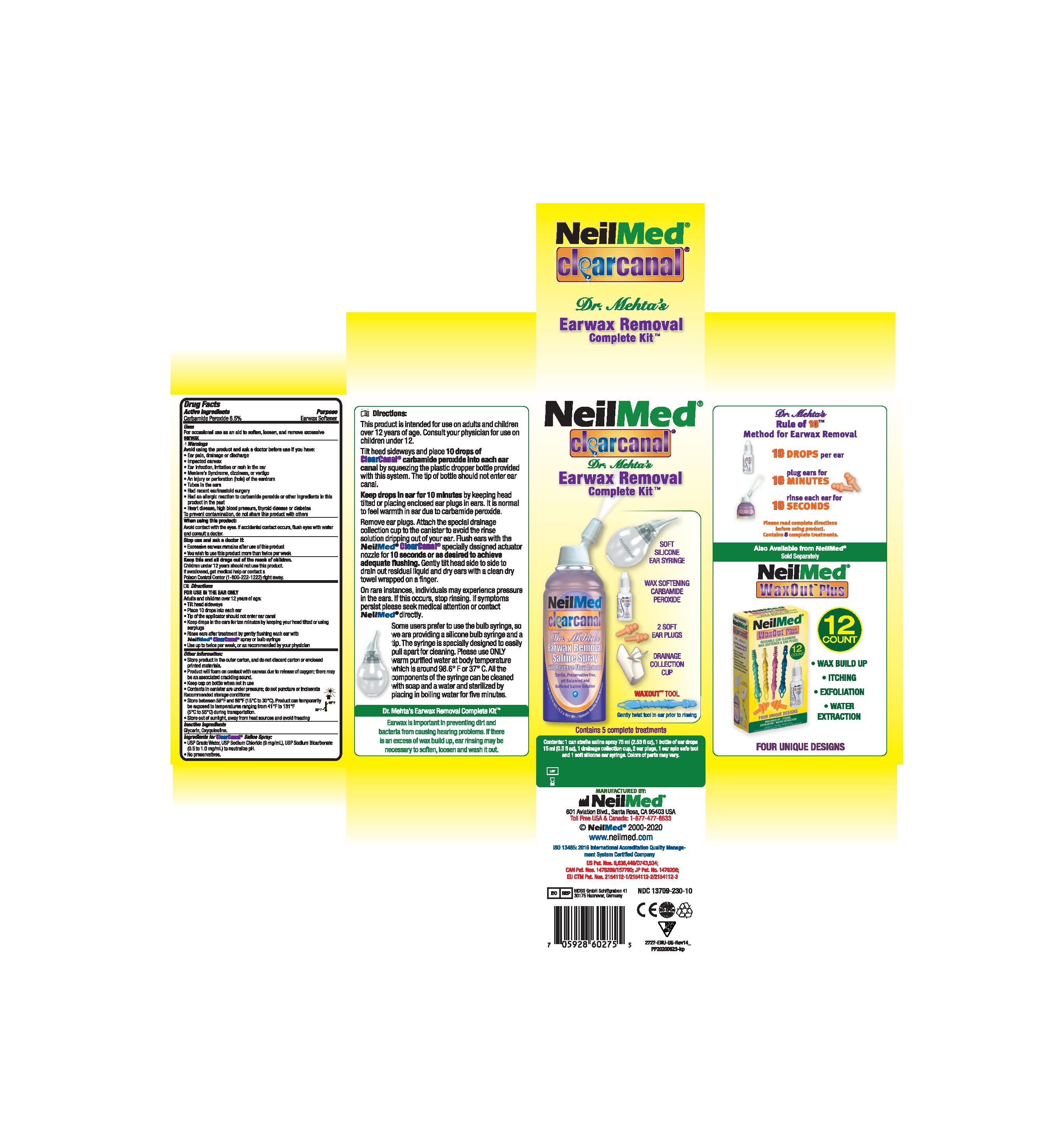
Ear Wax Removal Drops Directions
Step 1 Ear Wax Removal Drops Directions:
- FOR USE IN THE EAR ONLY.
- Adults and children over 12 years of age: tilt head sideways and place 5 to 10 drops into ear.
- Tip of applicator should not enter ear canal.
- Keep drops in ear for several minutes by keeping head tilted or placing enclosed ear plugs in ear.
- Use twice daily for upto 4 days if needed or as directed by a doctor.
- Any wax remaining after treatment may be removed by gently flushing the ear with NeilMed ClearCanal rinsing system with special ear rinse adapter.
- Children under 12 years of age: consult a doctor.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. If accidental contact with eyes occurs, flush eyes with water and consult a doctor.
Do not use:
* If you have had an allergic reaction to Carbamide Peroxide.
* If you have ear drainage or discharge, ear pain, irritation or rash in the ear, or are dizzy; consult a doctor
* If you have an injury or perforation (hole) of the eardrum or after ear surgery, unless directed by a doctor
* For more than four consecutive days
Stop use ad ask a doctor if excessive ear wax remains after use of this product for four consecutive days
Ear Wax Removal Complete System
Ear wax is an important part of your body's system for keeping dirt, bacteria and other things from causing hearing problems. When rinsing is necessary, this cleaning system helps to soften, loosen and remove the excessive ear wax.
The system consists of kit with 1 bottle carbamide peroxide solution 15mL and 1 can sterile saline spray 177mL (or 125mL).
Warnings:
Do not use:
If you have had an allergic reaction to carbamide peroxide.
If you have ear drainage or discharge, ear pain, irritation or rash in the ear, or are dizzy; consult a doctor
If you have an injury or perforation (hole) of the eardrum or after ear surgery, unless directed by a doctor
For more than four consecutive days
When using this product:
void contact with the eyes
Stop use ad ask a doctor if excessive ear wax remains after use of this product for four consecutive days.