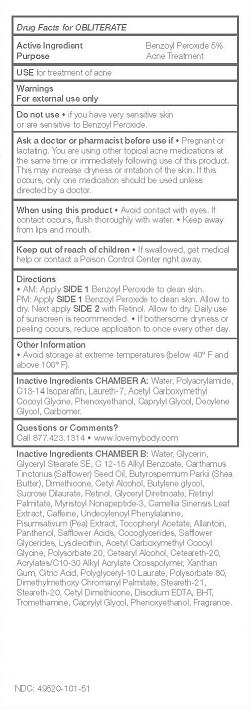
ASK A DOCTOR OR PHARMACIST BEFORE USE IF * PREGNANT OR LACTATING. YOU ARE USING OTHER TOPICAL ACNE MEDICATIONS AT THE SAME TIME OR IMMEDIATELY FOLLOWING USE OF THIS PRODUCT. THIS MAY INCREASE DRYNESS OR IRRITATION OF THE SKIN. IF THIS OCCURS, ONLY ONE MEDICATION SHOULD BE USED UNLESS DIRECTED BY A DOCTOR.
WHEN USING THIS PRODUCT * AVOID CONTACT WITH EYES. IF CONTACT OCCURS, FLUSH THOROUGHLY WITH WATER. * KEEP AWAY FROM LIPS AND MOUTH.
KEEP OUT OF REACH OF CHILDREN * IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
DIRECTIONS
* AM: APPLY SIDE 1 BENZOYL PEROXIDE TO CLEAN SKIN.
PM: APPLY SIDE 1 BENZOYL PEROXIDE TO CLEAN SKIN. ALLOW TO DRY. NEXT APPLY SIDE 2 WITH RETINOL. ALLOW TO DRY. DAILY USE OF SUNSCREEN IS RECOMMENDED. * IF BOTHERSOME DRYNESS OR PEELING OCCURS, REDUCE APPLICATION TO ONCE EVERY OTHER DAY.