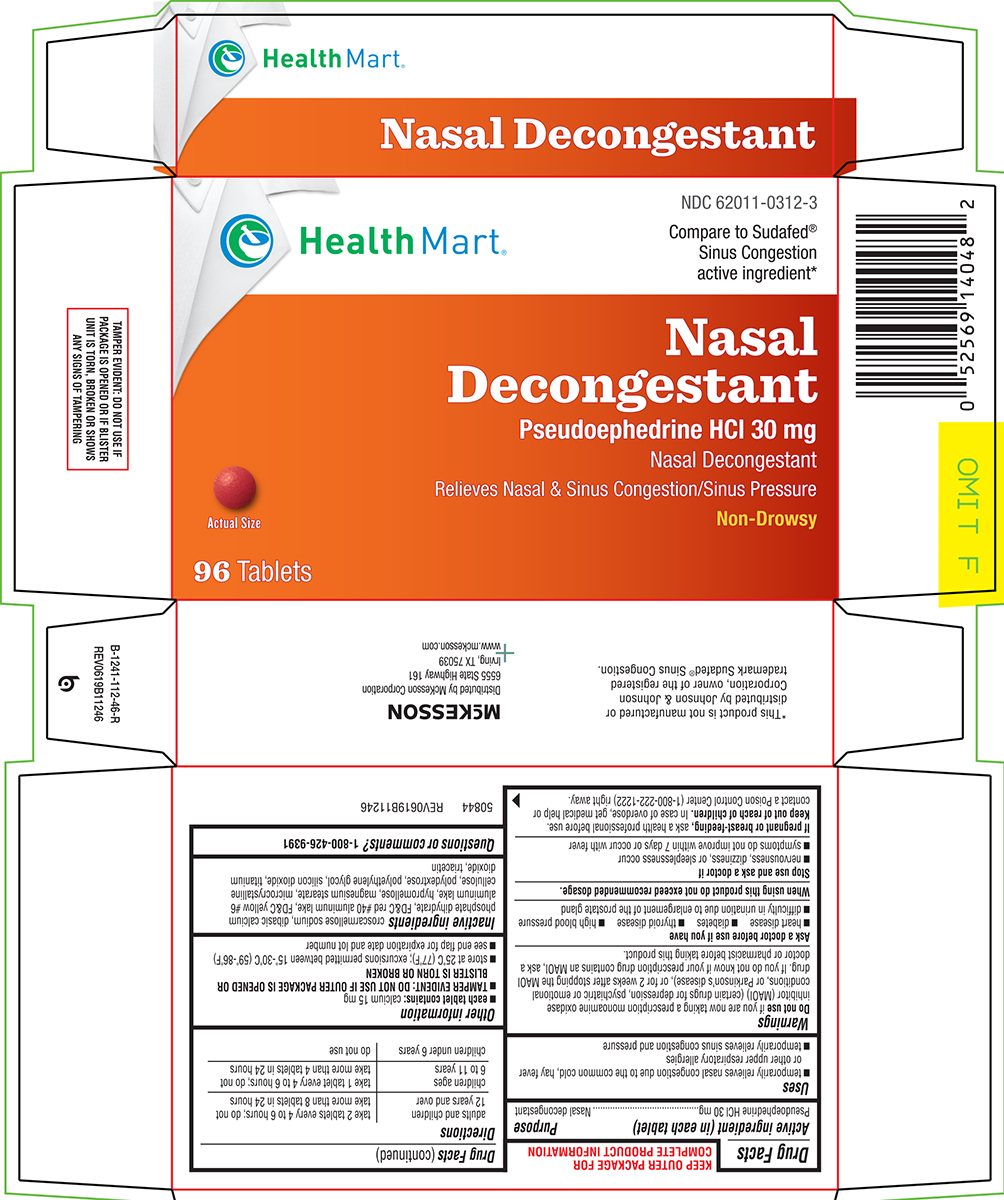
Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- diabetes
- heart disease
- high blood pressure
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
Directions
| adults and children 12 years and over | take 2 tablets every 4 to 6 hours; do not take more than 8 tablets in 24 hours |
| children ages 6 to 11 years | take 1 tablet every 4 to 6 hours; do not take more than 4 tablets in 24 hours |
| children under 6 years | do not use |
Other information
- each tablet contains: calcium 15 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients
croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, silicon dioxide, titanium dioxide, triacetin
Principal display panel
Health Mart®
NDC 62011-0312-3
Compare to Sudafed®
Sinus Congestion
active ingredient*
Nasal
Decongestant
Pseudoephedrine HCl 30 mg
Nasal Decongestant
Relieves Nasal & Sinus Congestion/Sinus Pressure
Non-Drowsy
Actual Size
96 Tablets
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERING
*This product is not manufactured or
distributed by Johnson & Johnson
Corporation, owner of the registered
trademark Sudafed® Congestion.
50844 REV0619B11246
Mckesson
Distributed by McKesson Corporation
6555 State Highway 161
Irving, TX 75039
www.mckesson.com
Healthmart 44-112