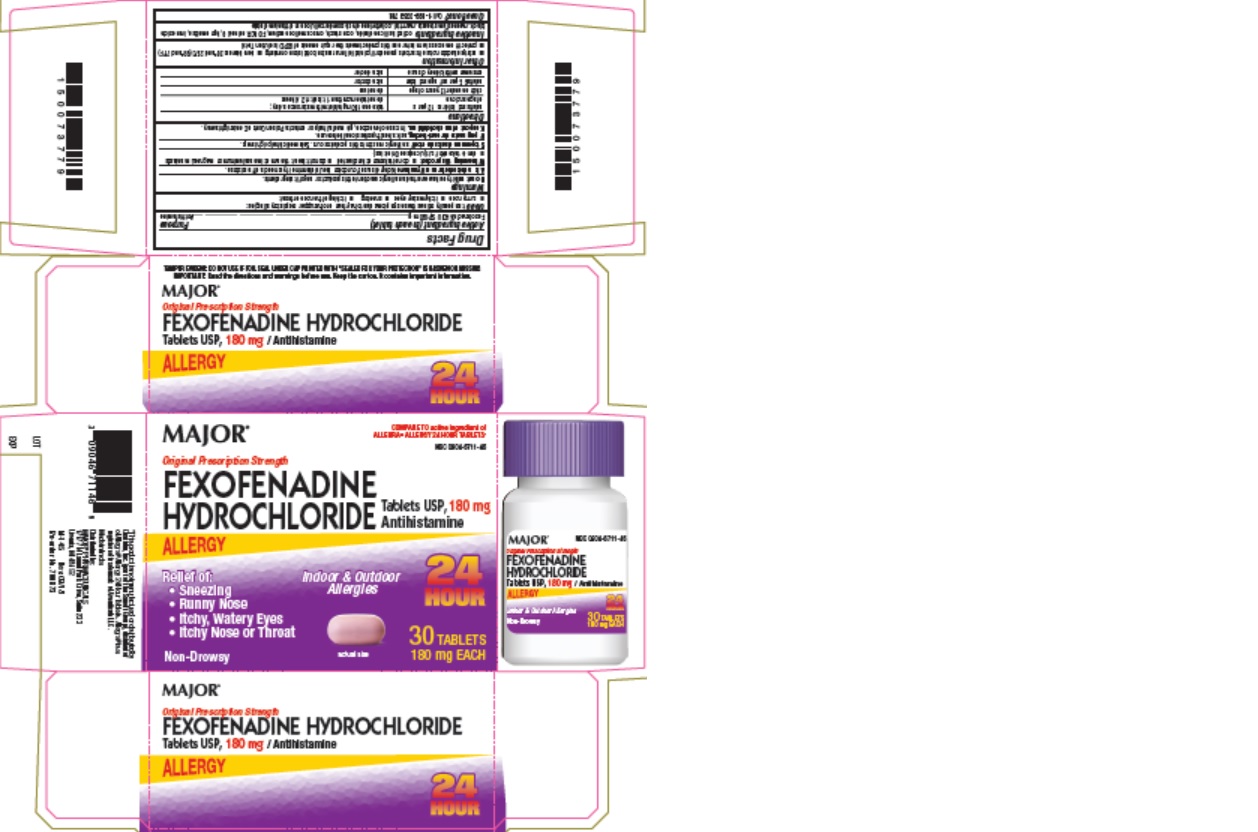
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Directions
| adults and children 12 years of age and over | take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Other information
- safety sealed: do not use if carton is opened or if printed foil inner seal on bottle is torn or missing
- store between 20° and 25°C (68° and 77°F)
- protect from excessive moisture
- this product meets the requirements of USP Dissolution Test 4
Inactive ingredients
colloidal silicon dioxide, corn starch, croscarmellose sodium, FD&C Red no. 40, hypromellose, iron oxide black, magnesium stearate, mannitol, polyethylene glycol, powder cellulose and titantium dioxide
Package Label - 30 Count Carton
MAJOR
COMPARE TO active ingredient of
ALLEGRA® ALLERGY 24 HOUR TABLETS*
NDC 0904-6711-46
Original Prescription Strength
FEXOFENADINE
HYDROCHLORIDE Tablets USP, 180 mg
Antihistamine
ALLERGY
Relief of:
- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Nose or Throat
Non-Drowsy
Indoor & Outdoor
Allergies
24
HOUR
30 TABLETS
180 mg EACH