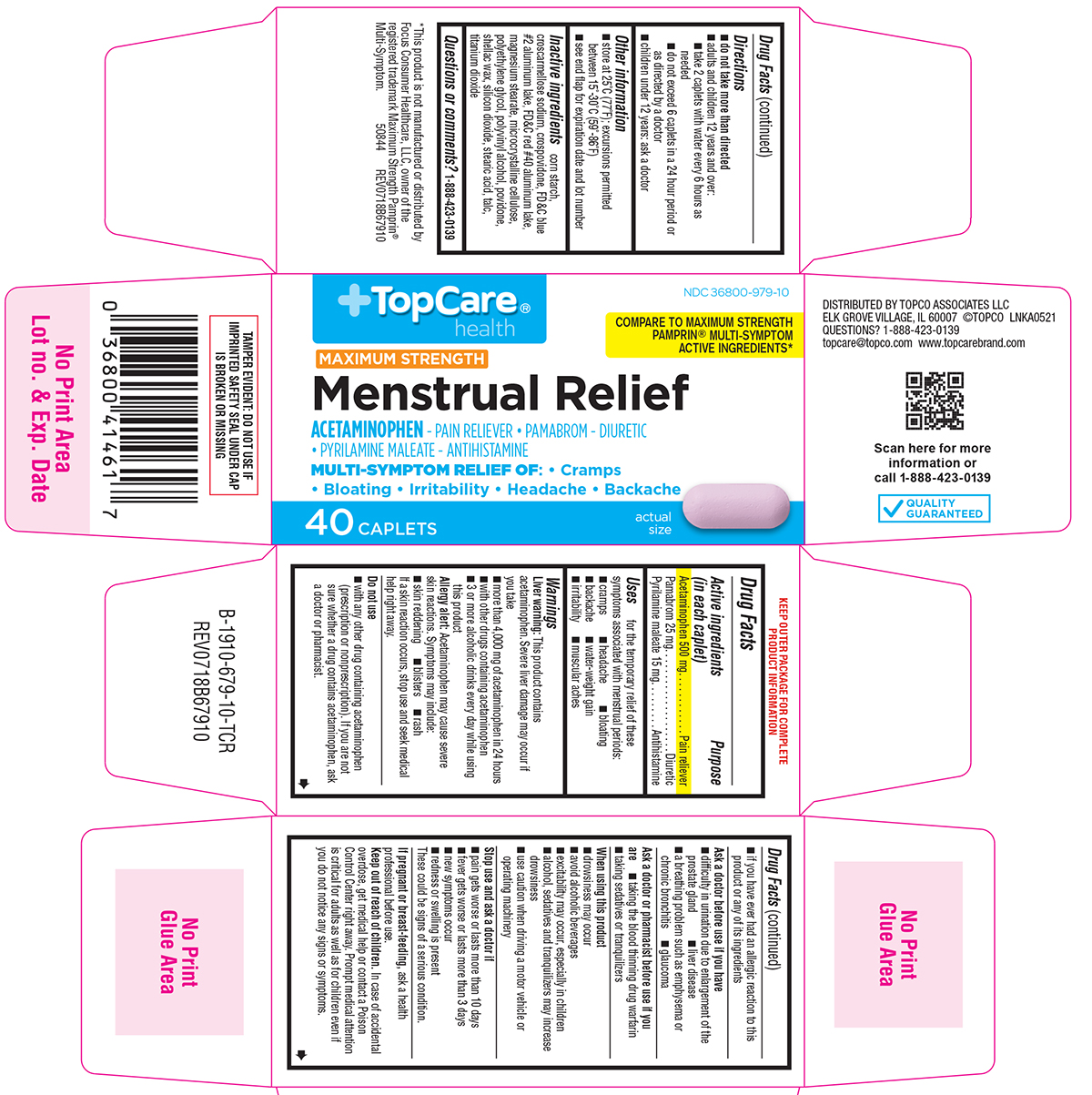
Uses
for the temporary relief of these symptoms associated with menstrual periods:
- cramps
- headache
- bloating
- backache
- water-weight gain
- irritability
- muscular aches
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- difficulty in urination due to enlargement of the prostate gland
- liver disease
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- drowsiness may occur
- avoid alcoholic beverages
- excitability may occur, especially in children
- alcohol, sedatives and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
Directions
- do not take more than directed
- adults and children 12 years and over:
- take 2 caplets with water every 6 hours as needed
- do not exceed 6 caplets in a 24 hour period or as directed by a doctor
- children under 12 years: ask a doctor
Other information
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, croscarmellose sodium, crospovidone, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, shellac wax, silicon dioxide, stearic acid, talc, titanium dioxide
Principal Display Panel
TopCare®
health
NDC 36800-979-10
COMPARE TO MAXIMUM STRENGTH
PAMPRIN® MULTI-SYMPTOM ACTIVE
INGREDIENTS*
MAXIMUM STRENGTH
Menstrual Relief
ACETAMINOPHEN - PAIN RELIEVER • PAMABROM - DIURETIC
• PYRILAMINE MALEATE - ANTIHISTAMINE
MULTI-SYMPTOM RELIEF OF: • Cramps
• Bloating • Irritability • Headache • Backache
40 CAPLETS
actual
size
TAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER CAP
IS BROKEN OR MISSING
DISTRIBUTED BY TOPCO ASSOCIATES LLC
ELK GROVE VILLAGE, IL 60007 ©TOPCO LNKA0521
QUESTIONS? 1-888-423-0139
topcare@topco.com www.topcarebrand.com
QUALITY
GUARANTEED
*This product is not manufactured or distributed by
Focus Consumer Healthcare, LLC, owner of the
registered trademark Maximum Strength Pamprin®
Multi-Symptom. 50844 REV0718B67910
TopCare 44-679