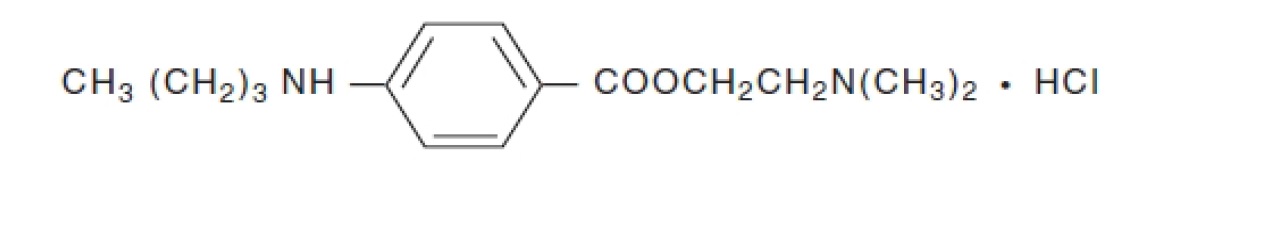TETRACAINE HYDROCHLORIDE- tetracaine hydrochloride solution/ drops
Paragon BioTeck, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% safely and effectively. See full prescribing information for Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%.
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%, for topical ophthalmic use Initial U.S. Approval: 1965 INDICATIONS AND USAGETetracaine Hydrochloride Ophthalmic Solution USP, 0.5%, is an ester local anesthetic indicated for procedures requiring a rapid and short-acting topical ophthalmic anesthetic. ( 1) DOSAGE AND ADMINISTRATIONOne drop topically in the eye(s) as needed. ( 2) DOSAGE FORMS AND STRENGTHSSterile, preserved, ophthalmic solution containing 0.5% tetracaine hydrochloride. ( 3) CONTRAINDICATIONSTetracaine Hydrochloride Ophthalmic Solution, 0.5% should not be used in patients with a history of hypersensitivity to any component of this preparation. ( 4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSOcular adverse events: transient stinging, burning, conjunctival redness, eye irritation, eye pain, ocular discomfort. ( 6) To report SUSPECTED ADVERSE REACTIONS, contactBausch & Lomb, a Division of Valeant Pharmaceuticals North America LLC,. at 1-800-321-4576or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2019 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% is indicated for procedures requiring
a rapid and short-acting topical ophthalmic anesthetic.
3 DOSAGE FORMS AND STRENGTHS
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% is a clear, colorless, ophthalmic
solution containing 0.5% w/v tetracaine hydrochloride equivalent to tetracaine 0.44% w/v.
4 CONTRAINDICATIONS
Tetracaine Hydrochloride Ophthalmic Solution, USP, 0.5% should not be used in patients with a
history of hypersensitivity to any component of this preparation.
5 WARNINGS AND PRECAUTIONS
5.1 Corneal Injury with Intracameral Use
Not for injection or intraocular use. Do not use intracamerally because use of Tetracaine
Hydrochloride Ophthalmic Solution USP, 0.5% may lead to damage of the corneal endothelial
cells.
6 ADVERSE REACTIONS
The following serious ocular adverse reactions are described elsewhere in the labeling:
- Corneal Injury with Intracameral Use [See Warnings and Precautions ( 5.1)]
- Corneal Toxicity [See Warnings and Precautions ( 5.2)]
- Corneal Injury due to Insensitivity [See Warnings and Precautions ( 5.3)]
The following adverse reactions have been identified following use of Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliable estimate their frequency or establish a causal relationship to drug exposure.
Ocular Adverse Reactions
Transient stinging, burning, and conjunctival redness, eye irritation, eye pain, ocular discomfort.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies with Tetracaine Hydrochloride Ophthalmic
Solution USP, 0.5% in pregnant women. Animal developmental and reproductive toxicity studies
with tetracaine hydrochloride have not been reported in the published literature.
8.2 Lactation
Risk Summary
There are no data to assess whether Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% is
excreted in human milk or to assess its effects on milk production/excretion. The developmental
and health benefits of breastfeeding should be considered along with the mother’s clinical need
for Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% and any potential adverse effects
on the breastfed child from Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%.
8.3 Females and Males of Reproductive Potential
No human data on the effect of Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% on
fertility are available.
8.4 Pediatric Use
Safety of Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% in the pediatric population
has been demonstrated in clinical trials. Efficacy of Tetracaine Hydrochloride Ophthalmic
Solution USP, 0.5% for use in pediatric patients has been extrapolated from adequate and well
controlled clinical trials in the adult population.
10 OVERDOSAGE
Prolonged use of a topical ocular anesthetic including Tetracaine Hydrochloride Ophthalmic
Solution USP, 0.5% may produce permanent corneal opacification and ulceration with
accompanying visual loss.
11 DESCRIPTION
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% is a sterile, clear, colorless, topical
local anesthetic for ophthalmic use containing tetracaine hydrochloride as the active
pharmaceutical ingredient.
Tetracaine hydrochloride is chemically designated as benzoic acid, 4-(butylamino)-,
2-(dimethylamino) ethyl ester, monohydrochloride. Its chemical formula is C15H24N2O2 •
HCl and it is represented by the chemical structure:
Tetracaine hydrochloride is a fine, white, crystalline, odorless powder with a molecular weight of
300.82
Active ingredient: tetracaine hydrochloride 0.5% w/v (equivalent to 0.44% w/v tetracaine)
Preservative: chlorobutanol 0.4%
Inactive ingredients: boric acid, potassium chloride, edetate disodium, water for injection USP.
Sodium hydroxide and/or hydrochloric acid may be added to adjust pH (3.7 – 6.0)
12 CLINICAL PHARMACOLOGY
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies to assess the genotoxicity of tetracaine hydrochloride have not been reported in the
published literature. Long-term animal studies have not been conducted to evaluate the
carcinogenic potential of tetracaine hydrochloride. Animal studies to assess the effects of
tetracaine hydrochloride on fertility have not been reported in the published literature.
14 CLINICAL STUDIES
Topical administration of Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% results in
localized temporary anesthesia. The maximum effect is achieved within 10–20 seconds after
instillation, with efficacy lasting 10–20 minutes. Duration of effect can be extended with
repeated dosing. [See Warnings and Precautions ( 5.2) and Overdosage ( 10)].
16 HOW SUPPLIED/STORAGE AND HANDLING
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5% is supplied as a sterile, aqueous,
topical ophthalmic solution with a fill volume of 15 mL in a 15 mL low-density polyethylene
plastic dropper bottle with a low-density polyethylene dropper tip and white polypropylene cap.
NDC 42702-170-15
After opening, this product can be used until the expiration date stamped on the bottle.
Storage: Store at 15°C to 25°C (59°F to 77°F). Protect from light. Do not use if solution
contains crystals, cloudy, or discolored.
17 PATIENT COUNSELING INFORMATION
Eye Care Precaution
Do not touch the dropper tip to any surface as this may contaminate the solution.
Advise patients that, due to the effect of the anesthetic, their eyes will be insensitive for up to 20
minutes and that care should be taken to avoid accidental injuries.
Manufactured for:
Paragon BioTeck, Inc.
4640 SW Macadam Ave, Ste 80
Portland, OR 97239
Manufactured and Distributed by:
Bausch & Lomb
8500 Hidden River Pkwy
Tampa, FL 33637
Revised: March 2019
| TETRACAINE HYDROCHLORIDE
tetracaine hydrochloride solution/ drops |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Paragon BioTeck, Inc. (078279037) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bausch & Lomb Incorporated | 079587625 | manufacture(42702-170) | |