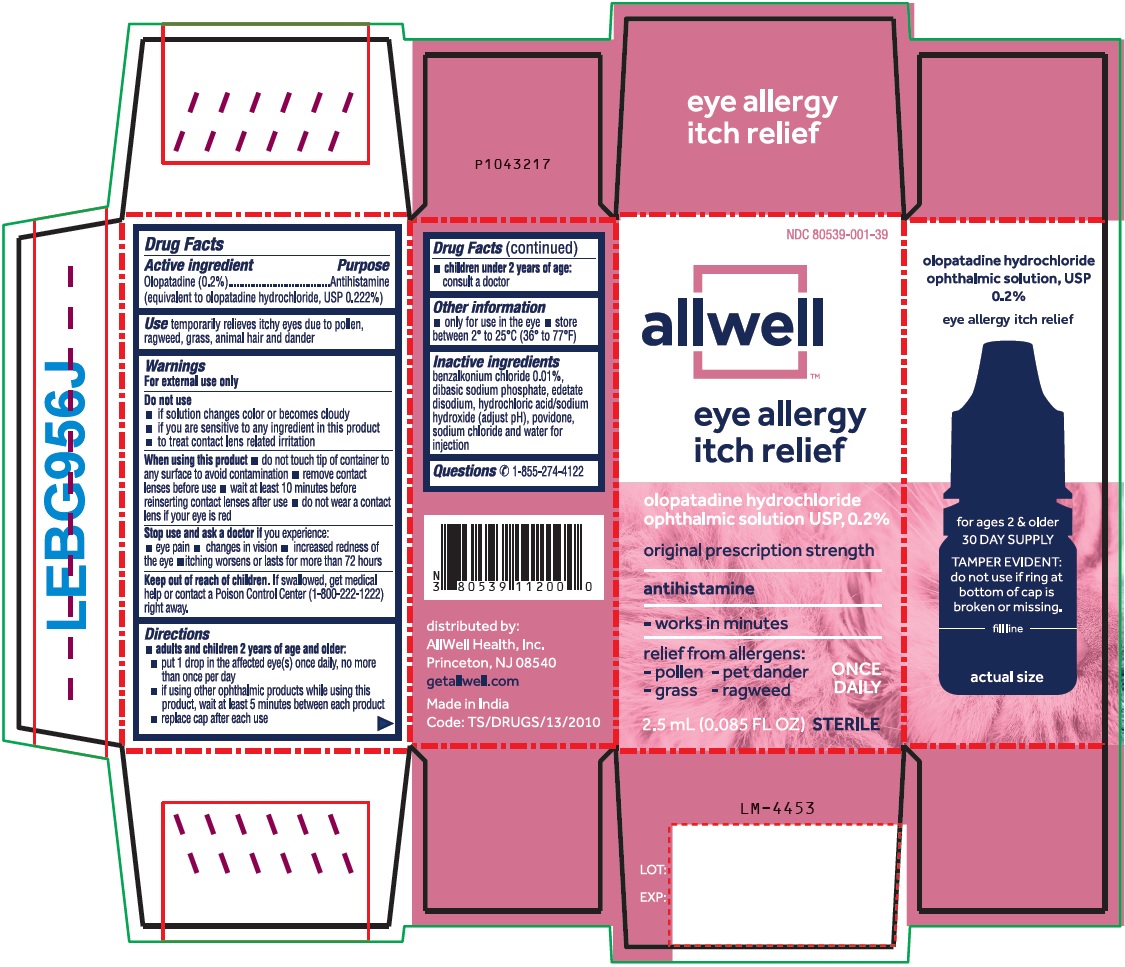
Drug Facts
Active ingredient
Olopatadine (0.2%)
(equivalent to olopatadine hydrochloride, USP 0.222%)
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
- to treat contact lens related irritation
When using this product
- do not touch tip of container to any surface to avoid contamination
- remove contact lenses before use
- wait at least 10 minutes before reinserting contact lenses after use
- do not wear a contact lens if your eye is red
Stop use and ask a doctor if you experience:
- eye pain
- changes in vision
- increased redness of the eye
- itching worsens or lasts for more than 72 hours
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
-
adults and children 2 years of age and older:
- put 1 drop in the affected eye(s) once daily, no more than once per day
- if using other ophthalmic products while using this product, wait at least 5 minutes between each product
- replace cap after each use
- children under 2 years of age:
consult a doctor
Inactive ingredients
benzalkonium chloride 0.01%, dibasic sodium phosphate, edetate disodium, hydrochloric acid/sodium hydroxide (adjust pH), povidone, sodium chloride and water for injection
Questions?
✆ 1-855-274-4122
distributed by:
AllWell Health, Inc.
Princeton, NJ 08540
getallwell.com
Made in India
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-0.2% (2.5 mL Container)
allwell ONCE DAILY
eye allergy itch relief
olopatadine hydrochloride
ophthalmic solution USP, 0.2%
antihistamine
STERILE 2.5 mL (0.085 FL OZ)

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-0.2% (2.5 mL Container Carton)
NDC 80539-001-39
allwell
eye allergy
itch relief
olopatadine hydrochloride
ophthalmic solution USP, 0.2%
original prescription strength
antihistamine
- works in minutes
relief from allergens:
- pollen - pet dander ONCE
- grass - ragweed DAILY
2.5 mL (0.085 FL OZ) STERILE