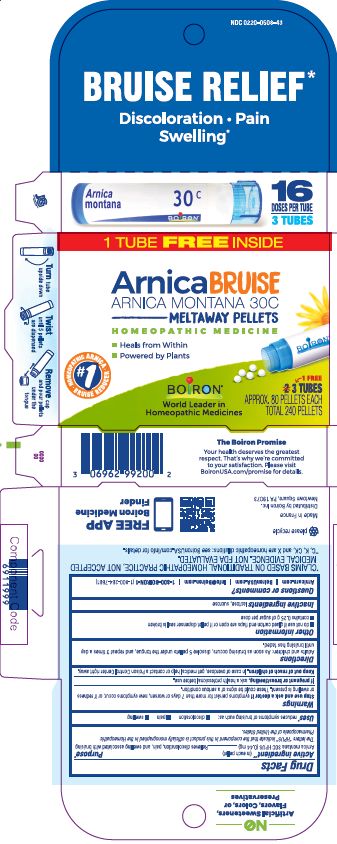
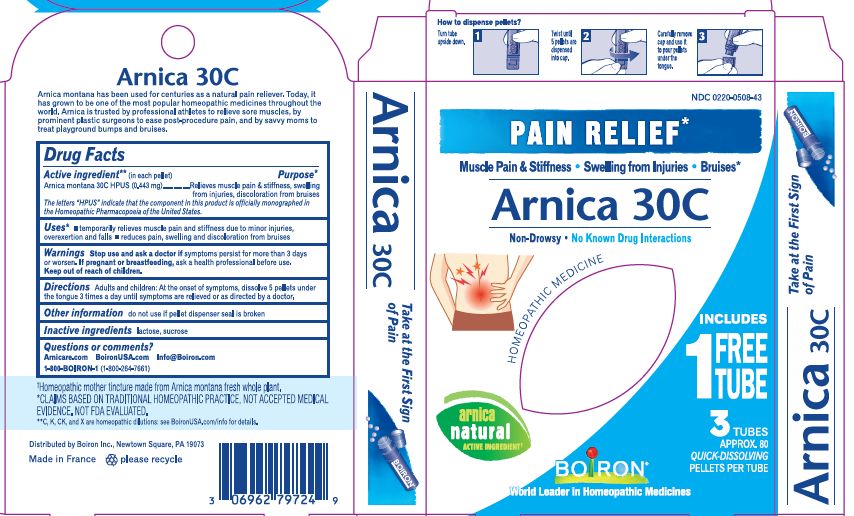
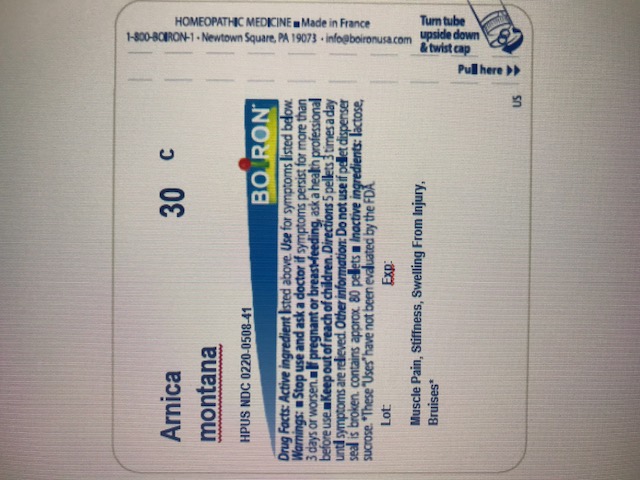
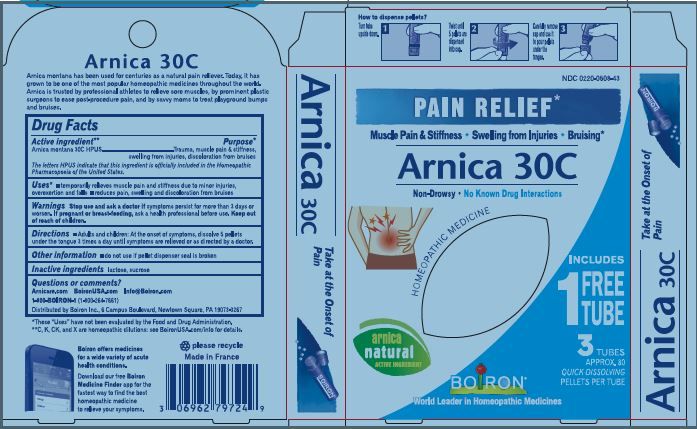
Active ingredient** (in each pellet)
Arnica montana 30C HPUS (0.443 mg)
The letters "HPUS" indicate that the component in this product is officially monographed in Homeopathic Pharmacopoeia of the United States.
Purpose*
Relieves muscle pain & stiffness, swelling from injuries, discoloration from bruises
Relieves discoloration, pain, and swelling with bruising
Uses*
temporarily relieves muscle pain and stiffness due to:
- minor injuries
- overexertion
- falls
reduces symptoms of bruising such as:
- pain
- swelling
- discoloration
Stop use and ask a doctor if symptoms persist for more than 3 days or worsen, new symptoms occur, or if redness or swelling is present. These could be signs of a serious condition.
Stop use and ask a doctor if symptoms persist for more than 7 days or worsen, new symptoms occur, or if redness or swelling is present. These could be signs of a serious condition.
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Directions
Adults and children: At the onset of symptoms, dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a doctor.
- do not use if glued carton end flaps are open or if the pellet dispenser seal is broken.
- contains 0.25 g of sugar per dose
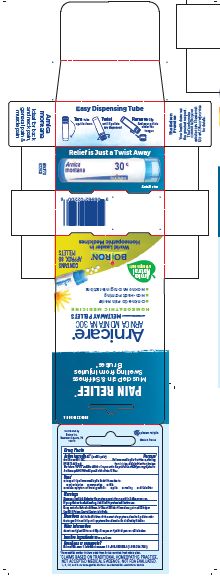
Pain Relief"
Muscle Pain & Stiffness, Swelling from Injuries, Bruises*
Bruise Relief*
Discoloration Pain Swelling*
No Known Drug Interactions
Plant-Powered Relief
No Artificial Colors, Flavors, Sweeteners, or Preservatives
How to dispense pellets? Turn tube upside down Twist until 5 pellets are dispensed Remove the cap and pour pellets under the tongue.
16 Doses per Tube
3 Tubes approx. 80 quick dissolving pellets per tube
Total 240 Pellets
*Claims are based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated
**C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.