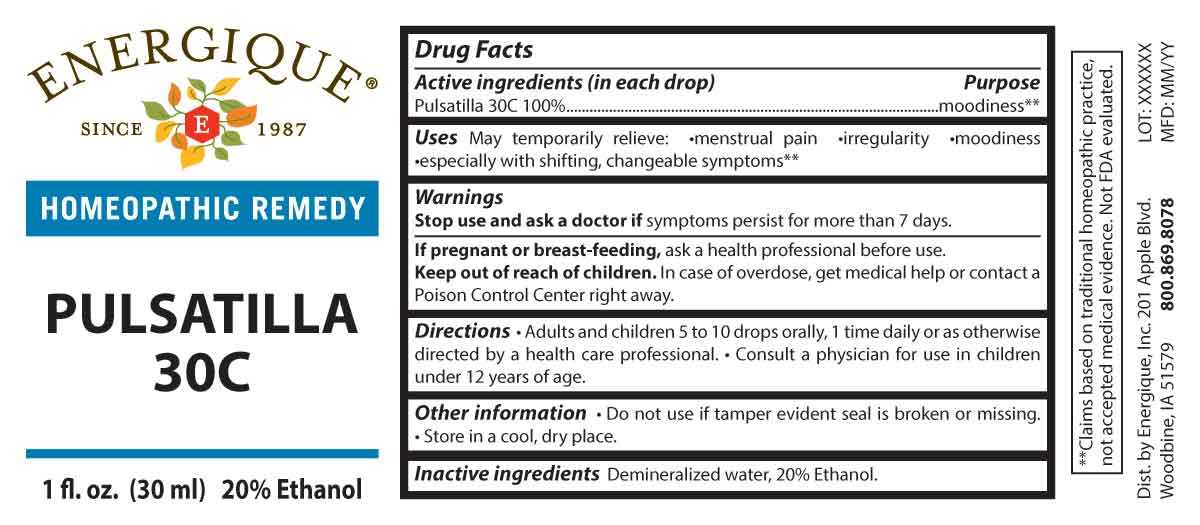
PURPOSE
Pulsatilla 30C - moodiness**
** Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
USES:
May temporarily relieve: • menstrual pain • irregularity • moodiness • especially with shifting, changeable symptoms**** Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
WARNINGS:
Stop use and ask a doctor if symptoms persist for more than 7 days.
If pregnant or breastfeeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
• Do not use if tamper evident seal is broken or missing.
• Store in a cool, dry place.
KEEP OUT OF REACH OF CHILDREN:
In case of overdose, get medical help or contact a Poison Control Center right away.