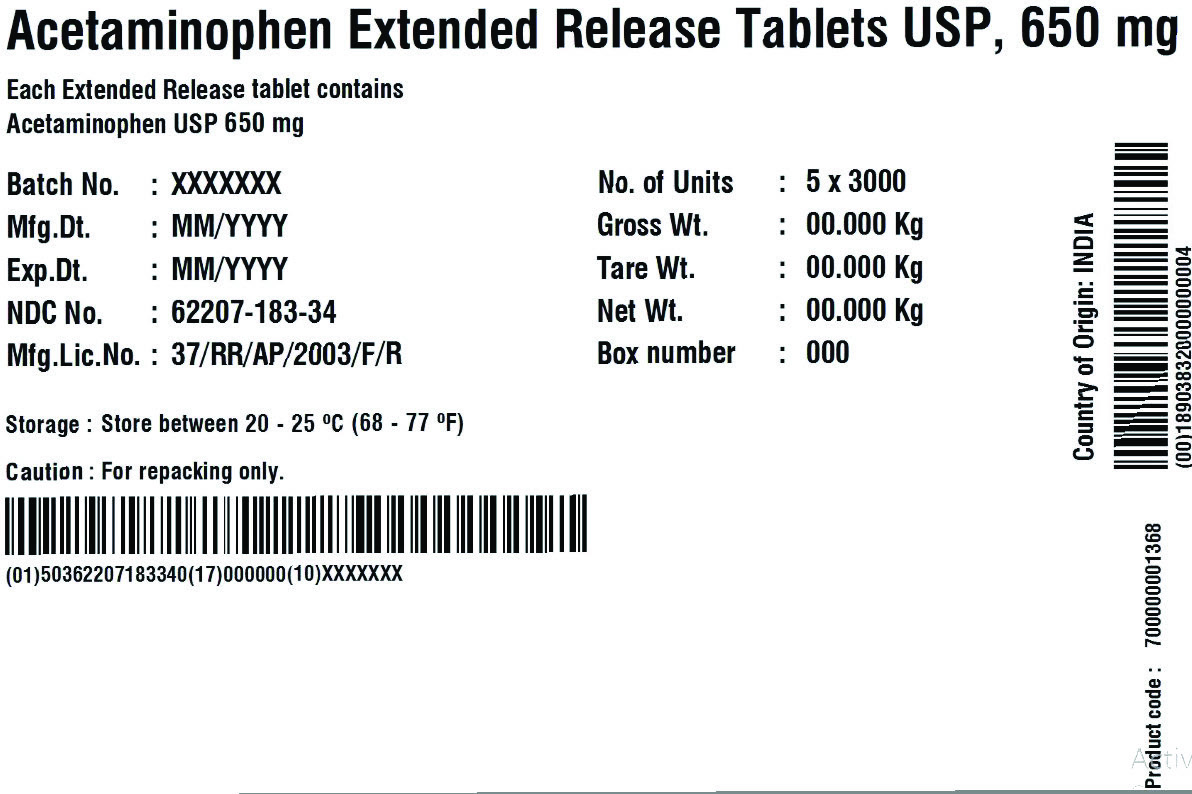
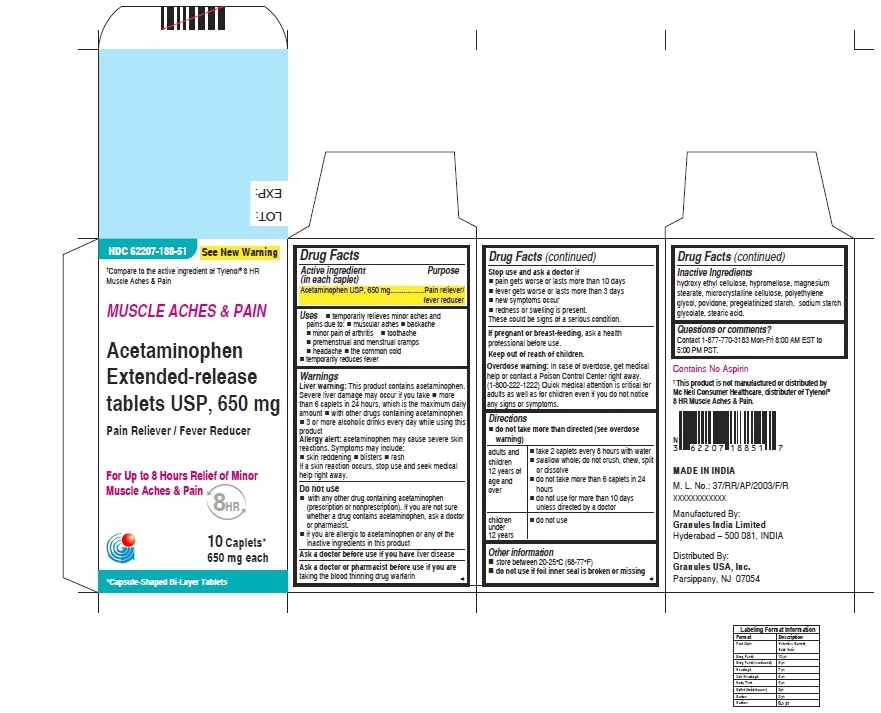
USES
For
Arthritis Pain label
• temporarily relieves minor aches and pains due to:
• minor pain of arthritis
• muscular aches
• backache
• premenstrual and menstrual cramps
• the common cold
• headache
• toothache
• temporarily reduces fever
For
Muscle Aches & Pain label
• temporarily relieves minor aches and pains due to:
• muscular aches
• backache
• minor pain of arthritis
• toothache
• premenstrual and menstrual cramps
• headache
• the common cold
• temporarily reduces fever
WARNINGS
Liver warning: This product contains acetaminophen. Severe Liver damage may occur if you take
• more than 6 caplets in 24 hours, which is the maximum daily amount
• with other drugs containing acetaminophen
• 3 or more alcoholic drinks everyday while using this product
Allergy alert: acetaminophen may cause severe skin reactions
Symptoms may include:
• skin reddening
• blisters
• rash
If a skin reaction occurs, stop use and seek medical help right away
Do not use
• with any other drugs containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
• if you are allergic to acetaminophen or any of the inactive ingredients in this product.
Stop use and ask doctor if
• pain gets worse or lasts more than 10 days
• fever gets worse or lasts more than 3 days
• new symptoms occur
• redness or swelling is present
These could be signs of a serious condition.
Keep out of reach of children
Overdose warning: In case of overdose, get medical help or contact a poison control center right away.(1-800 222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms
DIRECTIONS
For Arthritis Pain Label
•
do not take more than directed (see overdose warning)
| adults | • take 2 caplets every 8 hours with water
• swallow whole; do not crush, chew, split or dissolve • do not take more than 6 caplets in 24 hours • do not use for more than 10 days unless directed by a doctor |
| under 18 years
of age | • ask a doctor |
For Muscle Ache and Pain label
• do not take more than directed (see overdose warning)
| adults and children
12 years of age and over | • take 2 caplets every 8 hours with water
• swallow whole; do not crush, chew, split or dissolve • do not take more than 6 caplets in 24 hours • do not use for more than 10 days unless directed by a doctor |
| children under
12 years | • do not use |
OTHER INFORMATION
• store between 20-25°C (68-77°F)
• do not use if foil inner seal is broken or missing.