Active Ingredient
(in each liquid filled capsule)
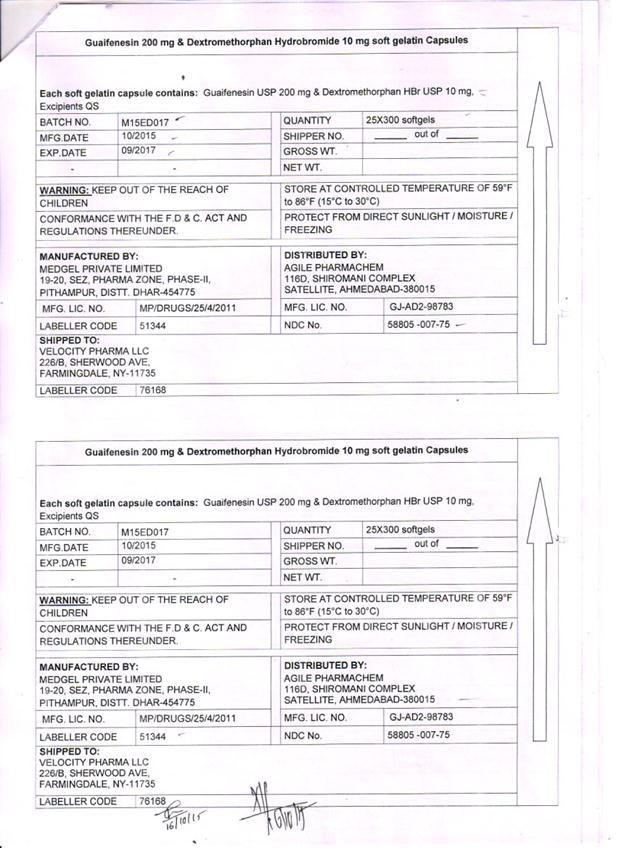
Dextromethorphan Hydrobromide, USP 10mg
Guaifenesin, USP 200mg
Uses
- Temporarily relieves cough due minor throat and bronchial irritation as may occur with a cold
- Helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes
Directions
- Do not take more than 12 capsules in any 24 hour period
- This adult product is not intend for use in children under 12 years of age
Age dose Adults and children 12 years and over 2 capsules every 4 hours Children under 12 years Do not use
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions or parkinson’s disease) or for 2 weeks after stopping the MAOI drug.
If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- cough that occurs with too much phlegm(mucus)
- cough that lasts or is chronic such as occur with smoking, asthma, chronic bronchitis, or emphysema
Stop use and ask a doctor if cough lasts more than 7 days, comes back, or is accompanied by fever, rash, or persistent headache. This could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.