Active ingredient(s)
Loratadine 10mg
Use(s)
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Do not use
if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if
or kidney disease. Your doctor should determine if you need a different dose.
When using this product
do not take more than directed. Taking more than directed may cause drowsiness.
Stop use and ask a doctor if
if an allergic reaction to this product occurs. Seek medical help right away.
Keep out of reach of children
In case of accidental overdose, contact a doctor or Poison Control Center (1-800-222-1222) right away.
Directions
Adults and children 6 years and over 1 tablet daily; not more than 1 tablet in 24 hours
Children under 6 years of age ask a doctor
Consumers with liver or kidney disease ask a doctor
Other information
- do not use if blister unit is torn or
Storage
- store between 20° to 25°C (68° to 77°F)
- protect from excessive moisture
Inactive ingredients
lactose monohydrate, magnesium stearate, pregelatinized starch, sodium starch glycolate
Questions
1-800-327-2704 Monday - Friday 8:00am - 5:00pm (CDT)
Principal Display Panel
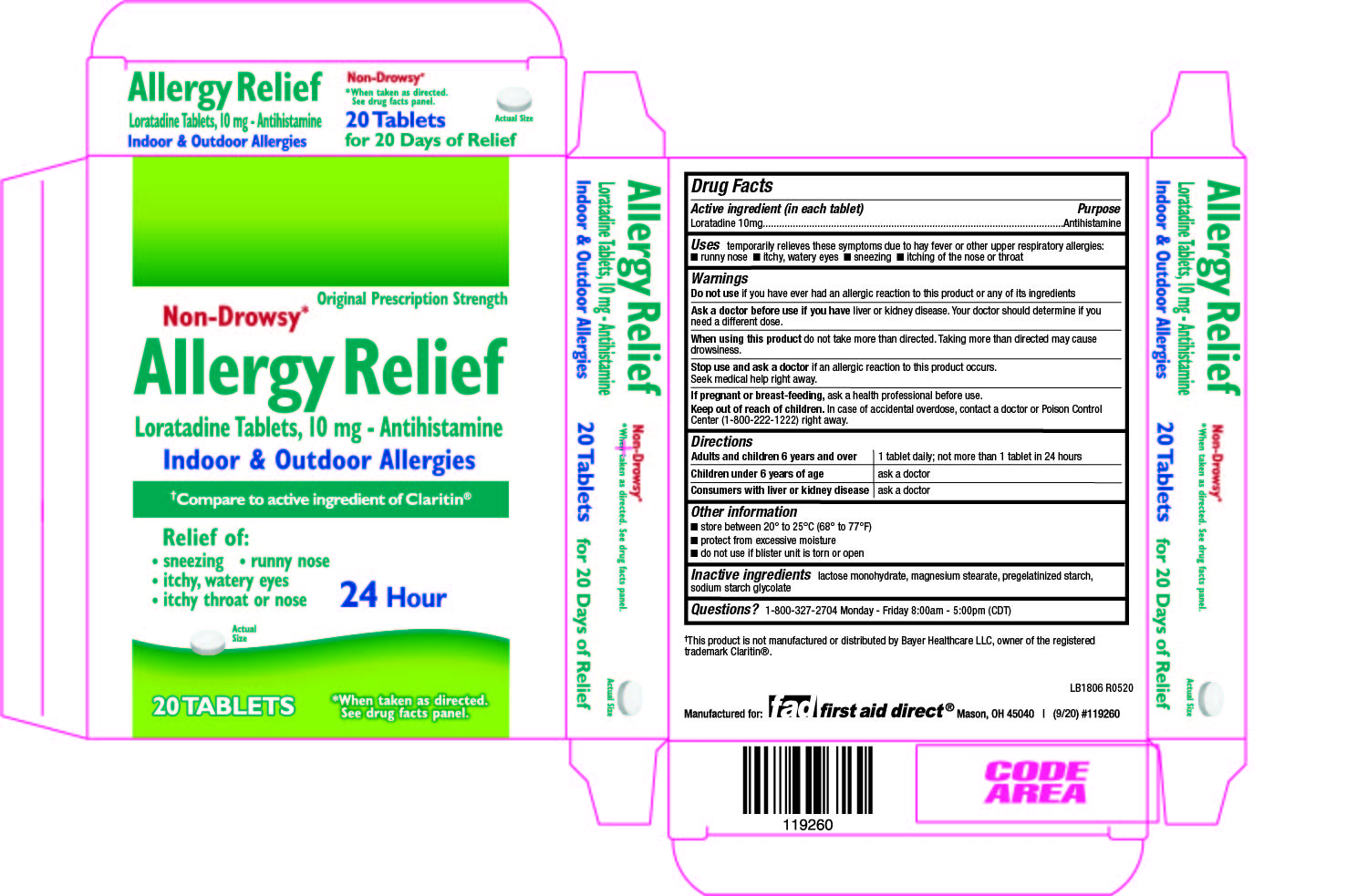
Allergy Relief Loratadine
