PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC 65841-734-10 in bottle of 1000 tablets
Pramipexole Dihydrochloride Tablets, 0.125 mg
Rx only
1000 tablets

NDC 65841-735-10 in bottle of 1000 tablets
Pramipexole Dihydrochloride Tablets, 0.25 mg
Rx only
1000 tablets

NDC 65841-736-10 in bottle of 1000 tablets
Pramipexole Dihydrochloride Tablets, 0.5 mg
Rx only
1000 tablets

NDC 65841-737-10 in bottle of 1000 tablets
Pramipexole Dihydrochloride Tablets, 1 mg
Rx only
1000 tablets

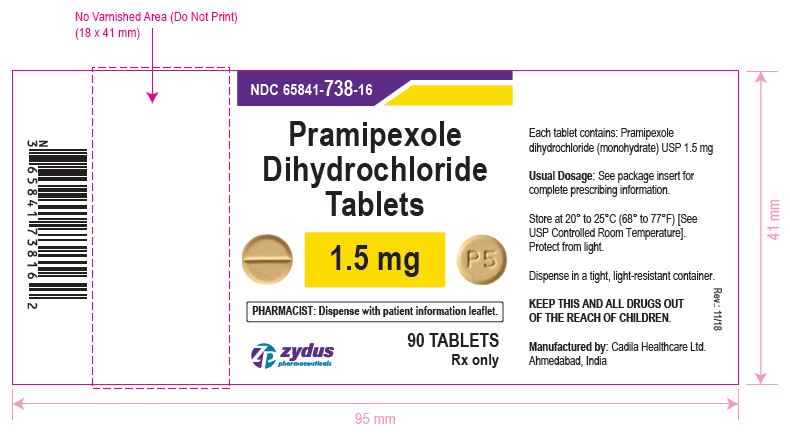
NDC 65841-738-16 in bottle of 90 tablets
Pramipexole Dihydrochloride Tablets, 1.5 mg
Rx only
90 tablets
NDC 65841-844-16 in bottle of 90 tablets
Pramipexole Dihydrochloride Tablets, 0.75 mg
Rx only
