Use
- •
- treats frequent heartburn (occurs 2 or more days a week)
- •
- not intended for immediate relief of heartburn; this drug may take 1 to 4 days for full effect
Warnings
Allergy alert: Do not use if you are allergic to omeprazole
Do not use if you have:
- •
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- •
- heartburn with lightheadedness, sweating or dizziness
- •
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- •
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have:
- •
- had heartburn over 3 months. This may be a sign of a more serious condition.
- •
- frequent wheezing, particularly with heartburn
- •
- unexplained weight loss
- •
- nausea or vomiting
- •
- stomach pain
Ask a doctor or pharmacist before use if you are
taking a prescription drug. Acid reducers may interact with certain prescription drugs.
Directions
- •
- for adults 18 years of age and older
- •
- this product is to be used once a day (every 24 hours), every day for 14 days
- •
- it may take 1 to 4 days for full effect; some people get complete relief of symptoms within 24 hours
14-Day Course of Treatment
- •
- swallow 1 tablet with a glass of water before eating in the morning
- •
- take every day for 14 days
- •
- do not take more than 1 tablet a day
- •
- do not use for more than 14 days unless directed by your doctor
- •
- swallow whole. Do not chew or crush tablets
Repeated 14-Day Courses (if needed)
- •
- you may repeat a 14-day course every 4 months
- •
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- •
- children under 18 years of age: ask a doctor. Heartburn in children may sometimes be caused by a serious condition.
Other information
- •
- read the directions and warnings before use
- •
- keep the carton. It contains important information.
- •
- store at 20-25°C (68-77°F) and protect from moisture
Inactive ingredients
carnauba wax, ferric oxide red, ferric oxide yellow, hypromellose, hypromellose acetate succinate, lactose monohydrate, monoethanolamine, propylene glycol, sodium lauryl sulfate, sodium starch glycolate, sodium stearate, sodium stearyl fumarate, talc, titanium dioxide, triethyl citrate
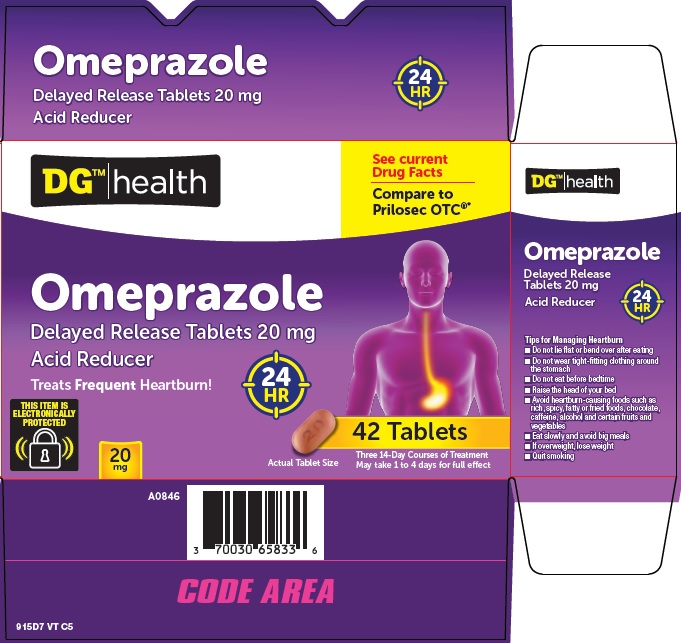
Principal Display Panel
See current Drug Facts
Compare to Prilosec OTC®
Omeprazole
Delayed Release Tablets 20 mg
Acid Reducer
Treats Frequent Heartburn!
24 HR
THIS ITEM IS ELECTRONICALLY PROTECTED
20 mg
42 Tablets
Actual Tablet Size
Three 14-Day Courses of Treatment
May take 1 to 4 days for full effect