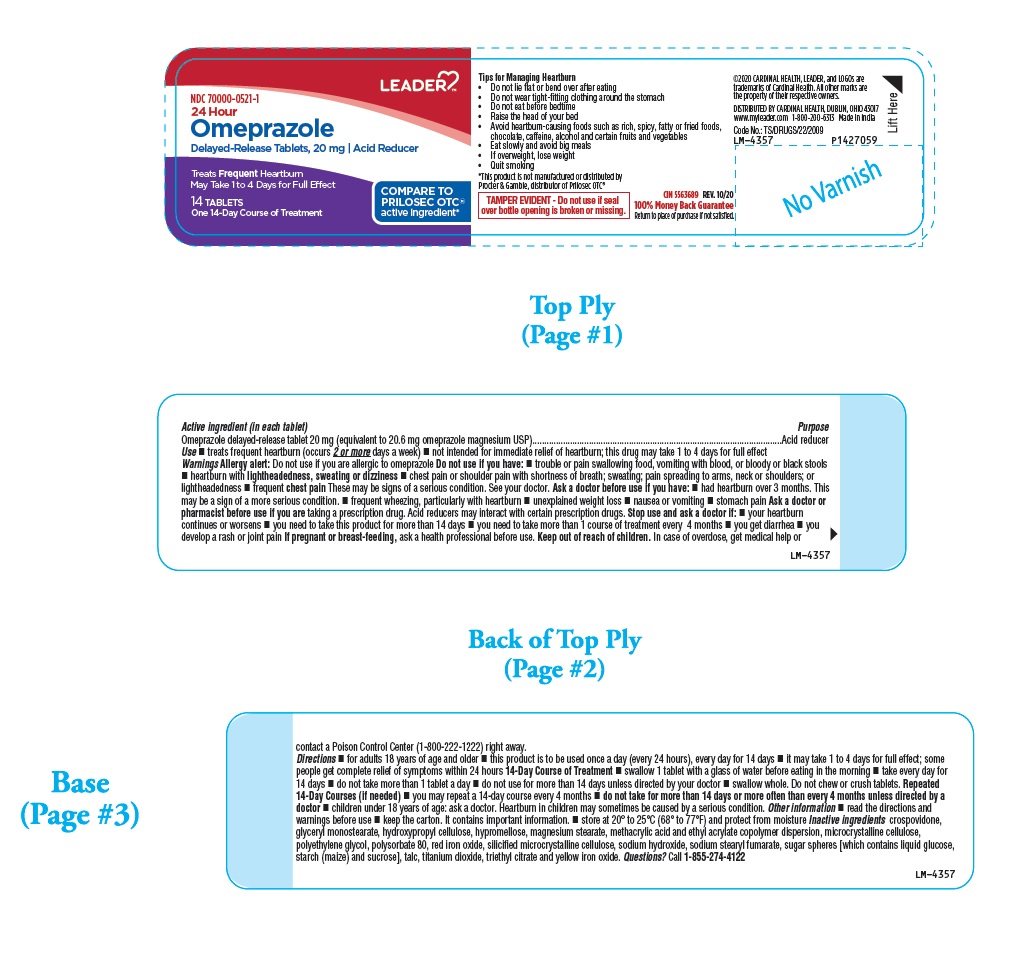
Tips for Managing Heartburn
- Do not lie flat or bend over after eating
- Do not wear tight-fitting clothing around the stomach
- Do not eat before bedtime
- Raise the head of your bed
- Avoid heartburn-causing foods such as rich, spicy, fatty or fried foods, chocolate, caffeine, alcohol and certain fruits and vegetables
- Eat slowly and avoid big meals
- If overweight, lose weight
- Quit smoking
Active ingredient (in each tablet)
Omeprazole delayed-release tablet 20 mg (equivalent to 20.6 mg omeprazole magnesium USP)
Use
- treats frequent heartburn (occurs 2 or more days a week)
- not intended for immediate relief of heartburn; this drug may take 1 to 4 days for full effect
Do not use if you have:
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have:
- had heartburn over 3 months. This may be a sign of a more serious condition.
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Ask a doctor or pharmacist before use if you are
taking a prescription drug.
Acid reducers may interact with certain prescription drugs.
Stop use and ask a doctor if:
- your heartburn continues or worsens
- you need to take this product for more than 14 days
- you need to take more than 1 course of treatment every 4 months
- you get diarrhea
- you develop a rash or joint pain
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
- for adults 18 years of age and older
- this product is to be used once a day (every 24 hours), every day for 14 days
- it may take 1 to 4 days for full effect; some people get complete relief of symptoms within 24 hours
14-Day Course of Treatment
- swallow 1 tablet with a glass of water before eating in the morning
- take every day for 14 days
- do not take more than 1 tablet a day
- do not use for more than 14 days unless directed by your doctor
- swallow whole. Do not chew or crush tablets.
Repeated 14-Day Courses (if needed)
- you may repeat a 14-day course every 4 months
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- children under 18 years of age: ask a doctor. Heartburn in children may sometimes be caused by a serious condition.
Other information
- read the directions and warnings before use
- keep the carton. It contains important information.
- store at 20º to 25ºC (68º to 77º F) and protect from moisture
Inactive ingredients
crospovidone, glyceryl monostearate, hydroxypropyl cellulose, hypromellose, magnesium stearate, methacrylic acid and ethyl acrylate copolymer dispersion, microcrystalline cellulose, polyethylene glycol, polysorbate 80, red iron oxide, silicified microcrystalline cellulose, sodium hydroxide, sodium stearyl fumarate, sugar spheres [which contains liquid glucose, starch (maize) and sucrose], talc, titanium dioxide, triethyl citrate and yellow iron oxide.
Questions?
Call 1-855-274-4122
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com 1-800-200-6313
Essential to Care™ since 1979
Made in India
Code: TS/DRUGS/22/2009
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (14 Tablet Bottle)
LEADERTM
NDC 70000-0521-1
24 Hour
Omeprazole
Delayed-Release Tablets, 20 mg | Acid Reducer
Treats Frequent Heartburn
May Take 1 to 4 Days for Full Effect
COMPARE TO
PRILOSEC OTC®
active ingredient*
14 TABLETS
One 14-Day Course of Treatment
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg Container Carton Label
LEADERTM
NDC 70000-0521-1
24 Hour
Omeprazole
Delayed-Release Tablets, 20 mg
Acid Reducer
Actual Size
Treats Frequent Heartburn
May Take 1 to 4 Days
for Full Effect
COMPARE TO
PRILOSEC OTC®
active ingredients*
100% Money
Back Guarantee
14 TABLETS
One 14-Day Course
of Treatment

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg Blister Carton Label
LEADERTM
NDC 70000-0521-2
24 Hour
Omeprazole
Delayed-Release Tablets, 20 mg
Acid Reducer
Treats Frequent Heartburn
May Take 1 to 4 Days
for Full Effect
COMPARE TO
PRILOSEC OTC®
active ingredients*
100% Money
Back Guarantee
Actual Size
14(1x14) TABLETS
One 14-Day Course of Treatment
