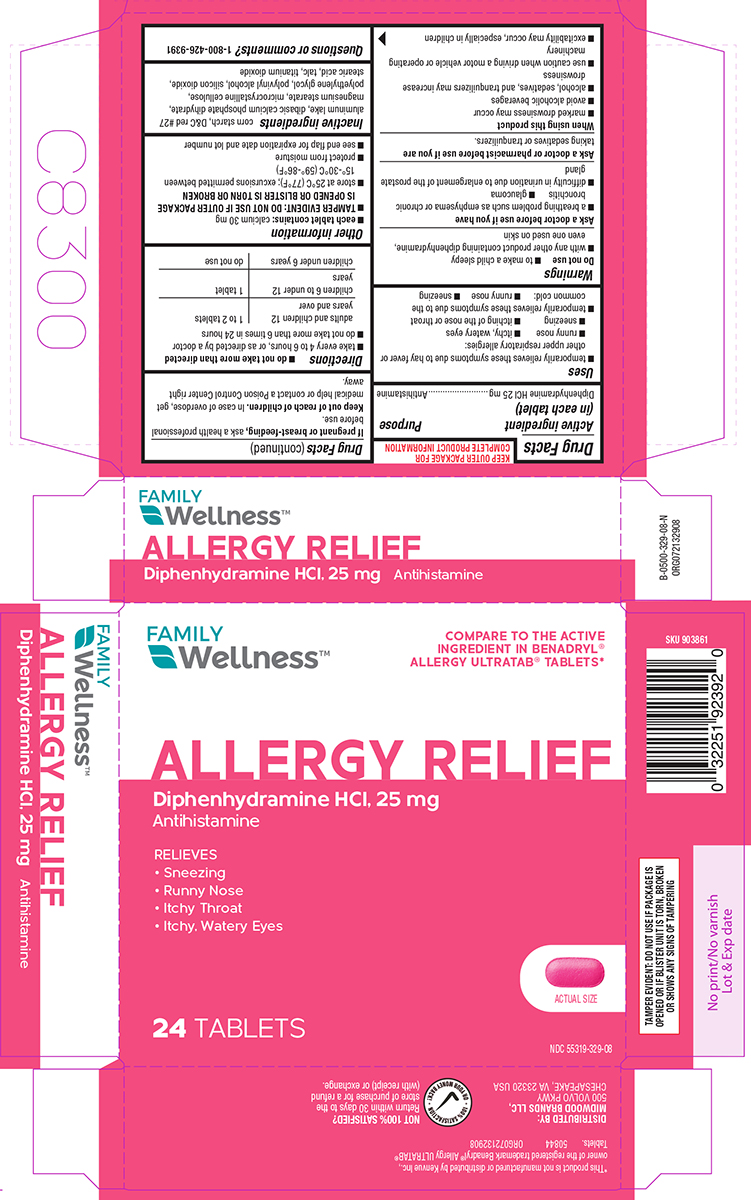
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Directions
- do not take more than directed
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years | 1 tablet |
| children under 6 years | do not use |
Other information
- each tablet contains: calcium 30 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15°-30°C (59°-86°F)
- protect from moisture
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, D&C red #27 aluminum lake, dicalcium phosphate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, stearic acid, talc, titanium dioxide
Principal Display Panel
FAMILY
Wellness™
COMPARE TO THE ACTIVE
INGREDIENT IN BENADRYL®
ALLERGY ULTRATAB® TABLETS*
ALLERGY RELIEF
Diphenhydramine HCl, 25 mg
Antihistamine
RELIEVES
• Sneezing
• Runny Nose
• Itchy Throat
• Itchy, Watery Eyes
48 TABLETS
ACTUAL SIZE
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING
DISTRIBUTED BY:
MIDWOOD BRANDS, LLC,
500 VOLVO PKWY
CHESAPEAKE, VA 23320 USA
NOT 100% SATISFIED?
Return within 30 days to the
store of purchase for a refund
(with receipt) or exchange.
*This product is not manufactured or distributed by Kenvue Inc.,
owner of the registered trademark Benadryl® Allergy ULTRATAB®
Tablets. 50844 ORG072132908
Family Wellness 44-329