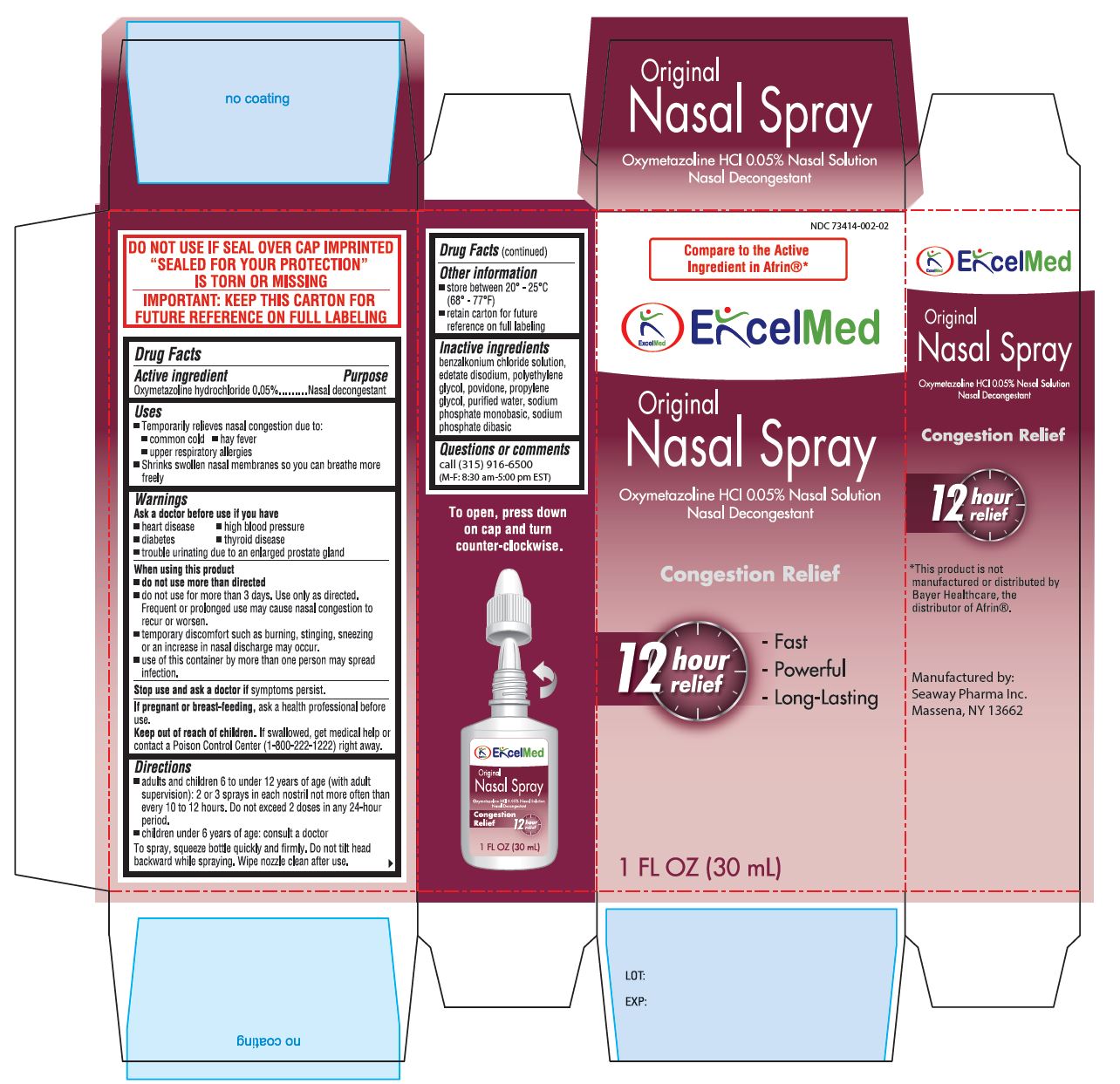
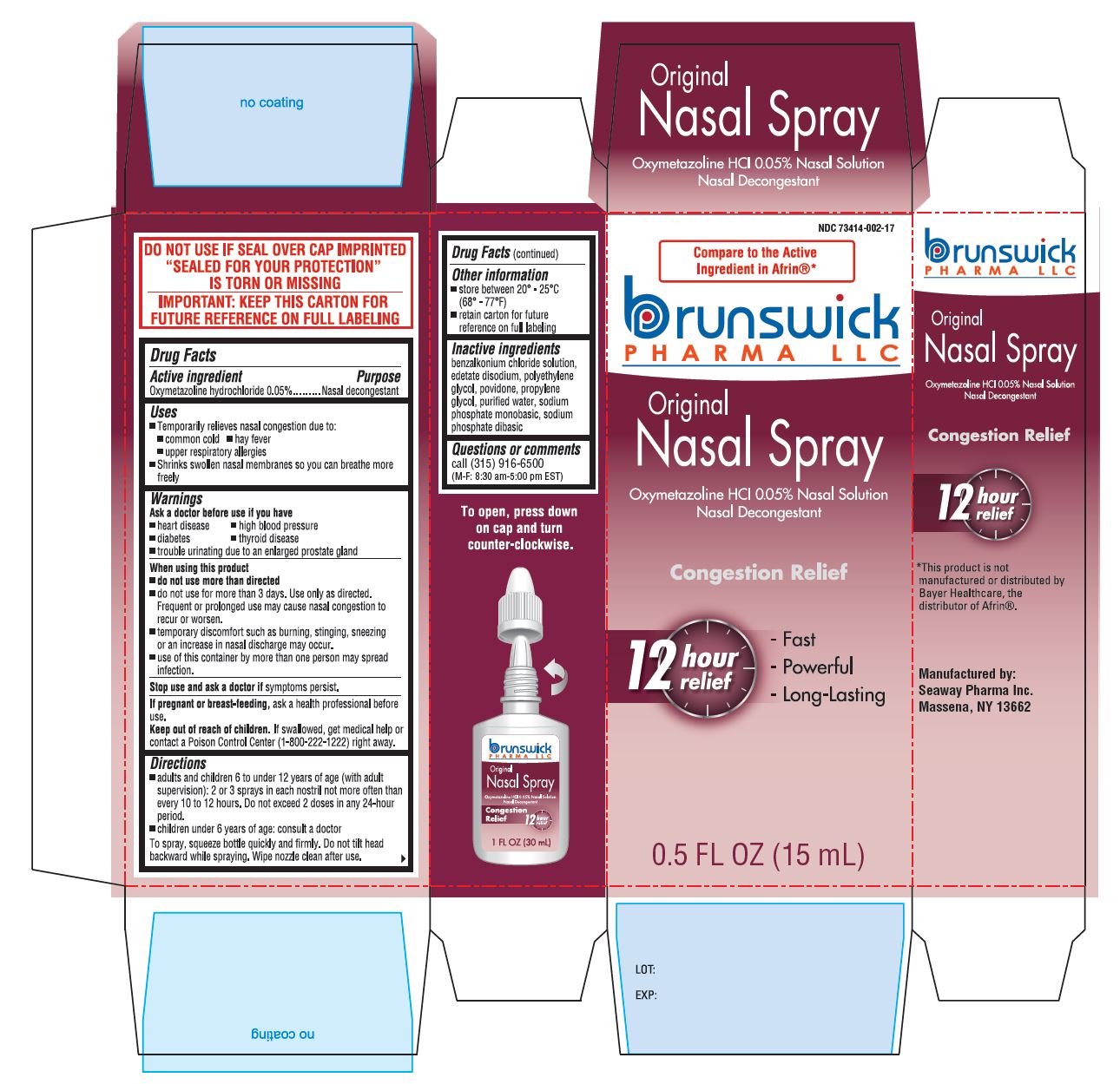
Uses
Temporarily relieves nasal congestion due to:
- common cold
- hay fever
- upper respiratory allergies
- shrinks swollen nasal membrances so you can breathe more freely.
- Frequent or prolonged use may cause nasal congestion to recur or worsen
- temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
- use of this container by more than one person may spread infection
Stop use and ask a doctor If symptoms persist.
If pregnant or breast feeding, ask a health professional before use.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
- adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostrill not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period.
- children under 6 years of age: consult a doctor.
To spray, squeeze bottle quickly and firmly. Do not tilt head backward while spraying. Wipe nozzle clean after use.
Inactive Ingredients
benzalkonium chloride solution, edetate disodium, polyethylene glycol, povidone, propylene glycol, purified water, sodium phosphate monobasic, sodium phosphate dibasic