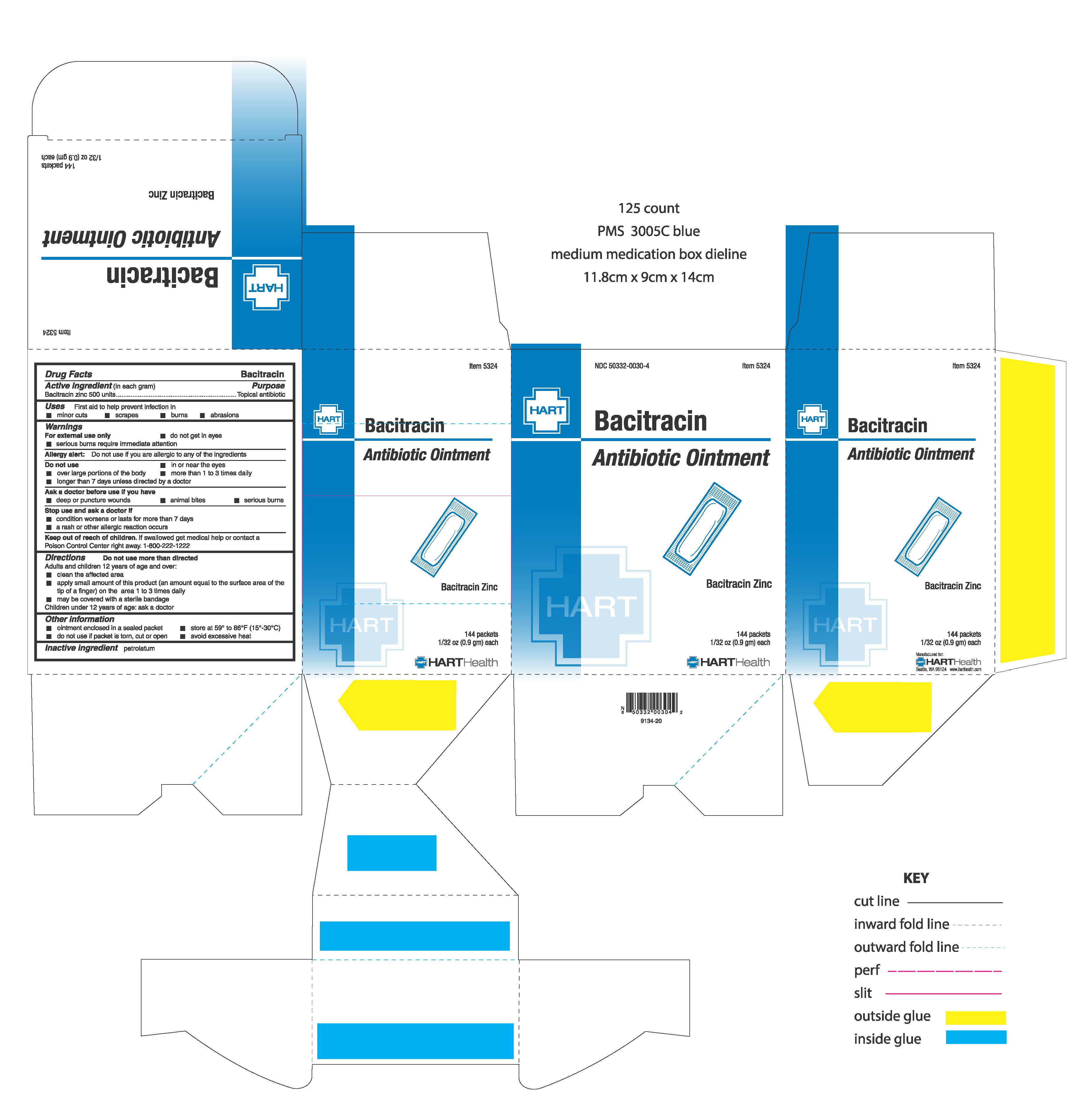
Warnings: For external use only
- do not get in eyes
- serious burns require immediate attention
Allergy alert: do not use if you are allergic to any of the ingredients
Do not use:
- in or near the eyes
- over large portions of the body
- more than 1 to 3 times daily
- longer than 7 days unless directed by a doctor
Stop use and ask a doctor if
- condition worsens or lasts for more than 7 days
- a rash or other allergic reaction occurs
Keep out of reach of children. If swallowed get medical help or contact a Poison Control Center right away. 1-800-222-1222.
Directions: do not use more than directed
Adults and children 12 years of age and over:
- clean the affected area
- apply small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Children under 12 years of age: ask a doctor