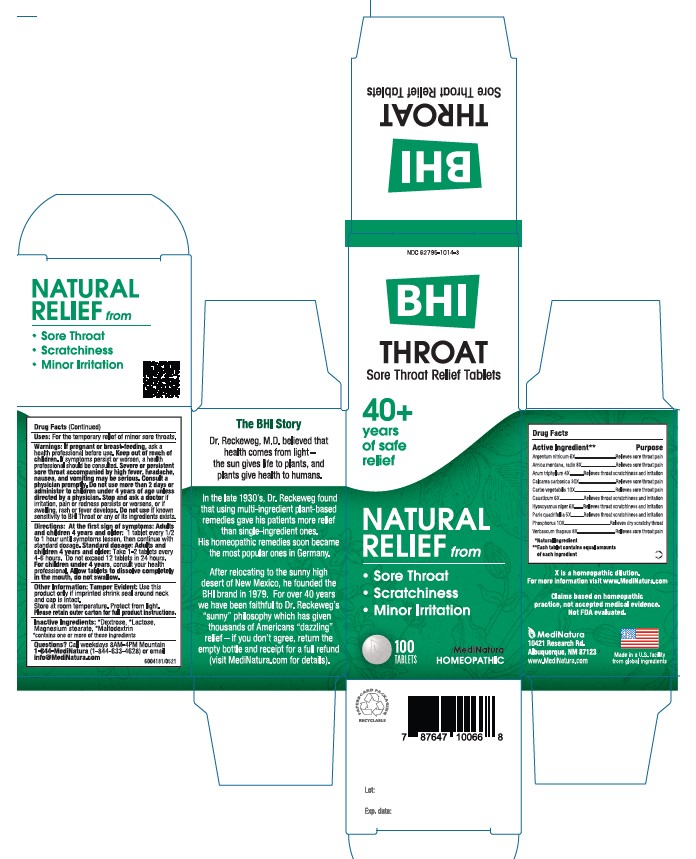
ACTIVE INGREDIENTS
Each tablet contains: Argentum nitricum 6X, *Arnica montana, radix 8X, *Arum triphyllum 4X, Calcarea carbonica 30X, *Carbo vegetabilis 10X, Causticum 6X, *Hyoscyamus niger 6X, *Paris quadrifolia 5X, *Phosphorus 10X, *Verbascum thapsus 8X 30 mg each.
*Natural Ingredients
INACTIVE INGREDIENT
Inactive Ingredients: **Dextrose, ** Lactose, Magnesium Stearate, **Maltodextrin
**contains one or more of these ingredients.
DIRECTIONS
At first sign of symptoms: Adults and children 4 years and older: 1 tablet every 1/2 to 1 hour until symptoms lessen, then continue with standard dosage.
Standard dosage: Adults and children 4 years and older: Take 1-2 tablets every 4 to 6 hours. Do not exceed 12 tablets in 24 hours.
For children under 4, consult your health professional.
Allow tablets to dissolve completely in the mouth, do not swallow
.
WARNINGS
If pregnant or breast-feeding , ask a health professional before use. Keep out of reach of children. If symptoms persist or worsen, a health professional should be consulted. Severe or persistent sore throat accompanied by high fever, headache, nausea, and vomiting may be serious. Consult physician promptly. Do not use more than 2 days or administer to children under 4 years of age unless directed by a physician. Stop and ask a doctor if irritation, pain or redness persists or worsens, or if swelling, rash or fever develops.Do not use if known sensitivity to Throat or any of its ingredients exists.