DESCRIPTION
DITROPAN XL® (oxybutynin chloride) is an antispasmodic, anticholinergic agent. Each DITROPAN XL® Extended Release Tablet contains 5 mg, 10 mg, or 15 mg of oxybutynin chloride USP, formulated as a once-a-day controlled-release tablet for oral administration. Oxybutynin chloride is administered as a racemate of R- and S-enantiomers.
Chemically, oxybutynin chloride is d,l (racemic) 4-diethylamino-2-butynyl phenylcyclohexylglycolate hydrochloride. The empirical formula of oxybutynin chloride is C22H31NO3•HCl.
Its structural formula is:
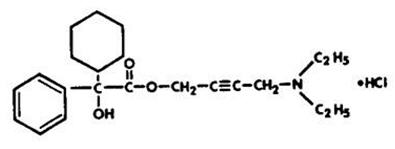
Oxybutynin chloride is a white crystalline solid with a molecular weight of 393.9. It is readily soluble in water and acids, but relatively insoluble in alkalis.
DITROPAN XL® also contains the following inert ingredients: cellulose acetate, hypromellose, lactose, magnesium stearate, polyethylene glycol, polyethylene oxide, synthetic iron oxides, titanium dioxide, polysorbate 80, sodium chloride, and butylated hydroxytoluene.
System Components and Performance
DITROPAN XL® uses osmotic pressure to deliver oxybutynin chloride at a controlled rate over approximately 24 hours. The system, which resembles a conventional tablet in appearance, comprises an osmotically active bilayer core surrounded by a semipermeable membrane. The bilayer core is composed of a drug layer containing the drug and excipients, and a push layer containing osmotically active components. There is a precision-laser drilled orifice in the semipermeable membrane on the drug-layer side of the tablet. In an aqueous environment, such as the gastrointestinal tract, water permeates through the membrane into the tablet core, causing the drug to go into suspension and the push layer to expand. This expansion pushes the suspended drug out through the orifice. The semipermeable membrane controls the rate at which water permeates into the tablet core, which in turn controls the rate of drug delivery. The controlled rate of drug delivery into the gastrointestinal lumen is thus independent of pH or gastrointestinal motility. The function of DITROPAN XL® depends on the existence of an osmotic gradient between the contents of the bilayer core and the fluid in the gastrointestinal tract. Since the osmotic gradient remains constant, drug delivery remains essentially constant. The biologically inert components of the tablet remain intact during gastrointestinal transit and are eliminated in the feces as an insoluble shell.
CLINICAL PHARMACOLOGY
Oxybutynin chloride exerts a direct antispasmodic effect on smooth muscle and inhibits the muscarinic action of acetylcholine on smooth muscle. Oxybutynin chloride exhibits only one-fifth of the anticholinergic activity of atropine on the rabbit detrusor muscle, but four to ten times the antispasmodic activity. No blocking effects occur at skeletal neuromuscular junctions or autonomic ganglia (antinicotinic effects).
Oxybutynin chloride relaxes bladder smooth muscle. In patients with conditions characterized by involuntary bladder contractions, cystometric studies have demonstrated that oxybutynin increases bladder (vesical) capacity, diminishes the frequency of uninhibited contractions of the detrusor muscle, and delays the initial desire to void. Oxybutynin thus decreases urgency and the frequency of both incontinent episodes and voluntary urination.
Antimuscarinic activity resides predominantly in the R-isomer. A metabolite, desethyloxybutynin, has pharmacological activity similar to that of oxybutynin in in vitro studies.
Pharmacokinetics
Absorption
Following the first dose of DITROPAN XL® (oxybutynin chloride), oxybutynin plasma concentrations rise for 4 to 6 hours; thereafter steady concentrations are maintained for up to 24 hours, minimizing fluctuations between peak and trough concentrations associated with oxybutynin.
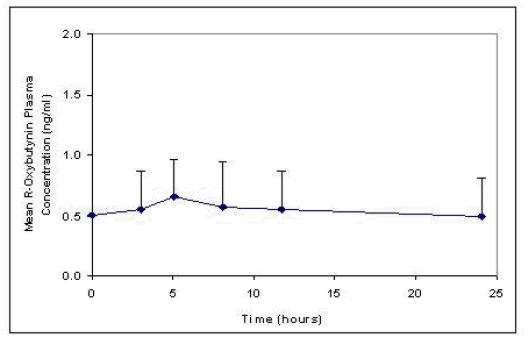
The relative bioavailabilities of R- and S-oxybutynin from DITROPAN XL® are 156% and 187%, respectively, compared with oxybutynin. The mean pharmacokinetic parameters for R- and S-oxybutynin are summarized in Table 1. The plasma concentration-time profiles for R- and S-oxybutynin are similar in shape; Figure 1 shows the profile for R-oxybutynin.
| Parameters (units) | R-Oxybutynin | S-Oxybutynin | ||
|---|---|---|---|---|
| Cmax (ng/mL) | 1.0 | (0.6) | 1.8 | (1.0) |
| Tmax (h) | 12.7 | (5.4) | 11.8 | (5.3) |
| t1/2 (h) | 13.2 | (6.2) | 12.4 | (6.1) |
| AUC (0–48) (ng∙h/mL) | 18.4 | (10.3) | 34.2 | (16.9) |
| AUCinf (ng∙h/mL) | 21.3 | (12.2) | 39.5 | (21.2) |
 |
Steady state oxybutynin plasma concentrations are achieved by Day 3 of repeated DITROPAN XL® dosing, with no observed drug accumulation or change in oxybutynin and desethyloxybutynin pharmacokinetic parameters.
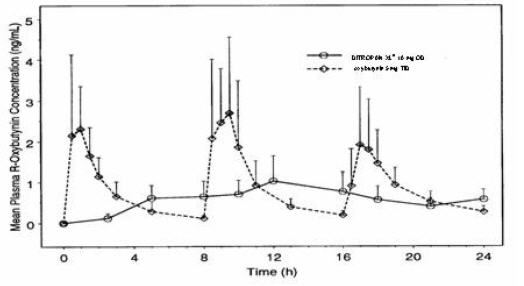
DITROPAN XL® steady state pharmacokinetics were studied in 19 children aged 5–15 years with detrusor overactivity associated with a neurological condition (e.g., spina bifida). The children were on DITROPAN XL® total daily dose ranging from 5 to 20 mg (0.10 to 0.77 mg/kg). Sparse sampling technique was used to obtain serum samples. When all available data are normalized to an equivalent of 5 mg per day DITROPAN XL®, the mean pharmacokinetic parameters derived for R- and S-oxybutynin and R- and S-desethyloxybutynin are summarized in Table 2. The plasma-time concentration profiles for R- and S-oxybutynin are similar in shape; Figure 2 shows the profile for R-oxybutynin when all available data are normalized to an equivalent of 5 mg per day.
| R-Oxybutynin | S-Oxybutynin | R- Desethyloxybutynin | S- Desethyloxybutynin | |
|---|---|---|---|---|
| Cmax (ng/mL) | 0.7 ± 0.4 | 1.3 ± 0.8 | 7.8 ± 3.7 | 4.2 ± 2.3 |
| Tmax (h) | 5.0 | 5.0 | 5.0 | 5.0 |
| AUC(ng∙h/mL) | 12.8 ± 7.0 | 23.7 ± 14.4 | 125.1 ± 66.7 | 73.6 ± 47.7 |
Distribution
Oxybutynin is widely distributed in body tissues following systemic absorption. The volume of distribution is 193 L after intravenous administration of 5 mg oxybutynin chloride. Both enantiomers of oxybutynin are highly bound (>99%) to plasma proteins. Both enantiomers of N-desethyloxybutynin are also highly bound (>97%) to plasma proteins. The major binding protein is alpha-1 acid glycoprotein.
Metabolism
Oxybutynin is metabolized primarily by the cytochrome P450 enzyme systems, particularly CYP3A4 found mostly in the liver and gut wall. Its metabolic products include phenylcyclohexylglycolic acid, which is pharmacologically inactive, and desethyloxybutynin, which is pharmacologically active. Following DITROPAN XL® administration, plasma concentrations of R- and S-desethyloxybutynin are 73% and 92%, respectively, of concentrations observed with oxybutynin.
Excretion
Oxybutynin is extensively metabolized by the liver, with less than 0.1% of the administered dose excreted unchanged in the urine. Also, less than 0.1% of the administered dose is excreted as the metabolite desethyloxybutynin.
Dose Proportionality
Pharmacokinetic parameters of oxybutynin and desethyloxybutynin (Cmax and AUC) following administration of 5–20 mg of DITROPAN XL® are dose proportional.
Special Populations
Geriatric
The pharmacokinetics of DITROPAN XL® were similar in all patients studied (up to 78 years of age).
Pediatric
The pharmacokinetics of DITROPAN XL® were evaluated in 19 children aged 5–15 years with detrusor overactivity associated with a neurological condition (e.g., spina bifida). The pharmacokinetics of DITROPAN XL® in these pediatric patients were consistent with those reported for adults (see Tables 1 and 2, and Figures 1 and 2 above).
Gender
There are no significant differences in the pharmacokinetics of oxybutynin in healthy male and female volunteers following administration of DITROPAN XL®.
Race
Available data suggest that there are no significant differences in the pharmacokinetics of oxybutynin based on race in healthy volunteers following administration of DITROPAN XL®.
Renal Insufficiency
There is no experience with the use of DITROPAN XL® in patients with renal insufficiency.
CLINICAL STUDIES
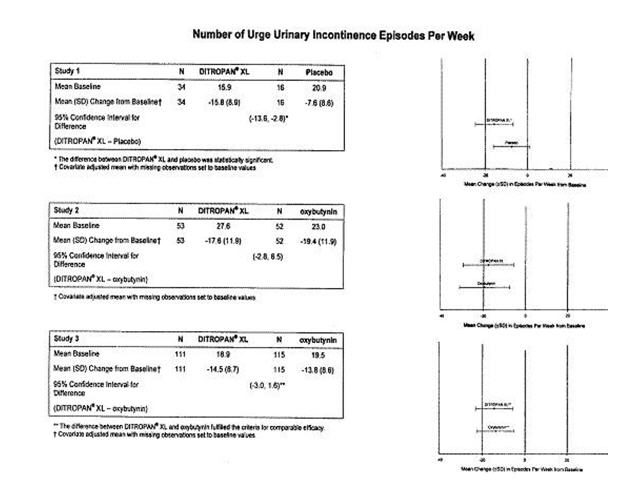
DITROPAN XL® (oxybutynin chloride) was evaluated for the treatment of patients with overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency in three controlled studies and one open-label study. The majority of patients were Caucasian (89.0%) and female (91.9%) with a mean age of 59 years (range, 18 to 98 years). Entry criteria required that patients have urge or mixed incontinence (with a predominance of urge) as evidenced by ≥ 6 urge incontinence episodes per week and ≥ 10 micturitions per day. Study 1 was a fixed-dose escalation design, whereas the other studies used a dose-adjustment design in which each patient's final dose was adjusted to a balance between improvement of incontinence symptoms and tolerability of side effects. Controlled studies included patients known to be responsive to oxybutynin or other anticholinergic medications, and these patients were maintained on a final dose for up to 2 weeks.
The efficacy results for the three controlled trials are presented in the following tables and figures.
INDICATIONS AND USAGE
DITROPAN XL® (oxybutynin chloride) is a once-daily controlled-release tablet indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency.
DITROPAN XL® is also indicated in the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
CONTRAINDICATIONS
DITROPAN XL® (oxybutynin chloride) is contraindicated in patients with urinary retention, gastric retention and other severe decreased gastrointestinal motility conditions, uncontrolled narrow-angle glaucoma and in patients who are at risk for these conditions.
DITROPAN XL® is also contraindicated in patients who have demonstrated hypersensitivity to the drug substance or other components of the product.
WARNINGS
Angioedema of the face, lips, tongue and/or larynx has been reported with oxybutynin. In some cases, angioedema occurred after the first dose. Angioedema associated with upper airway swelling may be life-threatening. If involvement of the tongue, hypopharynx, or larynx occurs, oxybutynin should be promptly discontinued and appropriate therapy and/or measures necessary to ensure a patent airway should be promptly provided.
PRECAUTIONS
Central Nervous System Effects
Oxybutynin is associated with anticholinergic central nervous system (CNS) effects (see ADVERSE REACTIONS). A variety of CNS anticholinergic effects have been reported, including hallucinations, agitation, confusion and somnolence. Patients should be monitored for signs of anticholinergic CNS effects, particularly in the first few months after beginning treatment or increasing the dose. If a patient experiences anticholinergic CNS effects, dose reduction or drug discontinuation should be considered.
DITROPAN XL® should be used with caution in patients with preexisting dementia treated with cholinesterase inhibitors due to the risk of aggravation of symptoms.
General
DITROPAN XL® (oxybutynin chloride) should be used with caution in patients with hepatic or renal impairment and in patients with myasthenia gravis due to the risk of symptom aggravation.
Urinary Retention
DITROPAN XL® should be administered with caution to patients with clinically significant bladder outflow obstruction because of the risk of urinary retention (see CONTRAINDICATIONS).
Gastrointestinal Disorders
DITROPAN XL® should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention (see CONTRAINDICATIONS).
DITROPAN XL®, like other anticholinergic drugs, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis and intestinal atony.
DITROPAN XL® should be used with caution in patients who have gastroesophageal reflux and/or who are concurrently taking drugs (such as bisphosphonates) that can cause or exacerbate esophagitis.
As with any other nondeformable material, caution should be used when administering DITROPAN XL® to patients with preexisting severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs in nondeformable controlled-release formulations.
Information for Patients
Patients should be informed that oxybutynin may produce angioedema that could result in life-threatening airway obstruction. Patients should be advised to promptly discontinue oxybutynin therapy and seek immediate medical attention if they experience edema of the tongue, edema of the laryngopharynx, or difficulty breathing.
Patients should be informed that heat prostration (fever and heat stroke due to decreased sweating) can occur when anticholinergics such as oxybutynin chloride are administered in the presence of high environmental temperature.
Because anticholinergic agents such as oxybutynin may produce drowsiness (somnolence) or blurred vision, patients should be advised to exercise caution.
Patients should be informed that alcohol may enhance the drowsiness caused by anticholinergic agents such as oxybutynin.
Patients should be informed that DITROPAN XL® should be swallowed whole with the aid of liquids. Patients should not chew, divide, or crush tablets. The medication is contained within a nonabsorbable shell designed to release the drug at a controlled rate. The tablet shell is eliminated from the body; patients should not be concerned if they occasionally notice in their stool something that looks like a tablet.
DITROPAN XL® should be taken at approximately the same time each day.
Drug Interactions
The concomitant use of oxybutynin with other anticholinergic drugs or with other agents which produce dry mouth, constipation, somnolence (drowsiness), and/or other anticholinergic-like effects may increase the frequency and/or severity of such effects.
Anticholinergic agents may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility. This may be of concern for drugs with a narrow therapeutic index.
Mean oxybutynin chloride plasma concentrations were approximately 2 fold higher when DITROPAN XL® was administered with ketoconazole, a potent CYP3A4 inhibitor. Other inhibitors of the cytochrome P450 3A4 enzyme system, such as antimycotic agents (e.g., itraconazole and miconazole) or macrolide antibiotics (e.g., erythromycin and clarithromycin), may alter oxybutynin mean pharmacokinetic parameters (i.e., Cmax and AUC). The clinical relevance of such potential interactions is not known. Caution should be used when such drugs are co-administered.
Carcinogenesis, Mutagenesis, Impairment of Fertility
A 24-month study in rats at dosages of oxybutynin chloride of 20, 80, and 160 mg/kg/day showed no evidence of carcinogenicity. These doses are approximately 6, 25, and 50 times the maximum human exposure, based on surface area.
Oxybutynin chloride showed no increase of mutagenic activity when tested in Schizosaccharomyces pompholiciformis, Saccharomyces cerevisiae, and Salmonella typhimurium test systems.
Reproduction studies with oxybutynin chloride in the mouse, rat, hamster, and rabbit showed no definite evidence of impaired fertility.
Pregnancy
Teratogenic Effects
Pregnancy Category B
Reproduction studies with oxybutynin chloride in the mouse, rat, hamster, and rabbit showed no definite evidence of impaired fertility or harm to the animal fetus. The safety of DITROPAN XL® administration to women who are or who may become pregnant has not been established. Therefore, DITROPAN XL® should not be given to pregnant women unless, in the judgment of the physician, the probable clinical benefits outweigh the possible hazards.
Nursing Mothers
It is not known whether oxybutynin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when DITROPAN XL® is administered to a nursing woman.
Pediatric Use
The safety and efficacy of DITROPAN XL® were studied in 60 children in a 24-week, open-label trial. Patients were aged 6–15 years, all had symptoms of detrusor overactivity in association with a neurological condition (e.g., spina bifida), all used clean intermittent catheterization, and all were current users of oxybutynin chloride. Study results demonstrated that administration of DITROPAN XL® 5 to 20 mg/day was associated with an increase from baseline in mean urine volume per catheterization from 108 mL to 136 mL, an increase from baseline in mean urine volume after morning awakening from 148 mL to 189 mL, and an increase from baseline in the mean percentage of catheterizations without a leaking episode from 34% to 51%.
Urodynamic results were consistent with clinical results. Administration of DITROPAN XL® resulted in an increase from baseline in mean maximum cystometric capacity from 185 mL to 254 mL, a decrease from baseline in mean detrusor pressure at maximum cystometric capacity from 44 cm H2O to 33 cm H2O, and a reduction in the percentage of patients demonstrating uninhibited detrusor contractions (of at least 15 cm H2O) from 60% to 28%.
DITROPAN XL® is not recommended in pediatric patients who cannot swallow the tablet whole without chewing, dividing, or crushing, or in children under the age of 6 (See DOSAGE AND ADMINISTRATION).
Geriatric Use
The rate and severity of anticholinergic effects reported by patients less than 65 years old and those 65 years and older were similar (see CLINICAL PHARMACOLOGY, Pharmacokinetics, Special Populations: Geriatric).
ADVERSE REACTIONS
Adverse Events with DITROPAN XL®
The safety and efficacy of DITROPAN XL® (oxybutynin chloride) were evaluated in a total of 580 participants who received DITROPAN XL® in 4 clinical trials (429 patients) and four pharmacokinetic studies (151 healthy volunteers). The 429 patients were treated with 5–30 mg/day for up to 4.5 months. Three of the 4 clinical trials allowed dose adjustments based on efficacy and adverse events and one was a fixed-dose escalation design. Safety information is provided for 429 patients from these three controlled clinical studies and one open-label study in the first column of Table 3 below.
Adverse events from two additional fixed-dose, active-controlled, 12-week treatment duration, postmarketing studies, in which 576 patients were treated with DITROPAN XL® 10 mg/day, are also listed in Table 3 (second column). The adverse events are reported regardless of causality.
| DITROPAN XL® | DITROPAN XL® | ||
|---|---|---|---|
| Body System | Adverse Event | 5–30 mg/day (n=429) | 10 mg/day (n=576) |
| General | headache | 10 | 6 |
| asthenia | 7 | 3 | |
| pain | 7 | 4 | |
| Digestive | dry mouth | 61 | 29 |
| constipation | 13 | 7 | |
| diarrhea | 9 | 7 | |
| nausea | 9 | 2 | |
| dyspepsia | 7 | 5 | |
| Nervous | somnolence | 12 | 2 |
| dizziness | 6 | 4 | |
| Respiratory | rhinitis | 6 | 2 |
| Special senses | blurred vision | 8 | 1 |
| dry eyes | 6 | 3 | |
| Urogenital | urinary tract infection | 5 | 5 |
The most common adverse events reported by the 429 patients receiving 5–30 mg/day DITROPAN XL® were the expected side effects of anticholinergic agents. The incidence of dry mouth was dose-related.
The discontinuation rate for all adverse events was 6.8% in the 429 patients from the 4 studies of efficacy and safety who received 5–30 mg/day. The most frequent adverse event causing early discontinuation of study medication was nausea (1.9%), while discontinuation due to dry mouth was 1.2%.
In addition, the following adverse events were reported by ≥ 1 to < 5% of all patients who received DITROPAN XL® in the 6 adjustable and fixed-dose efficacy and safety studies. Infections and infestations: nasopharyngitis, upper respiratory tract infection, sinusitis, bronchitis, cystitis; Psychiatric disorders: insomnia, depression, nervousness, confusional state; Nervous System Disorders: dysgeusia; Cardiac disorders: palpitations; Vascular disorders: hypertension; Respiratory, thoracic and mediastinal disorders: nasal dryness, cough, pharyngolaryngeal pain, dry throat; Gastrointestinal Disorders: gastroesophageal reflux disease, abdominal pain, loose stools, flatulence, vomiting; Skin and subcutaneous tissue disorders: dry skin, pruritis; Musculoskeletal and connective tissue disorders: back pain, arthralgia, pain in extremity; Renal and urinary disorders: urinary retention, urinary hesitation, dysuria; General disorders and administration site conditions: fatigue, edema peripheral, asthenia, chest pain; Investigations: blood pressure increased.
Postmarketing Surveillance
Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following additional adverse drug reactions have been reported from worldwide postmarketing experience with DITROPAN XL®: Psychiatric Disorders: psychotic disorder, agitation, hallucinations, memory impairment; Nervous System Disorders: convulsions; Eye Disorders: glaucoma; Cardiac Disorders: arrhythmia, tachycardia, QT interval prolongation; Vascular Disorders: flushing; Skin and Subcutaneous Tissue Disorders: rash; Renal and Urinary Disorders: impotence; General Disorders and Administration Site Conditions: hypersensitivity reactions, including angioedema with airway obstruction, urticaria, and face edema; rare anaphylactic reactions requiring hospitalization for emergency treatment; Injury, poisoning and procedural complications: fall.
Additional adverse events reported with some other oxybutynin chloride formulations include: cycloplegia, mydriasis, and suppression of lactation.
OVERDOSAGE
The continuous release of oxybutynin from DITROPAN XL® (oxybutynin chloride) should be considered in the treatment of overdosage. Patients should be monitored for at least 24 hours. Treatment should be symptomatic and supportive. Activated charcoal as well as a cathartic may be administered.
Overdosage with oxybutynin chloride has been associated with anticholinergic effects including central nervous system excitation, flushing, fever, dehydration, cardiac arrhythmia, vomiting, and urinary retention.
Ingestion of 100 mg oxybutynin chloride in association with alcohol has been reported in a 13-year-old boy who experienced memory loss, and a 34-year-old woman who developed stupor, followed by disorientation and agitation on awakening, dilated pupils, dry skin, cardiac arrhythmia, and retention of urine. Both patients fully recovered with symptomatic treatment.
DOSAGE AND ADMINISTRATION
DITROPAN XL® (oxybutynin chloride) must be swallowed whole with the aid of liquids, and must not be chewed, divided, or crushed.
DITROPAN XL® may be administered with or without food.
Adults
The recommended starting dose of DITROPAN XL® is 5 or 10 mg once daily at approximately the same time each day. Dosage may be adjusted in 5-mg increments to achieve a balance of efficacy and tolerability (up to a maximum of 30 mg/day). In general, dosage adjustment may proceed at approximately weekly intervals.
HOW SUPPLIED
DITROPAN XL® (oxybutynin chloride) Extended Release Tablets are available in three dosage strengths, 10 mg (pink), and 15 mg (gray) and are imprinted with "10 XL", or "15 XL". DITROPAN XL® Extended Release Tablets are supplied in
| 10 mg | 10 count bottle | NDC 54868-4502-1 |
|
| 30 count bottle | NDC 54868-4502-0 |
|
|
|
|
| 15 mg | 10 count bottle | NDC 54868-4835-1 |
|
| 30 count bottle | NDC 54868-4835-0 |
For more information call 1-888-395-1232 or visit www.DITROPANXL.com
Manufactured by:
ALZA Corporation, Vacaville, CA 95688
An ALZA OROS® Technology Product
DITROPAN XL® and OROS® are registered trademarks of ALZA Corporation.
Manufactured for:
Ortho Women's Health & Urology, Division of Ortho-McNeil-Janssen Pharmaceuticals, Inc.
Raritan, NJ 08869
Placeholder for Ortho Women's Health & Urology, Division of Ortho-McNeil-Janssen Pharmaceuticals, Inc. Logo
© Ortho-McNeil-Janssen Pharmaceuticals, Inc. 1998
10192603
Revised December 2011