FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Calcium Gluconate Injection is indicated for pediatric and adult patients for the treatment of acute symptomatic hypocalcemia.
Limitations of Use
The safety of Calcium Gluconate Injection for long term use has not been established.
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
- Calcium Gluconate Injection contains 100 mg of calcium gluconate per mL which contains 9.3 mg (i.e., 0.465 mEq) of elemental calcium.
- Dilute Calcium Gluconate Injection prior to use in 5% dextrose or normal saline and assess for potential drug or IV fluid incompatibilities [see Dosage and Administration (2.5)].
- Inspect Calcium Gluconate Injection visually prior to administration. The solution should appear clear and colorless to slightly yellow. Do not administer if there is particulate matter or discoloration.
- Use the diluted solution immediately after preparation.
- Administer Calcium Gluconate Injection intravenously via a secure intravenous line to avoid calcinosis cutis and tissue necrosis [see Warnings and Precautions (5.3)].
- Administer Calcium Gluconate Injection by bolus administration or continuous infusion:
For bolus intravenous administration:
- Dilute the dose [see Dosage and Administration (2.2)] of Calcium Gluconate Injection in 5% dextrose or normal saline to a concentration of 10-50 mg/mL prior to administration. Administer the dose slowly and DO NOT exceed an infusion rate of 200 mg/minute in adults or 100 mg/minute in pediatric patients, including neonates. Monitor patients, vitals and electrocardiograph (ECG) during administration [see Warnings and Precautions (5.4)].
For continuous intravenous infusion:
- Dilute Calcium Gluconate Injection in 5% dextrose or normal saline to a concentration of 5.8-10 mg/mL prior to administration. Administer at the rate recommended in Table 1 [see Dosage and Administration (2.2)] and monitor patients, vitals, calcium and ECG during the infusion [see Warnings and Precautions (5.4)].
- Calcium Gluconate Injection is supplied in single-dose vials.
2.2 Recommended Dosage
Individualize the dose of Calcium Gluconate Injection within the recommended range depending on the severity of symptoms of hypocalcemia, the serum calcium level, and the acuity of onset of hypocalcemia.
Table 1 provides dosing recommendations for Calcium Gluconate Injection in mg of calcium gluconate for neonates, pediatric and adult patients.
Table 1. Dosing Recommendations in mg of Calcium Gluconate for Neonate, Pediatric, and Adult Patients
|
Patient Population |
Initial Dose | Subsequent Doses (if needed)
|
|
| Bolus
| Continuous Infusion
|
||
| Neonate
(≤ 1 month) | 100 – 200 mg/kg | 100 - 200 mg/kg every 6 hours | Initiate at 17-33 mg/kg/hour |
| Pediatric
(> 1 month to < 17 years) | 29 - 60 mg/kg | 29 - 60 mg/kg every 6 hours | Initiate at 8-13 mg/kg/hour |
| Adult
| 1000 - 2000 mg | 1000 - 2000 mg every 6 hours | Initiate at 5.4 - 21.5 mg/kg/hour |
For bolus administration, DO NOT exceed an infusion rate of:
|
|||
2.3 Serum Calcium Monitoring
Measure serum calcium every 4 to 6 hours during intermittent infusions with Calcium Gluconate Injection and measure serum calcium every 1 to 4 hours during continuous infusion.
2.4 Dosage in Renal Impairment
For patients with renal impairment, initiate Calcium Gluconate Injection at the lowest dose of the recommended dose ranges for all age groups and monitor serum calcium levels every 4 hours.
2.5 Drug Incompatibilities
- Do not mix Calcium Gluconate Injection with ceftriaxone. Concurrent use of intravenous ceftriaxone and Calcium Gluconate Injection can lead to the formation of ceftriaxone-calcium precipitates. Concomitant use of ceftriaxone and intravenous calcium-containing products is contraindicated in neonates (28 days of age or younger) [see Contraindications (4)]. In patients older than 28 days of age, ceftriaxone and calcium-containing products may be administered sequentially, provided the infusion lines are thoroughly flushed between infusions with a compatible fluid. Ceftriaxone must not be administered simultaneously with intravenous calcium-containing solutions via a Y-site in any age group [see Warnings and Precautions (5.2), Drug Interactions (7.3)].
- Do not mix Calcium Gluconate Injection with fluids containing bicarbonate or phosphate. Calcium Gluconate Injection is not physically compatible with fluids containing phosphate or bicarbonate. Precipitation may result if mixed.
- Do not mix Calcium Gluconate Injection with minocycline injection. Calcium complexes minocycline rendering it inactive.
3 DOSAGE FORMS AND STRENGTHS
Calcium Gluconate Injection, USP is a clear, colorless to slightly yellow, solution free from visible particles available in the following:
- Single dose vial: 1,000 mg per 10 mL (100 mg per mL)
Each mL of Calcium Gluconate Injection, USP contains 9.3 mg (0.465 mEq) of elemental calcium.
4 CONTRAINDICATIONS
Calcium Gluconate Injection is contraindicated in:
- Hypercalcemia
- Neonates (28 days of age or younger) receiving ceftriaxone [see Warnings and Precautions (5.2)]
5 WARNINGS AND PRECAUTIONS
5.1 Arrhythmias with Concomitant Cardiac Glycoside Use
Cardiac arrhythmias may occur if calcium and cardiac glycosides are administered together. Hypercalcemia increases the risk of digoxin toxicity. Administration of Calcium Gluconate Injection should be avoided in patients receiving cardiac glycosides. If concomitant therapy is necessary, Calcium Gluconate Injection should be given slowly in small amounts and with close ECG monitoring [see Drug Inte ractions (7.1)].
5.2 End-Organ Damage due to Intravascular Ceftriaxone-Calcium Precipitates
Concomitant use of ceftriaxone and Calcium Gluconate Injection is contraindicated in neonates (28 days of age or younger) due to cases of fatal outcomes in neonates in which a crystalline material was observed in the lungs and kidneys at autopsy after ceftriaxone and calcium were administrated simultaneously through the same intravenous line. Concomitant administration can lead to the formation of ceftriaxone-calcium precipitates that may act as emboli, resulting in vascular spasm or infarction [see Contraindications (4)].
In patients older than 28 days of age, ceftriaxone and Calcium Gluconate Injection may be administered sequentially, provided the infusion lines are thoroughly flushed between infusions with a compatible fluid. Do not administer Ceftriaxone simultaneously with Calcium Gluconate Injection via a Y-site in any age group.
5.3 Tissue Necrosis and Calcinosis
Intravenous administration of Calcium Gluconate Injection and local trauma may result in calcinosis cutis due to transient increase in local calcium concentration. Calcinosis cutis can occur with or without extravasation of Calcium Gluconate Injection, is characterized by abnormal dermal deposits of calcium salts, and clinically manifests as papules, plaques, or nodules that may be associated with erythema, swelling, or induration. Tissue necrosis, ulceration, and secondary infection are the most serious complications.
If extravasation occurs or clinical manifestations of calcinosis cutis are noted, immediately discontinue intravenous administration at that site and treat as needed.
5.4 Hypotension, Bradycardia, and Cardiac Arrhythmias with Rapid Administration
Rapid injection of Calcium Gluconate Injection may cause vasodilation, decreased blood pressure, bradycardia, cardiac arrhythmias, syncope and cardiac arrest. To avoid adverse reactions that may follow rapid intravenous administration, Calcium Gluconate Injection should be diluted with 5% dextrose or normal saline and infused slowly. If rapid intravenous bolus of Calcium Gluconate Injection is required, the rate of intravenous administration should not exceed 200 mg/minute in adults and 100 mg/minute in pediatric patients and ECG monitoring during administration is recommended [see Dosage and Administration (2.1)].
5.5 Aluminum Toxicity
Calcium Gluconate Injection contains aluminum, up to 400 mcg per liter, that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration if kidney function is impaired. Premature neonates are particularly at risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum. Research indicates that patients with impaired kidney function, including premature neonates, who receive parenteral levels of aluminum at greater than 4 mcg/kg/day to 5 mcg/kg/day accumulate aluminum levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
6 ADVERSE REACTIONS
The following serious adverse reactions are also described elsewhere in the labeling:
- Arrhythmias with Concomitant Cardiac Glycoside Use [see Warnings and Precautions (5.1)]
- End-Organ Damage due to Intravascular Ceftriaxone-Calcium Precipitates [see Warnings and Precautions (5.2)]
- Tissue Necrosis and Calcinosis [see Warnings and Precautions (5.3)]
- Hypotension, Bradycardia, and Cardiac Arrhythmias [see Warnings and Precautions (5.4)]
- Aluminum toxicity [see Warnings and Precautions (5.5)]
The following adverse reactions associated with the use of calcium gluconate were identified in the literature. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
Cardiovascular: Vasodilation, decreased blood pressure, bradycardia, cardiac arrhythmia, syncope, cardiac arrest
Administration site reactions: Local soft tissue inflammation, local necrosis, calcinosis cutis and calcification due to extravasation
7 DRUG INTERACTIONS
7.1 Cardiac Glycosides
Hypercalcemia increases the risk of digoxin toxicity, while digoxin may be therapeutically ineffective in the presence of hypocalcemia. Synergistic arrhythmias may occur if calcium and cardiac glycosides are administered together. Avoid administration of Calcium Gluconate Injection in patients receiving cardiac glycosides; if considered necessary, administer Calcium Gluconate Injection slowly in small amounts and monitor ECG closely during administration.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk summary
Limited available data with Calcium Gluconate Injection use in pregnant women are insufficient to inform a drug associated risk of adverse developmental outcomes. There are risks to the mother and the fetus associated with hypocalcemia in pregnancy [see Clinical Considerations].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Disease-associated maternal risk
Maternal hypocalcemia can result in an increased rate of spontaneous abortion, premature and dysfunctional labor, and possibly preeclampsia.
Fetal/Neonatal adverse reactions
Infants born to mothers with hypocalcemia can have associated fetal and neonatal hyperparathyroidism, which in turn can cause fetal and neonatal skeletal demineralization, subperiosteal bone resorption, osteitis fibrosa cystica and neonatal seizures. Infants born to mothers with hypocalcemia should be carefully monitored for signs of hypocalcemia or hypercalcemia, including neuromuscular irritability, apnea, cyanosis and cardiac rhythm disorders.
8.2 Lactation
Risk summary
Calcium is present in human milk as a natural component of human milk. It is not known whether intravenous administration of Calcium Gluconate Injection can alter calcium concentration in human milk. There are no data on the effects of Calcium Gluconate Injection on the breastfed infant, or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Calcium Gluconate Injection and any potential adverse effects on the breastfed child from Calcium Gluconate Injection or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of Calcium Gluconate Injection have been established in pediatric patients for the treatment of acute, symptomatic hypocalcemia.
Pediatric approval for Calcium Gluconate Injection, including doses, is not based on adequate and well-controlled clinical studies. Safety and dosing recommendations in pediatric patients are based on published literature and clinical experience [see Dosage and Administration (2.2)].
Concomitant use of ceftriaxone and Calcium Gluconate Injection is contraindicated in neonates (28 days of age or younger) due to reports of fatal outcomes associated with the presence of lung and kidney ceftriaxone-calcium precipitates. In patients older than 28 days of age, ceftriaxone and Calcium Gluconate Injection may be administered sequentially, provided the infusion lines are thoroughly flushed between infusions with a compatible fluid [see Contraindications (4) and Warnings and Precautions (5.2)]. This product contains up to 400 mcg/L aluminum which may be toxic, particularly for premature neonates due to immature renal function. Parenteral administration of aluminum greater than 4 to 5 mcg/kg/day is associated with central nervous system and bone toxicity [see Warnings and Precautions (5.5)].
8.5 Geriatric Use
In general dose selection for an elderly patient should start at the lowest dose of the recommended dose range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
For patients with renal impairment, initiate Calcium Gluconate Injection at the lowest dose of the recommended dose ranges across all age groups. Monitor serum calcium levels every 4 hours [see Dosage and Administration (2.4)].
10 OVERDOSAGE
Overdosage of Calcium Gluconate Injection may result in hypercalcemia. Symptoms of hypercalcemia typically develop when the total serum calcium concentration is ≥12 mg/dL. Neurologic symptoms include depression, weakness, fatigue, and confusion at lower levels, with patients experiencing hallucinations, disorientation, hypotonicity, seizures, and coma. Effects on the kidney include diminished ability to concentrate urine and diuresis.
If overdose of Calcium Gluconate Injection occurs immediately discontinue administration and provide supportive treatments to restore intravascular volume as well as promote calcium excretion in the urine if necessary.
11 DESCRIPTION
Calcium Gluconate Injection, USP is a clear, colorless to slightly yellow, sterile, preservative-free, nonpyrogenic, supersaturated solution of calcium gluconate, a form of calcium, for intravenous use.
Calcium Gluconate is calcium D-gluconate (1:2) monohydrate. The structural formula is:
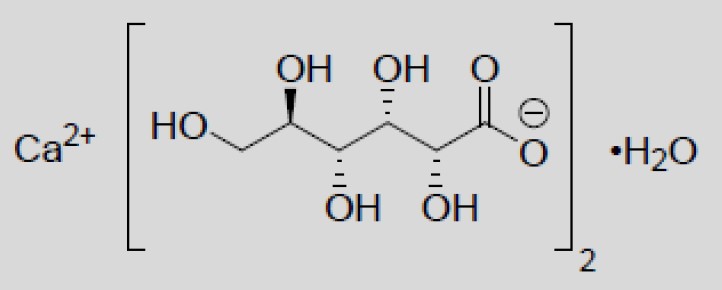
Molecular formula: C12H22CaO14• H2O
Molecular weight: 448.39
Solubility in water: 3.5 g/100 mL at 25°C
Calcium Gluconate Injection, USP is available as 1,000 mg per 10 mL (100 mg per mL) in a single-dose vial.
Each mL of Calcium Gluconate Injection, USP contains 100 mg of calcium gluconate (equivalent to 94 mg of calcium gluconate and 4.5 mg of calcium saccharate tetrahydrate), hydrochloric acid and/or sodium hydroxide for pH adjustment (6.0 to 8.2) and water for injection, q.s. It contains no antimicrobial agent.
Each mL of Calcium Gluconate Injection, USP contains 9.3 mg (0.465 mEq) of elemental calcium.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Intravenous administration of calcium gluconate increases serum ionized calcium level. Calcium gluconate dissociates into ionized calcium in plasma. Ionized calcium and gluconate are normal constituents of body fluids.
12.3 Pharmacokinetics
Absorption
Calcium Gluconate Injection is 100% bioavailable following intravenous injection.
Metabolism
Calcium itself does not undergo direct metabolism. The release of ionized calcium from intravenous administration of calcium gluconate is direct and does not seem to be affected by the first pass through the liver.
Distribution
Calcium in the body is distributed mainly in skeleton (99%). Only 1% of the total body calcium is distributed within the extracellular fluids and soft tissues. About 50% of total serum calcium is in the ionized form and represents the biologically active part. 8% to 10% serum calcium is bound to organic and inorganic acid and approximately 40% is protein-bound (primarily to albumin).
Elimination
Studies have shown a relationship between urinary calcium excretion and the intravenous administration of calcium gluconate, with a significant increase in urinary calcium excretion observed after the intravenous administration of calcium gluconate.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been conducted to evaluate the carcinogenic potential of Calcium Gluconate Injection. Calcium gluconate was not mutagenic with or without metabolic activation in the Ames test with Salmonella typhimurium (strains TA-1535, TA-1537, and TA-1538) or Saccharomyces cerevisiae (Strain D4). Fertility studies in animals have not been conducted with calcium gluconate administered by the intravenous route.
16 HOW SUPPLIED/STORAGE AND HANDLING
Calcium Gluconate Injection, USP is a clear, colorless to slightly yellow solution free from visible particles supplied as follows:
| NDC | Strength/Vial Size (mL) |
| 75834-171-01 | 1,000 mg calcium gluconate per 10 mL (100 mg per mL), in a 10 mL plastic, single-dose vial. |
| 75834-171-19 | 1,000 mg calcium gluconate per 10 mL (100 mg per mL), in a 10 mL plastic, single-dose vial, packaged in a carton of 25. |
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Do not freeze.
Preservative Free. Discard any unused portion in the single-dose vial immediately.
The diluted solution must be used immediately.
NOTE: Supersaturated solutions are prone to precipitation. The precipitate, if present, may be dissolved by warming the vial to 60° to 80°C, with occasional agitation, until the solution becomes clear. Shake vigorously. Allow to cool to room temperature before dispensing. Use injection only if clear immediately prior to use.
17 PATIENT COUNSELING INFORMATION
- Advise the patient that the risks associated with infusion including local tissue inflammation, local necrosis and calcinosis. [see Warnings and Precautions (5.3)].
Manufactured for:
Nivagen Pharmaceuticals, Inc.
Sacramento, CA 95827 USA
Toll free number: 1-877-977-0687
Rev. 10/2022

