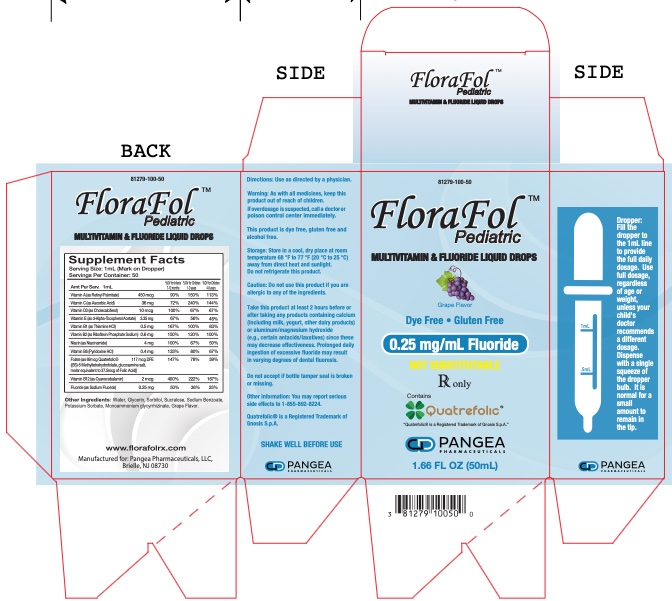
|
Supplement Facts | |||||
|
% Daily Value | |||||
|
Amount per serving 1mL |
Infants 7-12 months |
Children |
Children | ||
|
VITAMIN A (as Retinyl Palmitate) |
450 mcg |
90% |
150% |
113% | |
|
VITAMIN C (as Ascorbic Acid) |
36 mg |
72% |
240% |
144% | |
|
VITAMIN D3 (as Cholecalciferol) |
10 mcg |
100% |
67% |
67% | |
|
VITAMIN E (as d-Alpha-Tocopherol Acetate) |
3.35 mg |
45% |
38% |
32% | |
|
VITAMIN B1 (as Thiamine HCl) |
0.5 mg |
167% |
100% |
83% | |
|
VITAMIN B2 (as Riboflavin Phosphate Sodium) |
0.6 mg |
150% |
120% |
100% | |
|
NIACIN (as Niacinamide) |
4 mg |
100% |
67% |
50% | |
|
VITAMIN B6 (Pyridoxine HCl) |
0.4 mg |
133% |
80% |
67% | |
|
Folate (as 69 mcg Quatrefolic® |
117 mcg DFE |
147% |
78% |
59% | |
|
VITAMIN B12 (as Cyanocobalamin) |
2 mcg |
400% |
222% |
167% | |
|
FLUORIDE (as Sodium Fluoride) |
0.25 mg |
50% |
36% |
25% | |
Other Ingredients: Purified Water, Glycerin, Sorbitol, Sucralose, Sodium Benzoate, Potassium Sorbate, Sodium Carbonate, Monoammonium glycyrrhizinate, Grape Flavor.
INDICATIONS & USAGE
Florafol™ Pediatric 0.25 mg/mL Fluoride, is a multivitamin and fluoride prescription product providing essential vitamins, minerals and fluoride.
CONTRAINDICATIONS
Do not use in areas where drinking water exceeds 0.6 ppm Fluoride. Do not administer to pediatric patients less than 6 months old.
PRECAUTIONS
General
Do not use this product if you are allergic to any of the ingredients. The suggested dose should not be exceeded, since dental fluorosis may result from continued ingestion of large amounts of fluoride. Do not eat or drink dairy products within one hour of fluoride administration since these may decrease effectiveness.
Folic Acid
Folic Acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress.
DOSAGE AND ADMINISTRATION
See schedule below to determine dosage. Administer orally as prescribed by a healthcare practitioner. Use with calibrated dropper for measuring doses.
Fluoride Ion Level In Drinking Water (ppm)*
|
Age |
Less than 0.3ppm |
0.3-0.6 ppm |
Greater than 0.6 ppm |
|
Birth-6 months 6 months-3 years 3-6 years |
None 0.25 mg/day† 0.50 mg/day† |
None None 0.25 mg/day |
None None None |
|
6-16 years |
1.0 mg/day |
0.50 mg/day |
None |
* 1.0 part per million (ppm) = 1 milligram/liter (mg/L)
† 2.2 mg sodium fluoride contains 1 mg fluoride ion
Dietary Fluoride Supplement dosing schedule approved by the American Dental Association (ADA), American Academy of Pediatrics (AAP) & American Academy of Pediatric Dentistry (AAPD).
DESCRIPTION
Florafol™ Pediatric 0.25 mg/mL Fluoride, multivitamin and fluoride liquid drops is a dye free and gluten free grape flavored product.
HOW SUPPLIED
Florafol™ Pediatric 0.25 mg/mL Fluoride is supplied in 50 mL (1-2/3 fl. oz.) child-resistant bottles with accompanying calibrated dropper (Product Code: 81279-100-50).
Rx Only
STORAGE
Store in a cool, dry place at room temperature 68 °F to 77 °F (20 °C to 25 °C) away from direct heat and sunlight.
Do not refrigerate this product.
SHAKE WELL BEFORE USE
CAUTION
Do not use this product if you are allergic to any of the ingredients.
Take this product at least 2 hours before or after taking any products containing calcium (including milk, yogurt, other dairy products) or aluminum/magnesium hydroxide (e.g., certain antacids/laxatives) since these may decrease effectiveness. Prolonged daily ingestion of excessive fluoride may result in varying degrees of dental fluorosis.