Purpose
- relieves occasional constipation (irregularity)
- generally produces a bowel movement in 1 to 3 days
WARNINGS
Allergy alert: Do not use if you are allergic to polyethylene glycol
Do not use if you have kidney disease, except under the advice and supervision of a doctor
Ask a doctor before use if you have
- nausea, vomiting or abdominal pain
- a sudden change in bowel habits that lasts over 2 weeks
- irritable bowel syndrome
Ask a doctor or pharmacist before use if you are taking a prescription drug
When using this product you may have loose, watery, more frequent stools
Stop use and ask a doctor if
- you have rectal bleeding or your nausea, bloating, cramping or abdominal pain gets worse. These may be signs of a serious condition.
- you get diarrhea
- you need to use a laxative for longer than 1 week
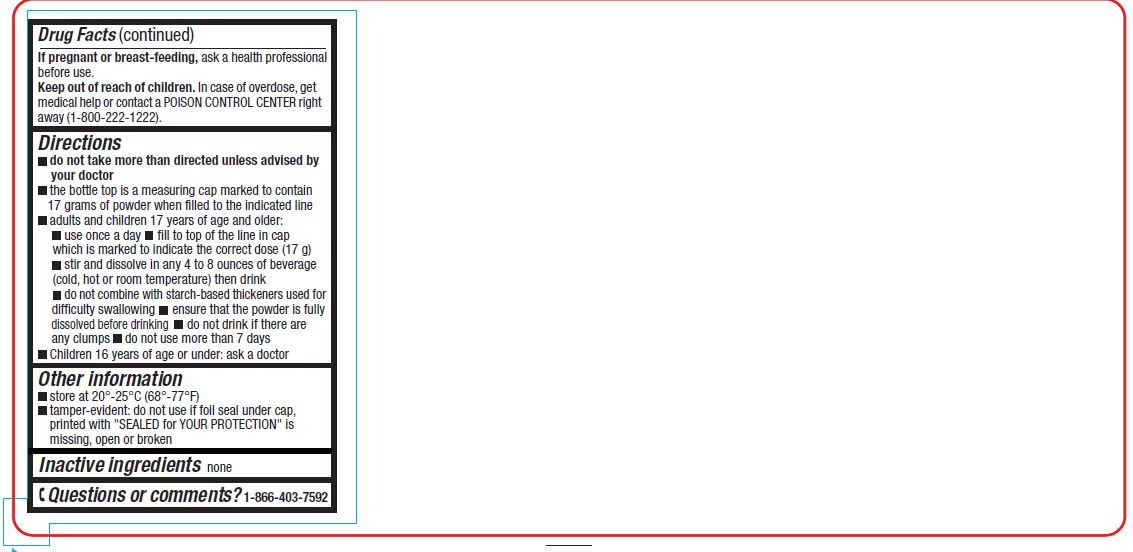
If pregnant or breast-feeding, ask a health professional before use.
Keep out of the reach of children
In case of overdose, get medical help or contact a POISON CONTROL CENTER right away. (1-800-222-1222)
Directions
- do not take more than directed unless advised by your doctor
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line.
- adults and children 17 years of age and older:
- use once a day
- fill to top of line in cap which is marked to indicate the correct dose (17 g)
- stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
- do not combine with starch-based thickeners used for difficulty swallowing
- ensure that the powder is fully dissolved before drinking
- do not drink if there are any clumps
- do not use more than 7 days
- children 16 years of age or under: ask a doctor
- store at 20°- 25°C (68°– 77°F)
- tamper-evident: do not use if printed foil seal under cap, printed with "SEALED for YOUR PROTECTION" is missing, open or broken
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
14 ONCE-DAILY DOSES
NET WT 8.3 OZ (238 g)

Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
50 ONCE-DAILY DOSES
NET WT 29.9 OZ (850 g)

Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
7 ONCE-DAILY DOSES
NET WT 4.1 OZ (119 g)

Original Prescription Strength
GaviLax
Polyethylene Glycol 3350
Powder for Oral Solution, Osmotic Laxative
30 ONCE-DAILY DOSES
NET WT 17.9 OZ (510 g)
