FEXOFENADINE HYDROCHLORIDE- fexofenadine hydrochloride tablet, film coated
PD-Rx Pharmaceuticals, Inc.
----------
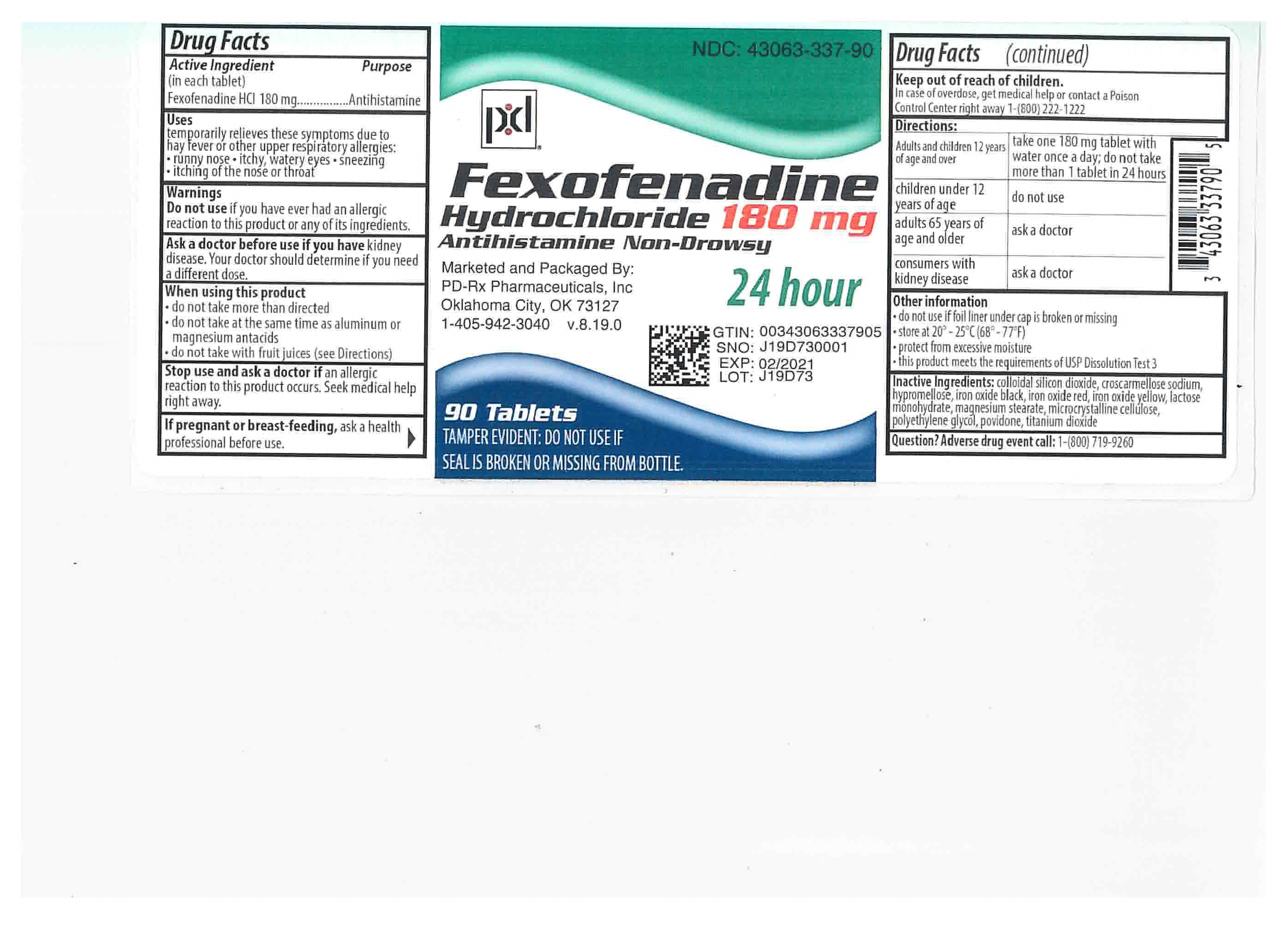
Perrigo Fexofenadine Hydrochloride Tablets, 180 mg Drug Facts
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Directions
|
adults and children 12 years of age and over |
take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours |
|
children under 12 years of age |
do not use |
|
adults 65 years of age and older |
ask a doctor |
|
consumers with kidney disease |
ask a doctor |
Other information
- do not use if foil liner under cap is broken or missing
- store between 20°-25°C (68°-77°F)
- protect from excessive moisture
- this product meets the requirements of USP Dissolution Test 3
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, hypromellose, iron oxide black, iron oxide red, iron oxide yellow, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, titanium dioxide
HOW SUPPLIED
Fexofenadine Hydrochloride 180mg , USP are available as the following:
180 mg peach, round, film-coated tablets, debossed "93" on obverse and "7253" on reverse. They are available as follows:
Bottles of 10: NDC 43063-337-10
Bottles of 30: NDC 43063-337-30
Bottles of 90: NDC 43063-337-90
| FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tablet, film coated |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - PD-Rx Pharmaceuticals, Inc. (156893695) |
| Registrant - PD-Rx Pharmaceuticals, Inc. (156893695) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| PD-Rx Pharmaceuticals, Inc. | 156893695 | repack(43063-337) | |