FULL PRESCRIBING INFORMATION
WARNING: SECONDARY EXPOSURE TO TESTOSTERONE
- Virilization has been reported in children who were secondarily exposed to testosterone gel [see Warnings and Precautions (5.2) and Adverse Reactions (6.2)].
- Children should avoid contact with unwashed or unclothed application sites in men using testosterone gel [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)].
- Healthcare providers should advise patients to strictly adhere to recommended instructions for use [see Dosage and Administration (2.2), Warnings and Precautions (5.2) and Patient Counseling Information (17)].
1 INDICATIONS AND USAGE
Vogelxo is indicated for replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous testosterone:
- Primary hypogonadism (congenital or acquired): testicular failure due to cryptorchidism, bilateral torsion, orchitis, vanishing testis syndrome, orchiectomy, Klinefelter's syndrome, chemotherapy, or toxic damage from alcohol or heavy metals. These men usually have low serum testosterone levels and gonadotropins (follicle-stimulating hormone [FSH], luteinizing hormone [LH]) above the normal range.
- Hypogonadotropic hypogonadism (congenital or acquired): gonadotropin or luteinizing hormone-releasing hormone (LHRH) deficiency or pituitary-hypothalamic injury from tumors, trauma, or radiation. These men have low testosterone serum levels but have gonadotropins in the normal or low range.
Limitations of Use:
- Safety and efficacy of Vogelxo in men with "age-related hypogonadism" (also referred to as "late-onset hypogonadism") have not been established.
- Safety and efficacy of Vogelxo in males less than 18 years old have not been established [see Use in Specific Populations (8.4)].
- Topical testosterone products may have different doses, strengths, or application instructions that may result in different systemic exposure [see Dosage and Administration (2) and Clinical Pharmacology (12.3)].
2 DOSAGE AND ADMINISTRATION
Prior to initiating Vogelxo, confirm the diagnosis of hypogonadism by ensuring that serum testosterone concentrations have been measured in the morning on at least two separate days and that these serum testosterone concentrations are below the normal range.
2.1 Dosing and Dose Adjustment
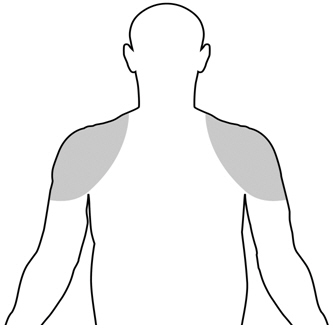
The recommended starting dose of Vogelxo is 50 mg of testosterone (one tube, one packet, or 4 pump actuations) applied topically once daily at approximately the same time each day to clean, dry intact skin of the shoulders and/or upper arms.
Dose Adjustment
To ensure proper dosing, serum testosterone concentrations should be measured. Morning, pre-dose serum testosterone concentrations should be measured approximately 14 days after initiation of therapy to ensure proper serum testosterone concentrations are achieved. If the serum testosterone concentration is below the normal range (300 ng/dL to 1,000 ng/dL), the daily Vogelxo dose may be increased from 50 mg testosterone (one tube, one packet, or 4 pump actuations) to 100 mg of testosterone (two tubes, two packets, or 8 pump actuations) once daily.
The maximum recommended dose of Vogelxo is 100 mg once daily.
2.2 Administration Instructions
Unit-Dose Tube or Packet
Upon opening the tube or packet the entire contents should be squeezed into the palm of the hand and immediately applied to the shoulders and/or upper arms (area of application should be limited to the area that will be covered by the patient's short sleeve t-shirt [see figure below]).
Table 1 has specific dosing guidelines for when the unit-dose tubes or packets are used.
| Prescribed Daily Dose | Number of Unit-Dose Tubes or Packets | Application Method |
|---|---|---|
| 50 mg testosterone | One unit-dose tube or packet (once daily) | Apply one unit-dose tube or packet to one upper arm and shoulder. |
| 100 mg testosterone | Two unit-dose tubes or packets (once daily) | Apply one unit-dose tube or packet to one upper arm and shoulder and then apply one unit-dose tube or packet to the opposite upper arm and shoulder. |
Multi-Dose Metered Pump
Patients should be instructed to prime the pump before using it for the first time by fully depressing the pump mechanism (actuation) 3 times and discard this portion of the product to assure precise dose delivery. After the priming procedure, patients should completely depress the pump one time (actuation) for every 12.5 mg of testosterone required to achieve the daily prescribed dosage. Table 2 has specific dosing guidelines for when the metered pump is used.
| Prescribed Daily Dose | Number of Pump Actuations | Application Method |
|---|---|---|
| 50 mg testosterone | 4 (once daily) | Apply 4 pump actuations to one upper arm and shoulder |
| 100 mg testosterone | 8 (once daily) | Apply 4 pump actuations to one upper arm and shoulder and then apply 4 pump actuations to the opposite upper arm and shoulder |
The prescribed amount of product should be delivered directly into the palm of the hand and immediately applied to the shoulders and/or upper arms (area of application should be limited to the area that will be covered by the patient's short sleeve t-shirt [see figure below]).
Do not apply Vogelxo to the genitals or to the abdomen.
Application sites should be allowed to dry completely prior to dressing.
Hands should be washed thoroughly with soap and water after Vogelxo has been applied.
Avoid fire, flame or smoking during the application of Vogelxo until the Vogelxo has dried [see Warnings and Precautions (5.2, 5.16)].
In order to prevent transfer to another person, clothing should be worn to cover the application site(s). If direct skin-to-skin contact of the application site(s) with another person is anticipated, the application site(s) must be washed thoroughly with soap and water [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
The patient should avoid swimming or showering or washing the administration site for a minimum of 2 hours after application [see Clinical Pharmacology (12.3)].
Strict adherence to the following precautions is advised in order to minimize the potential for secondary exposure to testosterone from Vogelxo treated skin:
- Children and women should avoid contact with unwashed or unclothed application site(s) of men using Vogelxo.
- Vogelxo should only be applied to the upper arms and shoulders. The area of application should be limited to the area that will be covered by a short sleeve t-shirt.
- Patients should wash their hands with soap and water immediately after applying Vogelxo.
- Patients should cover the application site(s) with clothing (e.g., a t-shirt) after the gel has dried.
- Prior to any situation in which direct skin-to-skin contact of the application site(s) with another person is anticipated, patients should wash the application site(s) thoroughly with soap and water to remove any testosterone residue.
- In the event that unwashed or unclothed skin to which Vogelxo has been applied comes in direct contact with the skin of another person, the general area of contact on the other person should be washed with soap and water as soon as possible.
3 DOSAGE FORMS AND STRENGTHS
Vogelxo (testosterone) gel is a clear to translucent hydroalcoholic topical gel for topical use available in unit-dose tubes, unit-dose packets, and multiple-dose metered pumps. Each tube or packet provides 50 mg testosterone in 5 g of gel. One pump actuation delivers 12.5 mg testosterone in 1.25 g of gel (4 actuations = 50 mg testosterone).
4 CONTRAINDICATIONS
- Vogelxo is contraindicated in men with carcinoma of the breast or known or suspected carcinoma of the prostate [see Warnings and Precautions (5.1)].
- Vogelxo is contraindicated in women who are pregnant. Vogelxo can cause virilization of the female fetus when administered to a pregnant woman. Pregnant women need to be aware of the potential for skin transfer of testosterone from men treated with Vogelxo. If a pregnant woman is exposed to Vogelxo, she should be apprised of the potential hazard to the fetus [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1)].
5 WARNINGS AND PRECAUTIONS
5.1 Worsening of Benign Prostatic Hyperplasia (BPH) and Potential Risk of Prostate Cancer
- Men with BPH treated with androgens are at an increased risk for worsening of signs and symptoms of BPH. Monitor patients with BPH for worsening signs and symptoms.
- Patients treated with androgens may be at increased risk for prostate cancer. Evaluate patients for prostate cancer prior to initiating and during treatment with androgens [see Contraindications (4)].
5.2 Potential for Secondary Exposure to Testosterone
Cases of secondary exposure resulting in virilization of children have been reported in postmarketing surveillance. Signs and symptoms have included enlargement of the penis or clitoris, development of pubic hair, increased erections and libido, aggressive behavior, and advanced bone age. In most cases, these signs and symptoms regressed with removal of the exposure to testosterone gel. In a few cases, however, enlarged genitalia did not fully return to age-appropriate normal size, and bone age remained modestly greater than chronological age. The risk of transfer was increased in some of these cases by not adhering to precautions for the appropriate use of the topical testosterone product. Children and women should avoid contact with unwashed or unclothed application sites in men using Vogelxo [see Dosage and Administration (2.2), Use in Specific Populations (8.1) and Clinical Pharmacology (12.3)].
Inappropriate changes in genital size or development of pubic hair or libido in children, or changes in body hair distribution, significant increase in acne, or other signs of virilization in adult women should be brought to the attention of a physician and the possibility of secondary exposure to testosterone gel should also be brought to the attention of a physician. Testosterone gel should be promptly discontinued until the cause of virilization has been identified.
5.3 Polycythemia
Increases in hematocrit, reflective of increases in red blood cell mass, may require lowering or discontinuation of testosterone. Check hematocrit prior to initiating treatment. It would also be appropriate to re-evaluate the hematocrit 3 to 6 months after starting treatment, and then annually. If hematocrit becomes elevated, stop therapy until hematocrit decreases to an acceptable concentration. An increase in red blood cell mass may increase the risk of thromboembolic events.
5.4 Venous Thromboembolism
There have been postmarketing reports of venous thromboembolic events, including deep vein thrombosis (DVT) and pulmonary embolism (PE), in patients using testosterone products, such as Vogelxo. Evaluate patients who report signs and symptoms of pain, edema, warmth and erythema in the lower extremity for DVT and those who present with acute shortness of breath for PE. If a venous thromboembolic event is suspected, discontinue treatment with Vogelxo and initiate appropriate workup and management [see Adverse Reactions (6.2)].
5.5 Cardiovascular Risk
Long term clinical safety trials have not been conducted to assess the cardiovascular outcomes of testosterone replacement therapy in men. To date, epidemiologic studies and randomized controlled trials have been inconclusive for determining the risk of major adverse cardiovascular events (MACE), such as non-fatal myocardial infarction, non-fatal stroke, and cardiovascular death, with the use of testosterone compared to non-use. Some studies, but not all, have reported an increased risk of MACE in association with use of testosterone replacement therapy in men. Patients should be informed of this possible risk when deciding whether to use or to continue to use Vogelxo.
5.6 Abuse of Testosterone and Monitoring of Serum Testosterone Concentrations
Testosterone has been subject to abuse, typically at doses higher than recommended for the approved indication and in combination with other anabolic androgenic steroids. Anabolic androgenic steroid abuse can lead to serious cardiovascular and psychiatric adverse reactions [see Drug Abuse and Dependence (9)].
If testosterone abuse is suspected, check serum testosterone concentrations to ensure they are within therapeutic range. However, testosterone levels may be in the normal or subnormal range in men abusing synthetic testosterone derivatives. Counsel patients concerning the serious adverse reactions associated with abuse of testosterone and anabolic androgenic steroids. Conversely, consider the possibility of testosterone and anabolic androgenic steroid abuse in suspected patients who present with serious cardiovascular or psychiatric adverse events.
5.7 Use in Women
Due to lack of controlled evaluations in women and potential virilizing effects, Vogelxo is not indicated for use in women [see Contraindications (4) and Use in Specific Populations (8.1, 8.2)].
5.8 Potential for Adverse Effects on Spermatogenesis
With large doses of exogenous androgens, including Vogelxo, spermatogenesis may be suppressed through feedback inhibition of pituitary follicle-stimulating hormone (FSH) which could possibly lead to adverse effects on semen parameters including sperm count.
5.9 Hepatic Adverse Effects
Prolonged use of high doses of orally active 17-alpha-alkyl androgens (e.g., methyltestosterone) has been associated with serious hepatic adverse effects (peliosis hepatis, hepatic neoplasms, cholestatic hepatitis, and jaundice). Peliosis hepatis can be a life-threatening or fatal complication. Long-term therapy with intramuscular testosterone enanthate, which elevate blood levels for prolonged periods, has produced multiple hepatic adenomas. Vogelxo is not known to produce these adverse effects. Nonetheless, patients should be instructed to report any signs or symptoms of hepatic dysfunction (e.g., jaundice). If these occur, promptly discontinue Vogelxo while the cause is evaluated.
5.10 Edema
Androgens, including Vogelxo, may promote retention of sodium and water. Edema, with or without congestive heart failure, may be a serious complication in patients with preexisting cardiac, renal, or hepatic disease. In addition to discontinuation of the drug, diuretic therapy may be required.
5.11 Gynecomastia
Gynecomastia occasionally develops and occasionally persists in patients being treated for hypogonadism [see Adverse Reactions (6.1)].
5.12 Sleep Apnea
The treatment of hypogonadal men with testosterone may potentiate sleep apnea in some patients, especially those with risk factors such as obesity or chronic lung diseases.
5.13 Lipids
Changes in the serum lipid profile may occur. Monitor the lipid profile periodically, particularly after starting testosterone therapy and after any dose increases.
5.14 Hypercalcemia
Androgens, including Vogelxo, should be used with caution in cancer patients at risk of hypercalcemia (and associated hypercalciuria). Regular monitoring of serum calcium concentrations is recommended in these patients.
5.15 Decreased Thyroxine-binding Globulin
Androgens, including Vogelxo, may decrease concentrations of thyroxine-binding globulins, resulting in decreased total T4 serum concentrations and increased resin uptake of T3 and T4. Free thyroid hormone levels remain unchanged, however, and there is no clinical evidence of thyroid dysfunction.
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In a controlled clinical study, 304 patients were treated with testosterone gel 50 mg or 100 mg or placebo gel for up to 90 days. Two hundred-five (205) patients received testosterone gel 50 mg or 100 mg daily and 99 patients received placebo. Subjects could be counted in both testosterone gel treatment groups if they received both 50 mg and 100 mg at different points in the study and experienced an adverse reaction at both dose levels. Adverse reactions reported by ≥1% of the testosterone gel patients and greater than placebo are listed in Table 3.
| Testosterone Gel 50 mg | Testosterone Gel 100 mg | Placebo | |
|---|---|---|---|
| Event | (n=103) | (n=149) | (n=99) |
| Application Site Reactions | 2% | 4% | 3% |
| Blood Pressure Increased | 1% | 1% | 0% |
| Gynecomastia | 1% | 0% | 0% |
| Headache | 1% | 1% | 0% |
| Hematocrit/Hemoglobin Increased | 1% | 2% | 0% |
| Hot Flushes | 1% | 0% | 0% |
| Insomnia | 1% | 0% | 0% |
| Mood Swings | 1% | 0% | 0% |
| Smell Disorder | 1% | 0% | 0% |
| Spontaneous Penile Erection | 1% | 0% | 0% |
| Taste Disorder | 1% | 1% | 0% |
The following adverse reactions occurred in fewer than 1% of patients but were greater in testosterone gel groups compared to the placebo group: activated partial thromboplastin time prolonged, blood creatinine increased, prothrombin time prolonged, appetite increased, sensitive nipples, and acne.
In this clinical trial of testosterone gel, six patients had adverse events that led to their discontinuation. These events included: depression with suicidal ideation, urinary tract infection, mood swings and hypertension. No testosterone gel patients discontinued due to skin reaction. In one foreign Phase 3 trial, one subject discontinued due to a skin-related adverse event.
In the pivotal U.S. and European Phase 3 trials combined, at the 50 mg dosage strength, the percentage of subjects reporting clinically notable increases in hematocrit or hemoglobin were similar to placebo. However, in the 100 mg dose group, 2.3% and 2.8% of patients had a clinically notable increase in hemoglobin (≥ 19 g/dL) or hematocrit (≥ 58%), respectively, compared to 1.0% and 1.5% of patients in the placebo group, respectively.
In the combined U.S. and European open label extension studies, approximately 140 patients received testosterone gel for at least 6 months. The results from these studies are consistent with those reported for the U.S. controlled clinical trial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of testosterone gel products. Because the reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Secondary Exposure to Testosterone in Children
Cases of secondary exposure to testosterone resulting in virilization of children have been reported in postmarketing surveillance of testosterone gel products. Signs and symptoms of these reported cases have included enlargement of the clitoris (with surgical intervention) or of the penis, development of pubic hair, increased erections and libido, aggressive behavior, and advanced bone age. In most cases with a reported outcome, these signs and symptoms were reported to have regressed with removal of the testosterone gel exposure. In a few cases, however, enlarged genitalia did not fully return to age-appropriate normal size, and bone age remained modestly greater than chronological age. In some of the cases, direct contact with the sites of application on the skin of men using testosterone gel was reported. In at least one reported case, the reporter considered the possibility of secondary exposure from items such as the testosterone gel user's shirts and/or other fabrics, such as towels and sheets [see Warnings and Precautions (5.2)].
7 DRUG INTERACTIONS
7.1 Insulin
Changes in insulin sensitivity or glycemic control may occur in patients treated with androgens. In diabetic patients, the metabolic effects of androgens may decrease blood glucose and, therefore, may necessitate a decrease in the dose of anti-diabetic medication.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Vogelxo is contraindicated in pregnant women. Testosterone is teratogenic and may cause fetal harm when administered to a pregnant woman based on data from animal studies and its mechanism of action [see Contraindications (4) and Clinical Pharmacology (12.1)]. Exposure of a female fetus to androgens may result in varying degrees of virilization. In animal developmental studies, exposure to testosterone in utero resulted in hormonal and behavioral changes in offspring and structural impairments of reproductive tissues in female and male offspring. These studies did not meet current standards for nonclinical development toxicity studies.
Data
Animal Data
In developmental studies conducted in rats, rabbits, pigs, sheep and rhesus monkeys, pregnant animals received intramuscular injection of testosterone during the period of organogenesis. Testosterone treatment at doses that were comparable to those used for testosterone replacement therapy resulted in structural impairments in both female and male offspring. Structural impairments observed in females included increased ano-genital distance, phallus development, empty scrotum, no external vagina, intrauterine growth retardation, reduced ovarian reserve, and increased ovarian follicular recruitment. Structural impairments seen in male offspring included increased or decreased testicular weight, larger seminal tubular lumen diameter, and higher frequency of occluded tubule lumen. Increased pituitary weight was seen in both sexes.
Testosterone exposure in utero also resulted in hormonal and behavioral changes in offspring. Hypertension was observed in pregnant female rats and their offspring exposed to doses approximately twice those used for testosterone replacement therapy.
8.3 Females and Males of Reproductive Potential
Infertility
During treatment with large doses of exogenous androgens, including Vogelxo, spermatogenesis may be suppressed through feedback inhibition of the hypothalamic-pituitary-testicular axis [see Warnings and Precautions (5.8)]. Reduced fertility is observed in some men taking testosterone replacement therapy. Testicular atrophy, subfertility, and infertility have also been reported in men who abuse anabolic androgenic steroids [see Drug Abuse and Dependence (9.2)]. With either type of use, the impact on fertility may be irreversible.
8.4 Pediatric Use
The safety and efficacy of Vogelxo in pediatric patients less than 18 years old have not been established. Improper use may result in acceleration of bone age and premature closure of epiphyses.
8.5 Geriatric Use
There is insufficient long-term safety data in geriatric patients to assess the potentially increased risks of cardiovascular disease and prostate cancer [see Warnings and Precautions (5.1)].
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Vogelxo contains testosterone, a Schedule III controlled substance in the Controlled Substances Act.
9.2 Abuse
Drug abuse is intentional non-therapeutic use of a drug, even once, for its rewarding psychological and physiological effects. Abuse and misuse of testosterone are seen in male and female adults and adolescents. Testosterone, often in combination with other anabolic androgenic steroids (AAS), and not obtained by prescription through a pharmacy, may be abused by athletes and bodybuilders. There have been reports of misuse by men taking higher doses of legally obtained testosterone than prescribed and continuing testosterone despite adverse events or against medical advice.
Abuse-Related Adverse Reactions
Serious adverse reactions have been reported in individuals who abuse anabolic androgenic steroids and include cardiac arrest, myocardial infarction, hypertrophic cardiomyopathy, congestive heart failure, cerebrovascular accident, hepatotoxicity, and serious psychiatric manifestations, including major depression, mania, paranoia, psychosis, delusions, hallucinations, hostility and aggression.
The following adverse reactions have also been reported in men: transient ischemic attacks, convulsions, hypomania, irritability, dyslipidemias, testicular atrophy, subfertility, and infertility.
The following additional adverse reactions have been reported in women: hirsutism, virilization, deepening of voice, clitoral enlargement, breast atrophy, male-pattern baldness, and menstrual irregularities.
The following adverse reactions have been reported in male and female adolescents: premature closure of bony epiphyses with termination of growth, and precocious puberty.
Because these reactions are reported voluntarily from a population of uncertain size and may include abuse of other agents, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
9.3 Dependence
Behaviors Associated with Addiction
Continued abuse of testosterone and other anabolic steroids, leading to addiction is characterized by the following behaviors:
- Taking greater dosages than prescribed
- Continued drug use despite medical and social problems due to drug use
- Spending significant time to obtain the drug when supplies of the drug are interrupted
- Giving a higher priority to drug use than other obligations
- Having difficulty in discontinuing the drug despite desires and attempts to do so
- Experiencing withdrawal symptoms upon abrupt discontinuation of use
Physical dependence is characterized by withdrawal symptoms after abrupt drug discontinuation or a significant dose reduction of a drug. Individuals taking supratherapeutic doses of testosterone may experience withdrawal symptoms lasting for weeks or months which include depressed mood, major depression, fatigue, craving, restlessness, irritability, anorexia, insomnia, decreased libido and hypogonadotropic hypogonadism.
Drug dependence in individuals using approved doses of testosterone for approved indications has not been documented.
10 OVERDOSAGE
There were no reports of overdose in the testosterone gel clinical trials. There is a single report in the literature of acute overdosage after injection of testosterone enanthate. This subject had serum testosterone concentrations of up to 11,400 ng/dL, which were implicated in a cerebrovascular accident.
Treatment of overdosage would consist of discontinuation of Vogelxo, washing the application site with soap and water, and appropriate symptomatic and supportive care.
11 DESCRIPTION
Vogelxo (testosterone) gel, for topical use is a clear to translucent hydroalcoholic topical gel containing testosterone, an androgen. Vogelxo provides continuous transdermal delivery of testosterone for 24 hours, following a single application to intact, clean, dry skin of the shoulders and/or upper arms.
Vogelxo is available in unit-dose tubes, unit-dose packets, and a metered-dose pump. One 5-g or two 5-g tubes/packets of Vogelxo contains 50 mg or 100 mg of testosterone, respectively. One pump actuation dispenses 1.25 g of gel, which contains 12.5 mg of testosterone. Four pump actuations or eight pump actuations contain 50 mg or 100 mg of testosterone, respectively. Each metered-dose pump container is capable of dispensing 60 pump actuations.
The active pharmacological ingredient in Vogelxo is testosterone. Testosterone USP is a white to practically white crystalline powder chemically described as 17-β hydroxyandrost-4-en-3-one. The structural formula is shown in the following figure:
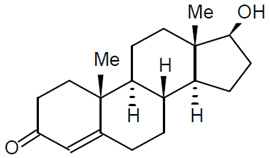
Testosterone (C19H28O2) MW: 288.42
Inactive ingredients in Vogelxo are carbomer copolymer Type B, carbomer homopolymer Type C, diisopropyl adipate, ethyl alcohol, glycerin, methyl laurate, oleyl alcohol, polyethylene glycol, propylene glycol, purified water, and tromethamine.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Endogenous androgens, including testosterone and dihydrotestosterone (DHT) are responsible for the normal growth and development of the male sex organs and for maintenance of secondary sex characteristics. These effects include the growth and maturation of the prostate, seminal vesicles, penis, and scrotum; the development of male hair distribution, such as facial, pubic, chest, and axillary hair; laryngeal enlargement, vocal cord thickening, and alterations in body musculature and fat distribution.
Male hypogonadism, a clinical syndrome resulting from insufficient secretion of testosterone, has two main etiologies. Primary hypogonadism is caused by defects of the gonads, such as Klinefelter's syndrome or Leydig cell aplasia, whereas secondary hypogonadism is the failure of the hypothalamus (or pituitary) to produce sufficient gonadotropins (FSH, LH).
12.3 Pharmacokinetics
In a single-dose, replicate crossover clinical study evaluating 58 hypogonadal males, the serum testosterone exposures (AUC0-24 and AUC0-t) and maximum testosterone concentration (Cmax) following a topical administration of 100 mg testosterone administration as a 2 × 5 g Vogelxo tubes (applied to the shoulders/upper arms) were bioequivalent to those following a topical administration of an approved testosterone gel product.
Absorption
Testosterone gel delivers physiologic amounts of testosterone, producing circulating testosterone concentrations that approximate normal levels (e.g., 300 – 1000 ng/dL) seen in healthy men.
The skin serves as a reservoir for the sustained release of testosterone into the systemic circulation. Approximately 10% of the testosterone applied on the skin surface is absorbed into the systemic circulation during a 24-hour period.
Single Dose
In a single dose, replicate crossover study, when Vogelxo 100 mg was applied, absorption of testosterone into the blood continued for the entire 24 hour dosing period. The average (± SD) AUC0-24 and AUC0-t and Cmax were 6625 (±3671) ng∙hr/dL, 10425 (±5521) ng∙hr/dL, and 573 (±284) ng/dL, respectively.
Multiple Dose
With single daily applications of testosterone gel 50 mg and 100 mg, follow-up measurements at 30 and 90 days after starting treatment have confirmed that serum testosterone and DHT concentrations are generally maintained within the normal range.
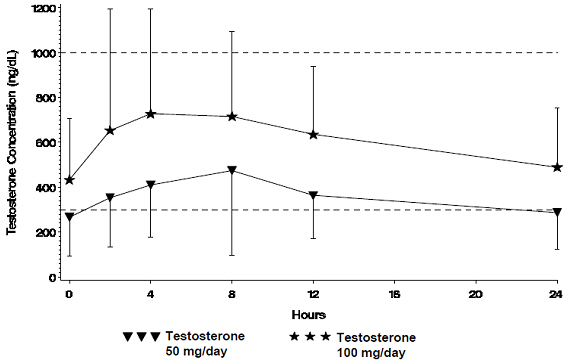
Figure 1 summarizes the 24-hour pharmacokinetic profile of testosterone for patients maintained on testosterone gel 50 mg or testosterone gel 100 mg for 30 days.
| Figure 1 Mean Steady-State Serum Testosterone (±SD) (ng/dL) Concentrations on Day 30 in Patients Applying Testosterone Once Daily |
|
|
The average daily testosterone concentration produced by testosterone gel 100 mg at Day 30 was 612 (± 286) ng/dL and by testosterone gel 50 mg at Day 30 was 365 (± 187) ng/dL.
Distribution
Circulating testosterone is primarily bound in the serum to sex hormone-binding globulin (SHBG) and albumin. Approximately 40% of testosterone in plasma is bound to SHBG, 2% remains unbound (free) and the rest is loosely bound to albumin and other proteins.
Metabolism
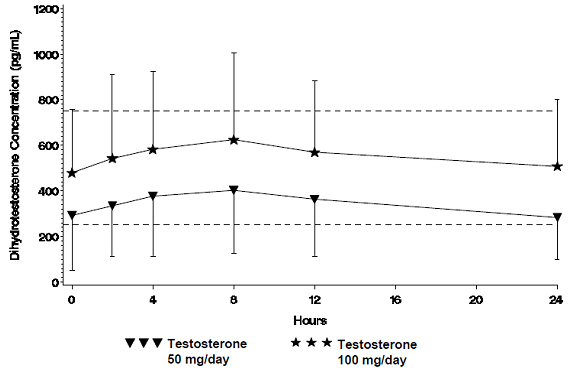
Testosterone is metabolized to various 17-keto steroids through two different pathways. The major active metabolites of testosterone are estradiol and DHT. The average daily DHT concentration produced by testosterone gel 100 mg at Day 30 was 555 (± 293) pg/mL and by testosterone gel 50 mg at Day 30 was 346 (± 212) pg/mL.
Figure 2 summarizes the 24-hour pharmacokinetic profile of DHT for patients maintained on testosterone gel 50 mg or testosterone gel 100 mg for 30 days.
| Figure 2 Mean Steady-State Serum Dihydrotestosterone (±SD) (pg/mL) Concentrations on Day 30 in Patients Applying Testosterone Once Daily |
|
|
Excretion
There is considerable variation in the half-life of testosterone concentration as reported in the literature, ranging from 10 to 100 minutes. About 90% of a dose of testosterone given intramuscularly is excreted in the urine as glucuronic acid and sulfuric acid conjugates of testosterone and its metabolites. About 6% of a dose is excreted in the feces, mostly in the unconjugated form. Inactivation of testosterone occurs primarily in the liver.
Potential for Transfer from Male Patients to Female Partners
The potential for dermal testosterone transfer following Vogelxo use was evaluated in a clinical study between males dosed with Vogelxo and their untreated female partners. Two (2) hours after application of 50 mg of testosterone from 5 g of Vogelxo to upper arm and shoulder of one side by the male subjects, the couples (N = 48 couples) engaged in a 15 minute session of skin-to-skin contact. Serum concentrations of testosterone were monitored in the female subjects for 24 hours after the transfer procedure. Under these study conditions, unprotected female partners had a mean testosterone AUC0-24 and Cmax that were 2.8 and 4 times greater than their mean baseline values, respectively. When a shirt covered the application site or the application site was washed, study results showed less than 10% increase in testosterone AUC0-24 and Cmax, compared to baseline in these females.
Effect of Hand Washing
In a clinical study conducted to evaluate the effect of hand washing on the residual amount of testosterone, 36 healthy male subjects received 50 mg of testosterone from 5 g of Vogelxo on a hand and applied testosterone gel to the upper arm and shoulder of one side. Subjects washed their hands with liquid soap and warm water immediately after drug application. Then the hand was allowed to air dry or patted dry with a cloth towel. A skin swab sample was collected and analyzed for testosterone content. A mean (SD) of 0.16 (0.46) to 0.65(1.03) µg of residual testosterone (i.e., 99% reduction compared to when hands were not washed) was recovered after washing hands with liquid soap and warm water.
Effect of Showering
The effect of showering (with mild soap) at 1, 2 and 6 hours post application of testosterone gel 100 mg was evaluated in a clinical trial in 12 men. The study demonstrated that the overall effect of washing was to decrease testosterone concentrations; however, when washing occurred two or more hours post drug application, serum testosterone concentrations remained within the normal range.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
Testosterone has been tested by subcutaneous injection and implantation in mice and rats. In mice, the implant induced cervical-uterine tumors, which metastasized in some cases. There is suggestive evidence that injection of testosterone into some strains of female mice increases their susceptibility to hepatoma. Testosterone is also known to increase the number of tumors and decrease the degree of differentiation of chemically induced carcinomas of the liver in rats.
14 CLINICAL STUDIES
14.1 Clinical Study in Hypogonadal Males
Testosterone gel was evaluated in a randomized multicenter, multi-dose, active and placebo controlled 90-day study in 406 adult males with morning testosterone levels ≤300 ng/dL. The study was double-blind for the doses of testosterone gel and placebo, but open label for the non-scrotal testosterone transdermal system. During the first 60 days, patients were evenly randomized to testosterone gel 50 mg, testosterone gel 100 mg, placebo gel, or testosterone transdermal system. At Day 60, patients receiving testosterone gel were maintained at the same dose, or were titrated up or down within their treatment group, based on 24-hour averaged serum testosterone concentration levels obtained on Day 30.
Of 192 hypogonadal men who were appropriately titrated with testosterone gel and who had sufficient data for analysis, 74% achieved an average serum testosterone level within the normal range (300 to 1,000 ng/dL) on treatment Day 90.
Table 4 summarizes the mean testosterone concentrations on Day 30 for patients receiving testosterone gel 50 mg or 100 mg.
| Testosterone gel 50 mg | Testosterone gel 100 mg | Placebo gel | |
|---|---|---|---|
| n=94 | n=95 | n=93 | |
| Cavg (ng/dL) | 365 ± 187 | 612 ± 286 | 216 ± 79 |
| Cmax (ng/dL) | 538 ± 371 | 897 ± 565 | 271 ± 110 |
| Cmin (ng/dL) | 223 ± 126 | 394 ±189 | 164 ± 64 |
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Vogelxo is supplied in unit-dose tubes in cartons of 30 and unit-dose packets in cartons of 30. Each tube or packet contains 50 mg testosterone in 5 g of gel.
Vogelxo is also supplied in a metered-dose pump that delivers 12.5 mg of testosterone per complete pump actuation. Each 88 g metered-dose pump is capable of dispensing 75 g of gel or 60-metered pump actuations. Each pump actuation delivers 1.25 g of gel. The metered-dose pump is supplied in cartons of 2.
Vogelxo is available as follows:
| NDC Number | Strength | Package Size |
|---|---|---|
| 0245-0871-05 | 50 mg of testosterone | 30 tubes (5 g of gel per tube) |
| 0245-0871-65 | 50 mg of testosterone | 1 tube (5 g of gel per tube) |
| 0245-0871-35 | 50 mg of testosterone | 30 packets (5 g of gel per packet) |
| 0245-0871-89 | 50 mg of testosterone | 1 packet (5 g of gel per packet) |
| 0245-0872-42 | 12.5 mg of testosterone per pump actuation | 2 × 75 g pumps (each pump dispenses 60 metered 1.25 g of gel) |
16.2 Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
16.3 Handling and Disposal
Used Vogelxo tubes, packets or pumps should be discarded in household trash in a manner that prevents accidental exposure of women, children, or pets [see Boxed Warning and Warnings and Precautions (5.2)]. Contents are flammable [see Warnings and Precautions (5.16)].
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Men with Known or Suspected Carcinoma of the Breast or Prostate
Men with known or suspected prostate or breast cancer should not use Vogelxo [see Contraindications (4) and Warnings and Precautions (5.1)].
Potential for Secondary Exposure to Testosterone and Steps to Prevent Secondary Exposure
Secondary exposure to testosterone in children and women can occur with the use of testosterone gel products in men. Cases of secondary exposure to testosterone have been reported in children.
Physicians should advise patients of the reported signs and symptoms of secondary exposure which may include the following:
- In children; unexpected sexual development including inappropriate enlargement of the penis or clitoris, premature development of pubic hair, increased erections, and aggressive behavior
- In women; changes in hair distribution, increase in acne, or other signs of testosterone effects
- The possibility of secondary exposure to Vogelxo should be brought to the attention of a healthcare provider
- Vogelxo should be promptly discontinued until the cause of virilization is identified
Strict adherence to the following precautions is advised to minimize the potential for secondary exposure to testosterone from Vogelxo in men [see Medication Guide]
- Children and women should avoid contact with unwashed or unclothed application site(s) of men using Vogelxo
- Patients using Vogelxo should apply the product as directed and strictly adhere to the following:
- Wash hands with soap and water immediately after application
- Cover the application site(s) with clothing after the gel has dried
- Wash the application site(s) thoroughly with soap and water prior to any situation where skin-to-skin contact of the application site with another person is anticipated
- In the event that unwashed or unclothed skin to which Vogelxo has been applied comes in contact with the skin of another person, the general area of contact on the other person should be washed with soap and water as soon as possible [see Dosage and Administration (2.2), Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
Potential Adverse Reactions with Androgens
Patients should be informed that treatment with androgens may lead to adverse reactions which include:
- Changes in urinary habits, such as increased urination at night, trouble starting the urine stream, passing urine many times during the day, having an urge to go the bathroom right away, having a urine accident, or being unable to pass urine or weak urine flow
- Breathing disturbances, including those associated with sleep or excessive daytime sleepiness.
- Too frequent or persistent erections of the penis
- Nausea, vomiting, changes in skin color, or ankle swelling
Patients Should Be Advised of the Following Instructions for Use
- Read the Medication Guide before starting Vogelxo therapy and reread it each time the prescription is renewed.
- Vogelxo should be applied and used appropriately to maximize the benefits and to minimize the risk of secondary exposure in children and women.
- Keep Vogelxo out of the reach of children. The package is not child resistant.
- Vogelxo is an alcohol-based product and is flammable; therefore avoid fire, flame or smoking until the gel has dried.
- It is important to adhere to all recommended monitoring.
- Report any changes in their state of health, such as changes in urinary habits, breathing, sleep, and mood.
- Vogelxo is prescribed to meet the patient's specific needs; therefore, the patient should never share Vogelxo with anyone.
- Vogelxo should be applied topically once daily at approximately the same time each day to clean dry skin of the shoulders and/or upper arms.
- Vogelxo should not be applied to the scrotum, penis, or abdomen.
- Wait 2 hours before washing or swimming following application of Vogelxo. This will ensure that the greatest amount of Vogelxo is absorbed.
Distributed by
UPSHER-SMITH LABORATORIES, LLC
Maple Grove, MN 55369
MADE IN CANADA
Vogelxo is a registered trademark of Upsher-Smith Laboratories, LLC.
Revised 04/2020
| MEDICATION GUIDE VOGELXO® (voh-JELKS-oh) CIII (testosterone) gel, for topical use |
|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. |
|
What is the most important information I should know about Vogelxo?
|
|
| What is Vogelxo?
Vogelxo is a prescription medicine that contains testosterone. Vogelxo is used to treat adult males who have low or no testosterone due to certain medical conditions.
Vogelxo is not meant for use in women. |
Do not use Vogelxo if you:
|
Before using Vogelxo, tell your healthcare provider about all of your medical conditions including if you:
Especially, tell your healthcare provider if you take:
|
How should I use Vogelxo?
|
| What are the possible side effects of Vogelxo? Vogelxo can cause serious side effects including: See "What is the most important information I should know about Vogelxo?"
The most common side effects of Vogelxo include:
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Vogelxo. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
| General information about the safe and effective use of Vogelxo.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Vogelxo for a condition for which it was not prescribed. Do not give Vogelxo to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Vogelxo that is written for health professionals. |
| What are the ingredients in Vogelxo? Active ingredient: testosterone Inactive ingredients: carbomer copolymer Type B, carbomer homopolymer Type C, diisopropyl adipate, ethyl alcohol, glycerin, methyl laurate, oleyl alcohol, polyethylene glycol, propylene glycol, purified water, and tromethamine Distributed by UPSHER-SMITH LABORATORIES, LLC Maple Grove, MN 55369 MADE IN CANADA For more information about Vogelxo, call 1-888-650-3789 or go to www.upsher-smith.com |
Revised 4/2020
INSTRUCTIONS FOR USE
VOGELXO® (voh-JELKS-oh), CIII
(testosterone)
gel, for topical use
Read this Instructions for Use for Vogelxo before you start using it and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment.
Applying Vogelxo:
Vogelxo comes in tubes, packets, or in a pump.
- Before applying Vogelxo, make sure that your shoulders and upper arms are clean, dry, and there is no broken skin.
- The application sites for Vogelxo are the shoulders and the upper arms that will be covered by a short sleeve t-shirt (See Figure A). Do not apply Vogelxo to any other parts of your body such as your penis, scrotum, or stomach area (abdomen).
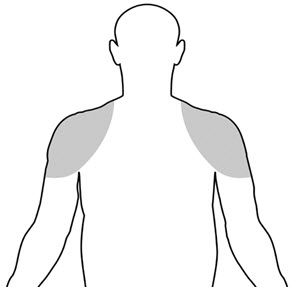 |
| (Figure A) |
If you are using Vogelxo tubes:
- Remove the cap from the tube and use the top of the cap to puncture the metal seal on the top of the tube.
- Squeeze from the bottom of the tube to the top.
- Squeeze all of the Vogelxo out of the tube into the palm of your hand.
- Vogelxo is to be applied only to the areas of your shoulders and upper arms that will be covered by a short sleeve t-shirt.
- Apply Vogelxo to the application site. Rub the gel onto your skin for several seconds.
- Vogelxo is flammable until dry. Let Vogelxo dry before smoking or going near an open flame.
- Let the application site dry for a few minutes before putting on a t-shirt.
- Wash your hands with soap and water right away after applying Vogelxo.
- Put the cap back on the tube.
- Avoid showering, swimming, or bathing for at least 2 hours after you apply Vogelxo.
If you are using Vogelxo packets:
- Tear open the packet completely at the notch on the top edge. Squeeze from the bottom of the packet to the top.
- Squeeze all of the Vogelxo out of the packet into the palm of your hand. Apply Vogelxo to the application site. Rub the gel onto your skin for several seconds.
- Let the application site dry for a few minutes before putting on a t-shirt.
- Wash your hands with soap and water right away after applying Vogelxo.
If you are using the Vogelxo pump:
- Before using a new bottle of Vogelxo for the first time, you will need to prime the pump. To prime the Vogelxo pump, remove the cap and slowly push the pump all the way down 3 times.
- Do not use any Vogelxo that came out while priming. Wash it down the sink to avoid accidental exposure to others. Your Vogelxo pump is now ready to use.
- Remove the cap from the pump. Then position the nozzle over the palm of your hand and slowly push the pump all the way down. Your healthcare provider will tell you the number of times to press the pump for each dose.
- Apply Vogelxo to the application site. Rub the gel onto your skin for several seconds.
- Let the application site dry for a few minutes before putting on a t-shirt.
- Wash your hands with soap and water right away.
- Put the cap back on the pump.
How should I store Vogelxo?
- Store Vogelxo at room temperature between 68º to 77ºF (20º to 25ºC).
- Safely throw away used Vogelxo containers in the household trash. Be careful to prevent accidental exposure of children or pets.
- Keep Vogelxo away from fire.
Keep Vogelxo and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration Revised:
Revised 4/2020
<
PRINCIPAL DISPLAY PANEL - 50 mg Tube Carton
NDC 0245-0871-05
Vogelxo®
(testosterone) gel
50 mg testosterone per tube
Each unit-dose tube
contains 5 grams of gel.
CIII
For topical use only.
Topical testosterone products may have different doses, strengths,
or application instructions that may result in different systemic exposure.
To be applied to the shoulders and upper arms.
PHARMACIST: Dispense the Medication Guide provided separately
to each patient.
Contains 30 Unit-dose Tubes
Rx only
UPSHER-SMITH
PRINCIPAL DISPLAY PANEL - 12.5 mg Bottle Carton
NDC 0245-0872-42
Vogelxo®
(testosterone) gel
12.5 mg of testosterone
per pump actuation*
*Each actuation delivers
1.25 grams of gel.
Multi-dose pump capable of
dispensing 60 metered
pump actuations.
CIII
For topical use only.
Topical testosterone products may have different
doses, strengths, or application instructions that
may result in different systemic exposure.
To be applied to the shoulders and upper arms.
PHARMACIST: Dispense the Medication Guide
provided separately to each patient.
2 canisters containing 88 grams each
Rx only
UPSHER-SMITH