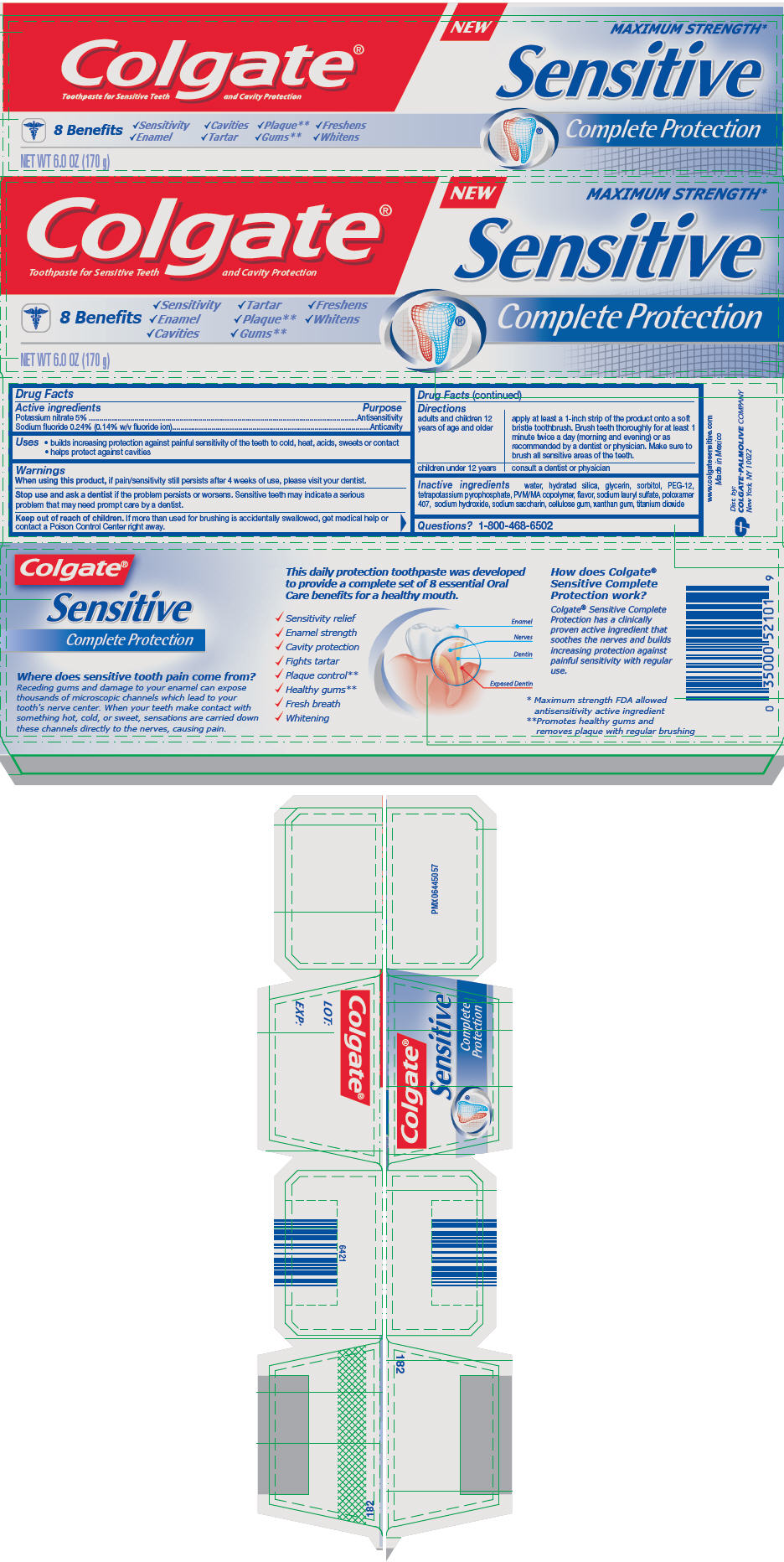
COLGATE SENSITIVE COMPLETE PROTECTION- sodium fluoride and potassium nitrate paste, dentifrice
Colgate-Palmolive Company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
| Active ingredients | Purpose |
| Potassium nitrate 5% | Antisensitivity |
| Sodium fluoride 0.24% (0.14% w/v fluoride ion) | Anticavity |
Uses
- builds increasing protection against painful sensitivity of the teeth to cold, heat, acids, sweets or contact
- helps protect against cavities
Warnings
When using this product, if pain/sensitivity still persists after 4 weeks of use, please visit your dentist.
Stop use and ask a dentist if the problem persists or worsens. Sensitive teeth may indicate a serious problem that may need prompt care by a dentist.
Keep out of reach of children. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Directions
| adults and children 12 years of age and older | apply at least a 1-inch strip of the product onto a soft bristle toothbrush. Brush teeth thoroughly for at least 1 minute twice a day (morning and evening) or as recommended by a dentist or physician. Make sure to brush all sensitive areas of the teeth. |
| children under 12 years | consult a dentist or physician |
Inactive ingredients
water, hydrated silica, glycerin, sorbitol, PEG-12, tetrapotassium pyrophosphate, PVM/MA copolymer, flavor, sodium lauryl sulfate, poloxamer 407, sodium hydroxide, sodium saccharin, cellulose gum, xanthan gum, titanium dioxide
Questions?
1-800-468-6502
Dist. by:
COLGATE-PALMOLIVE COMPANY
New York, NY 10022
PRINCIPAL DISPLAY PANEL - 170 g Tube Carton
Colgate®
Toothpaste for Sensitive Teeth and Cavity Protection
8 Benefits
✓Sensitivity ✓Tartar ✓Freshens
✓Enamel ✓Plaque** ✓Whitens
✓Cavities ✓Gums**
NET WT 6.0 OZ (170 g)
NEW
MAXIMUM STRENGTH*
Sensitive
Complete Protection