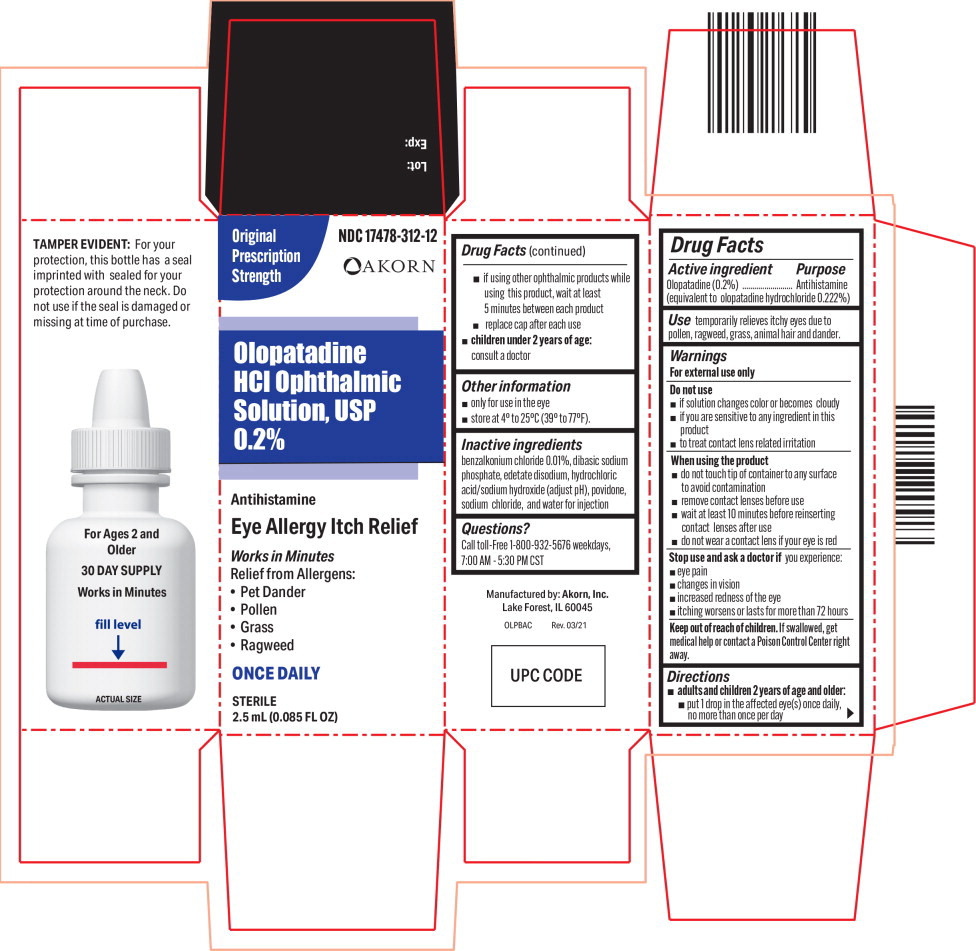
Warnings
For external use only
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
- to treat contact lens related irritation
When using the product
- do not touch tip of container to any surface to avoid contamination
- remove contact lenses before use
- wait at least 10 minutes before reinserting contact lenses after use
- do not wear a contact lens if your eye is red
Directions
-
adults and children 2 years of age and older:
- put 1 drop in the affected eye(s) once daily, no more than once per day
- if using other ophthalmic products while using this product, wait at least 5 minutes between each product
- replace cap after each use
- children under 2 years of age: consult a doctor
Inactive ingredients
benzalkonium chloride 0.01%, dibasic sodium phosphate, edetate disodium, hydrochloric acid/sodium hydroxide (adjust pH), povidone, sodium chloride, and water for injection
Principal Display Panel Text for Container Label:
NDC 17478-312-12
Olopatadine HCl Ophthalmic
Solution, USP 0.2%
Antihistamine
ONCE DAILY
STERILE 2.5 mL (0.085 FL OZ)
Principal Display Panel Text for Carton Label:
Original NDC 17478-312-12
Prescription
Strength Akorn logo
Olopatadine
HCl Ophthalmic
Solution, USP
0.2%
Antihistamine
Eye Allergy Itch Relief
Works in Minutes
Relief from Allergens:
• Pet Dander
• Pollen
• Grass
• Ragweed
ONCE DAILY
STERILE
2.5 mL (0.085 FL OZ)