DESCRIPTION
Dexamethasone Sodium Phosphate Injection, USP is a sterile, clear, colorless solution, essentially free from visible particles and a water-soluble inorganic ester of dexamethasone which produces a rapid response even when injected intramuscularly.
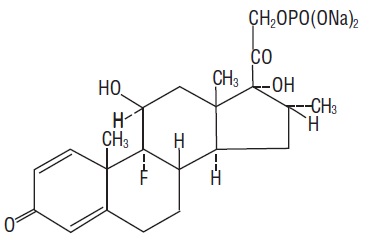
Dexamethasone Sodium Phosphate USP, C22H28FNa2O8P, has a molecular weight of 516.41 and chemically is Pregn-4-ene-3, 20-dione, 9-fluoro-11, 17-dihydroxy-16-methyl-21 (phosphonooxy)-, disodium salt, (11β, 16α).
It occurs as a white to practically white powder, is exceedingly hygroscopic, is soluble in water and its solutions have a pH between 7.0 and 8.5. It has the following structural formula:
Dexamethasone Sodium Phosphate Injection, USP is available in 10 mg/mL concentration.
Each mL of Dexamethasone Sodium Phosphate Injection USP, 10 mg/mL, contains 10.93 mg dexamethasone sodium phosphate, USP equivalent to 10 mg dexamethasone phosphate; 10 mg benzyl alcohol; Made isotonic with 13.5 mg sodium citrate, dihydrate; and Water for Injection, q.s. pH adjusted with citric acid or sodium hydroxide.
ACTIONS — Naturally occurring glucocorticoids (hydrocortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs are primarily used for their potent anti-inflammatory effects in disorders of many organ systems.
Glucocorticoids cause profound and varied metabolic effects. In addition, they modify the body’s immune responses to diverse stimuli.
INDICATIONS AND USAGE
A. Intravenous or intramuscular administration. When oral therapy is not feasible and the strength, dosage form, and route of administration of the drug reasonably lend the preparation to the treatment of the condition, those products labeled for intravenous or intramuscular use are indicated as follows:
1. Endocrine disorders. Primary or secondary adrenocortical insufficiency (hydrocortisone or cortisone is the drug of choice; synthetic analogs may be used in conjunction with mineralocorticoids where applicable; in infancy, mineralocorticoid supplementation is of particular importance).
Acute adrenocortical insufficiency (hydrocortisone or cortisone is the drug of choice; mineralocorticoid supplementation may be necessary, particularly when synthetic analogs are used).
Preoperatively, and in the event of serious trauma or illness, in patients with known adrenal insufficiency or when adrenocortical reserve is doubtful.
Shock unresponsive to conventional therapy if adrenocortical insufficiency exists or is suspected.
Congenital adrenal hyperplasia.
Nonsuppurative thyroiditis.
Hypercalcemia associated with cancer.
2. Rheumatic disorders. As adjunctive therapy for short-term administration (to tide the patient over an acute episode or exacerbation) in:
Post-traumatic osteoarthritis.
Synovitis of osteoarthritis.
Rheumatoid arthritis, including juvenile rheumatoid arthritis (selected cases may require low-dose maintenance therapy).
Acute and subacute bursitis.
Epicondylitis.
Acute nonspecific tenosynovitis.
Acute gouty arthritis.
Psoriatic arthritis.
Ankylosing spondylitis.
3. Collagen diseases. During an exacerbation or as maintenance therapy in selected cases of:
Systemic lupus erythematosus.
Acute rheumatic carditis.
4. Dermatologic diseases.
Pemphigus.
Severe erythema multiforme (Stevens-Johnson Syndrome).
Exfoliative dermatitis.
Bullous dermatitis herpetiformis.
Severe seborrheic dermatitis.
Severe psoriasis.
Mycosis fungoides.
5. Allergic states. Control of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment in:
Bronchial asthma.
Contact dermatitis.
Atopic dermatitis.
Serum sickness.
Seasonal or perennial allergic rhinitis.
Drug hypersensitivity reactions.
Urticarial transfusion reactions.
Acute noninfectious laryngeal edema (epinephrine is the drug of first choice).
6. Ophthalmic diseases. Severe acute and chronic allergic and inflammatory processes involving the eye, such as:
Herpes zoster ophthalmicus.
Iritis, iridocyclitis.
Chorioretinitis.
Diffuse posterior uveitis and choroiditis.
Optic neuritis.
Sympathetic ophthalmia.
Anterior segment inflammation.
Allergic conjunctivitis.
Allergic corneal marginal ulcers.
Keratitis.
7. Gastrointestinal diseases. To tide the patient over a critical period of the disease in:
Ulcerative colitis (systemic therapy).
Regional enteritis (systemic therapy).
8. Respiratory diseases:
Symptomatic Sarcoidosis.
Berylliosis.
Fulminating or disseminated pulmonary tuberculosis when used concurrently with appropriate anti-tuberculosis chemotherapy.
Loeffler's syndrome not manageable by other means.
Aspiration pneumonitis.
9. Hematologic disorders:
Acquired (autoimmune) hemolytic anemia.
Idiopathic thrombocytopenic purpura in adults (I.V. only; I.M. administration is contraindicated).
Secondary thrombocytopenia in adults.
Erythroblastopenia (RBC anemia).
Congenital (erythroid) hypoplastic anemia.
10. Neoplastic diseases. For palliative management of:
Leukemias and lymphomas in adults.
Acute leukemia of childhood.
11. Edematous states. To induce diuresis or remission of proteinuria in the nephrotic syndrome, without uremia, of the idiopathic type or that due to lupus erythematosus.
12. Nervous system.
Acute exacerbations of multiple sclerosis.
13. Miscellaneous.
Tuberculous meningitis with subarachnoid block or impending block when used concurrently with appropriate anti-tuberculosis chemotherapy.
Trichinosis with neurologic or myocardial involvement.
Diagnostic testing of adrenocortical hyperfunction.
Cerebral edema of diverse etiologies in conjunction with adequate neurological evaluation and management.
WARNINGS
Serious Neurologic Adverse Reactions with Epidural Administration
Serious neurologic events, some resulting in death, have been reported with epidural injection of corticosteroids. Specific events reported include, but are not limited to, spinal cord infarction, paraplegia, quadriplegia, cortical blindness, and stroke. These serious neurologic events have been reported with and without use of fluoroscopy. The safety and effectiveness of epidural administration of corticosteroids have not been established, and corticosteroids are not approved for this use.
In patients on corticosteroid therapy subject to any unusual stress, increased dosage of rapidly acting corticosteroids before, during and after the stressful situation is indicated.
Immunosuppression and Increased Risk of Infection
Corticosteroids, including dexamethasone sodium phosphate, suppress the immune system and increase the risk of infection with any pathogen, including viral, bacterial, fungal, protozoan, or helminthic pathogens. Corticosteroids can:
- Reduce resistance to new infections
- Exacerbate existing infections
- Increase the risk of disseminated infections
- Increase the risk of reactivation or exacerbation of latent infections
- Mask some signs of infection
Corticosteroid-associated infections can be mild but can be severe and at times fatal. The rate of infectious complications increases with increasing corticosteroid dosages.
Monitor for the development of infection and consider dexamethasone sodium phosphate withdrawal or dosage reduction as needed.
Tuberculosis
If dexamethasone sodium phosphate is used to treat a condition in patients with latent tuberculosis or tuberculin reactivity, tuberculosis may occur. Closely monitor such patients for reactivation. During prolonged therapy, patients with latent tuberculosis or tuberculin reactivity should receive chemoprophylaxis.
Varicella Zoster and Measles Viral Infections
Varicella and measles can have a serious or even fatal course in non-immune patients taking corticosteroids, including dexamethasone sodium phosphate. In corticosteroid-treated patients who have not had these diseases or are non-immune, particular care should be taken to avoid exposure to varicella and measles:
- If a dexamethasone sodium phosphate-treated patient is exposed to varicella, prophylaxis with varicella zoster immune globulin may be indicated. If varicella develops, treatment with antiviral agents may be considered.
- If a dexamethasone sodium phosphate-treated patient is exposed to measles, prophylaxis with immunoglobulin may be indicated.
Hepatitis B Virus Reactivation
Hepatitis B virus reactivation can occur in patients who are hepatitis B carriers treated with immunosuppressive dosages of corticosteroids, including dexamethasone sodium phosphate. Reactivation can also occur infrequently in corticosteroid-treated patients who appear to have resolved hepatitis B infection.
Screen patients for hepatitis B infection before initiating immunosuppressive (e.g., prolonged) treatment with dexamethasone sodium phosphate. For patients who show evidence of hepatitis B infection, recommend consultation with physicians with expertise in managing hepatitis B regarding monitoring and consideration for hepatitis B antiviral therapy.
Fungal Infections
Corticosteroids, including dexamethasone sodium phosphate, may exacerbate systemic fungal infections; therefore, avoid dexamethasone sodium phosphate use in the presence of such infections unless dexamethasone sodium phosphate is needed to control drug reactions. For patients on chronic dexamethasone sodium phosphate therapy who develop systemic fungal infections, dexamethasone sodium phosphate withdrawal or dosage reduction is recommended.
Amebiasis
Corticosteroids, including dexamethasone sodium phosphate, may activate latent amebiasis. Therefore, it is recommended that latent amebiasis or active amebiasis be ruled out before initiating dexamethasone sodium phosphate in patients who have spent time in the tropics or patients with unexplained diarrhea.
Strongyloides Infestation
Corticosteroids, including dexamethasone sodium phosphate, should be used with great care in patients with known or suspected Strongyloides (threadworm) infestation. In such patients, corticosteroid-induced immunosuppression may lead to Strongyloides hyperinfection and dissemination with widespread larval migration, often accompanied by severe enterocolitis and potentially fatal gram-negative septicemia.
Cerebral Malaria
Avoid corticosteroids, including dexamethasone sodium phosphate, in patients with cerebral malaria.
Kaposi’s Sarcoma
Kaposi’s sarcoma has been reported to occur in patients receiving corticosteroid therapy, most often for chronic conditions. Discontinuation of corticosteroids may result in clinical improvement of Kaposi’s sarcoma.
Prolonged use of corticosteroids may produce posterior subcapsular cataracts, glaucoma with possible damage to the optic nerves, and may enhance the establishment of secondary ocular infections due to fungi or viruses.
Usage in Pregnancy. Since adequate human reproduction studies have not been done with corticosteroids, use of these drugs in pregnancy, nursing mothers or women of childbearing potential requires that the possible benefits of the drug be weighed against the potential hazards to the mother and embryo or fetus. Infants born of mothers who have received substantial doses of corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism.
Average and large doses of cortisone or hydrocortisone can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium. These effects are less likely to occur with the synthetic derivatives except when used in large doses. Patients with a stressed myocardium should be observed carefully and the drug administered slowly since premature ventricular contractions may occur with rapid administration. Dietary salt restriction and potassium supplementation may be necessary. All corticosteroids increase calcium excretion.
While on corticosteroid therapy patients should not be vaccinated against smallpox. Other immunization procedures should not be undertaken in patients who are on corticosteroids, especially in high doses, because of possible hazards of neurological complications and lack of antibody response.
Because rare instances of anaphylactoid reactions have occurred in patients receiving parenteral corticosteroid therapy, appropriate precautionary measures should be taken prior to administration, especially when the patient has a history of allergy to any drug.
PRECAUTIONS
Drug-induced secondary adrenocortical insufficiency may be minimized by gradual reduction of dosage. This type of relative insufficiency may persist for months after discontinuation of therapy; therefore, in any situation of stress occurring during that period, hormone therapy should be reinstituted. Since mineralocorticoid secretion may be impaired, salt and/or a mineralocorticoid should be administered concurrently.
There is an enhanced effect of corticosteroids in patients with hypothyroidism and in those with cirrhosis.
Corticosteroids should be used cautiously in patients with ocular herpes simplex for fear of corneal perforation.
The lowest possible dose of corticosteroid should be used to control the condition under treatment, and when reduction in dosage is possible, the reduction must be gradual.
Psychic derangements may appear when corticosteroids are used ranging from euphoria, insomnia, mood swings, personality changes, and severe depression to frank psychotic manifestations. Also, existing emotional instability or psychotic tendencies may be aggravated by corticosteroids.
Aspirin should be used cautiously in conjunction with corticosteroids in hypoprothrombinemia.
Steroids should be used with caution in nonspecific ulcerative colitis, if there is a probability of impending perforation, abscess or other pyogenic infection, also in diverticulitis, fresh intestinal anastomoses, active or latent peptic ulcer, renal insufficiency, hypertension, osteoporosis, and myasthenia gravis.
Growth and development of infants and children on prolonged corticosteroid therapy should be carefully followed.
Patients who are on immunosuppressant doses of corticosteroids should be warned to avoid exposure to chickenpox or measles and, if exposed, to obtain medical advice.
Intra-articular injection of a corticosteroid may produce systemic as well as local effects.
Appropriate examination of any joint fluid present is necessary to exclude a septic process.
A marked increase in pain accompanied by local swelling, further restriction of joint motion, fever, and malaise are suggestive of septic arthritis. If this complication occurs and the diagnosis of sepsis is confirmed, appropriate antimicrobial therapy should be instituted.
Local injection of a steroid into a previously infected joint is to be avoided. Corticosteroids should not be injected into unstable joints.
Although controlled clinical trials have shown corticosteroids to be effective in speeding the resolution of acute exacerbations of multiple sclerosis they do not show that they affect the ultimate outcome or natural history of the disease. The studies do show that relatively high doses of corticosteroids are necessary to demonstrate a significant effect. (See Dosage and Administration Section).
Since complications of treatment with glucocorticoids are dependent on the size of the dose and the duration of treatment a risk/benefit decision must be made in each individual case as to dose and duration of treatment and as to whether daily or intermittent therapy should be used.
ADVERSE REACTIONS
Fluid and electrolyte disturbances:
Sodium retention
Fluid retention
Congestive heart failure in susceptible patients
Potassium loss
Hypokalemic alkalosis
Hypertension
Musculoskeletal:
Muscle weakness
Steroid myopathy
Loss of muscle mass
Osteoporosis
Vertebral compression fractures
Aseptic necrosis of femoral and humeral heads
Pathologic fracture of long bones
Gastrointestinal:
Peptic ulcer with possible subsequent perforation and hemorrhage
Pancreatitis
Abdominal distention
Ulcerative esophagitis
Dermatological:
Impaired wound healing
Thin fragile skin
Facial erythema
Increased sweating
May suppress reactions to skin tests
Petechiae and ecchymoses
Neurological:
Convulsions
Increased intracranial pressure with papilledema (pseudotumor cerebri) usually after treatment
Vertigo
Headache
Ophthalmic:
Posterior subcapsular cataracts
Increased intraocular pressure
Glaucoma
Endocrine:
Menstrual irregularities
Development of cushingoid state
Suppression of growth in children
Secondary adrenocortical and pituitary unresponsiveness, particularly in times of stress, as in trauma, surgery, or illness
Decreased carbohydrate tolerance
Manifestations of latent diabetes mellitus
Increased requirements for insulin or oral hypoglycemic agents in diabetics
Metabolic:
Negative nitrogen balance due to protein catabolism
Miscellaneous:
Hyperpigmentation or hypopigmentation
Subcutaneous and cutaneous atrophy
Sterile abscess
Charcot-like arthropathy
Itching, burning, tingling in the ano-genital region
DOSAGE AND ADMINISTRATION
A. Intravenous or intramuscular administration. The initial dosage of Dexamethasone Sodium Phosphate Injection, USP may vary from 0.5 mg/day to 9 mg/day depending on the specific disease entity being treated. In situations of less severity, lower doses will generally suffice while in selected patients higher initial doses may be required. Usually the parenteral dosage ranges are one-third to one-half the oral dose given every 12 hours. However, in certain overwhelming, acute, life-threatening situations, administration of dosages exceeding the usual dosages may be justified and may be in multiples of the oral dosages.
For the treatment of unresponsive shock high pharmacologic doses of this product are currently recommended. Reported regimens range from 1 to 6 mg/kg of body weight as a single intravenous injection to 40 mg initially followed by repeat intravenous injection every 2 to 6 hours while shock persists.
For the treatment of cerebral edema in adults an initial intravenous dose of 10 mg is recommended followed by 4 mg intramuscularly every six hours until maximum response has been noted. This regimen may be continued for several days postoperatively in patients requiring brain surgery. Oral dexamethasone, 1 to 3 mg t.i.d., should be given as soon as possible and dosage tapered off over a period of five to seven days. Nonoperative cases may require continuous therapy to remain free of symptoms of increased intracranial pressure. The smallest effective dose should be used in children, preferably orally. This may approximate 0.2 mg/kg/24 hours in divided doses.
In treatment of acute exacerbations of multiple sclerosis daily doses of 200 mg of prednisolone for a week followed by 80 mg every other day or 4 to 8 mg dexamethasone every other day for 1 month have been shown to be effective.
The initial dosage should be maintained or adjusted until a satisfactory response is noted. If after a reasonable period of time there is a lack of satisfactory clinical response, Dexamethasone Sodium Phosphate Injection, USP should be discontinued and the patient transferred to other appropriate therapy. It should be emphasized that dosage requirements are variable and must be individualized on the basis of the disease under treatment and the response of the patient.
After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage which will maintain an adequate clinical response is reached. It should be kept in mind that constant monitoring is needed in regard to drug dosage. Included in the situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient’s individual drug responsiveness and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this later situation it may be necessary to increase the dosage of Dexamethasone Sodium Phosphate Injection, USP for a period of time consistent with the patient’s condition. If after a long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
HOW SUPPLIED
Dexamethasone Sodium Phosphate Injection, USP is a sterile, clear, colorless solution, essentially free from visible particles and is supplied as follows:
100 mg per 10 mL (10 mg per mL)
10 mL Multiple-Dose Vials
in a carton of 10 NDC 55150-305-10
Protect from light. Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
The vial stopper is not made with natural rubber latex.
Distributed by:
Eugia US LLC
279 Princeton-Hightstown Rd.
E. Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
Revised: February 2024