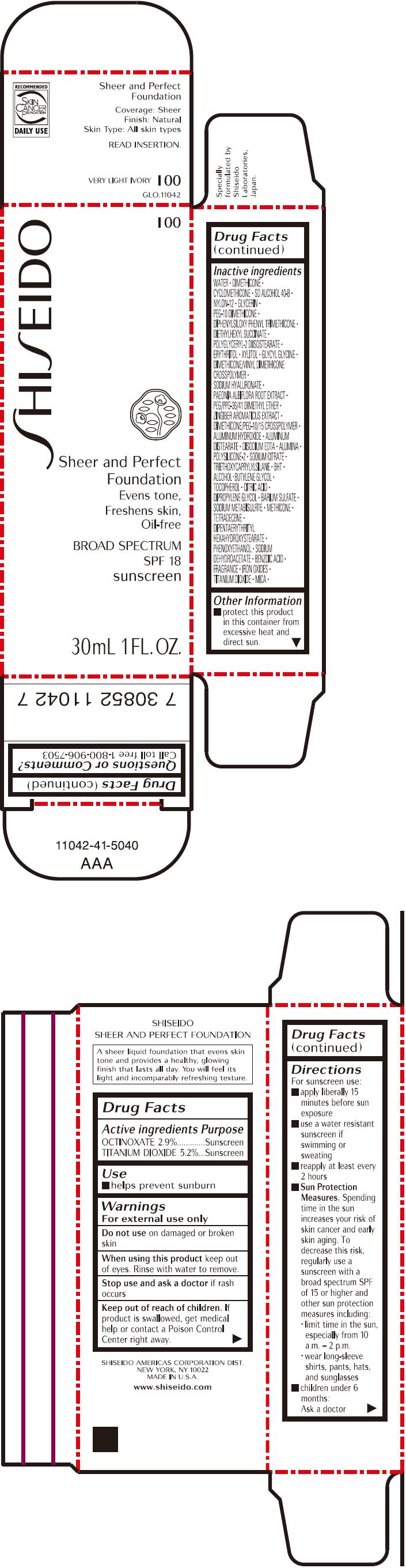
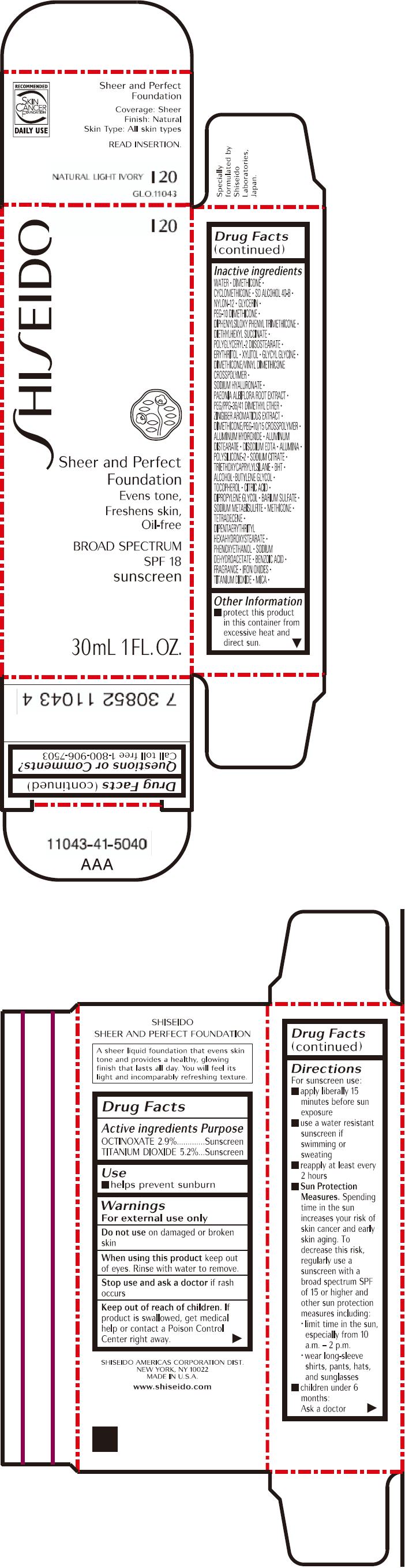
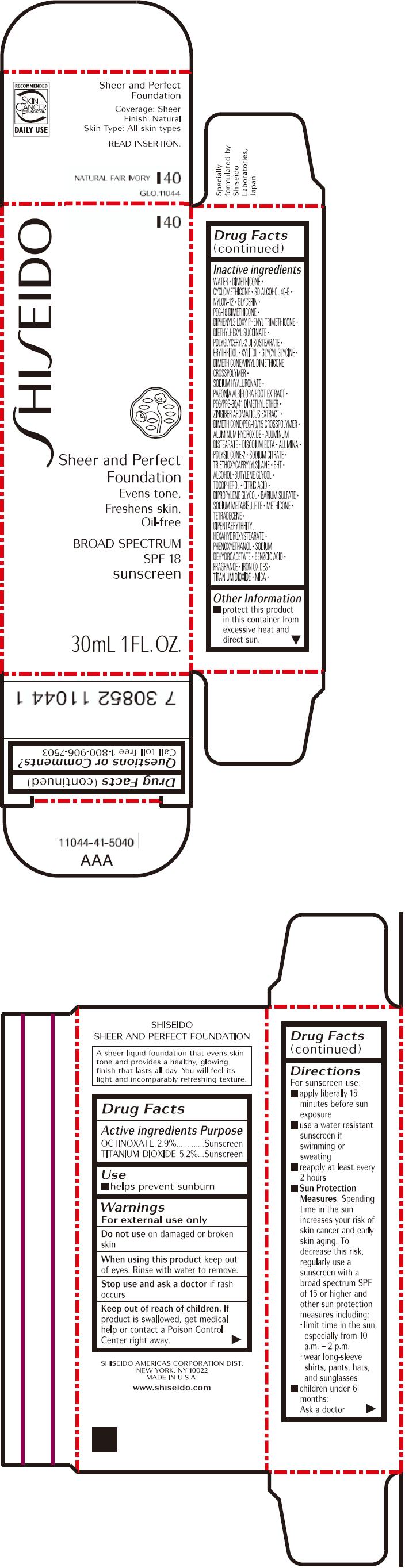
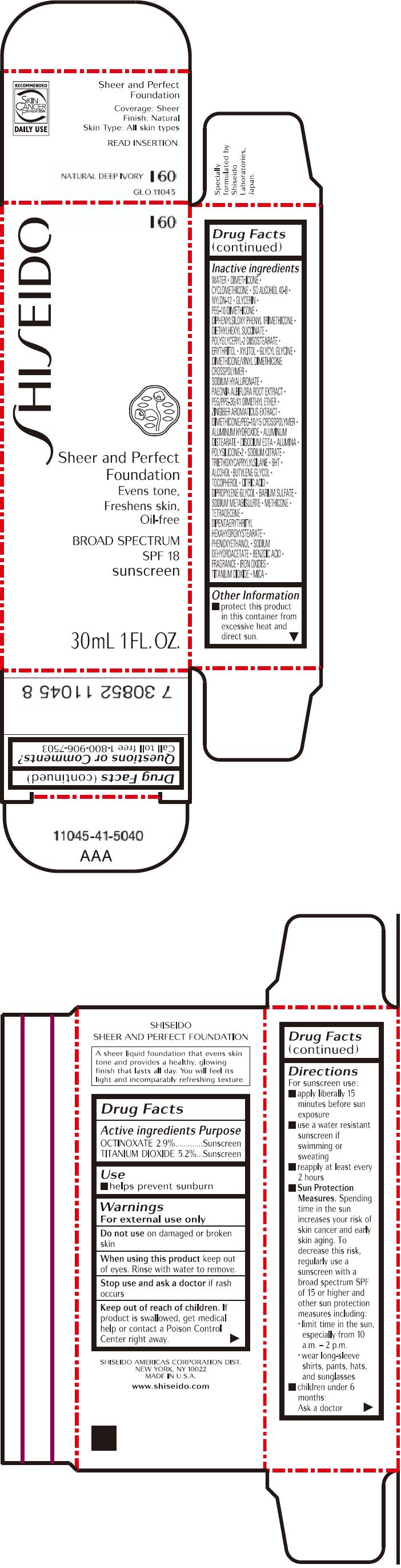
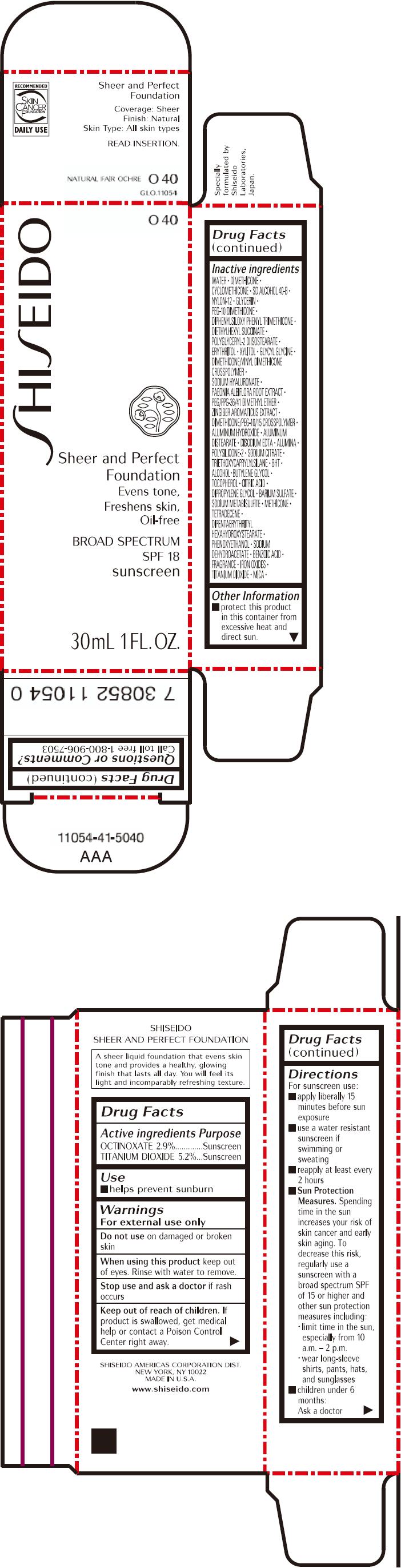
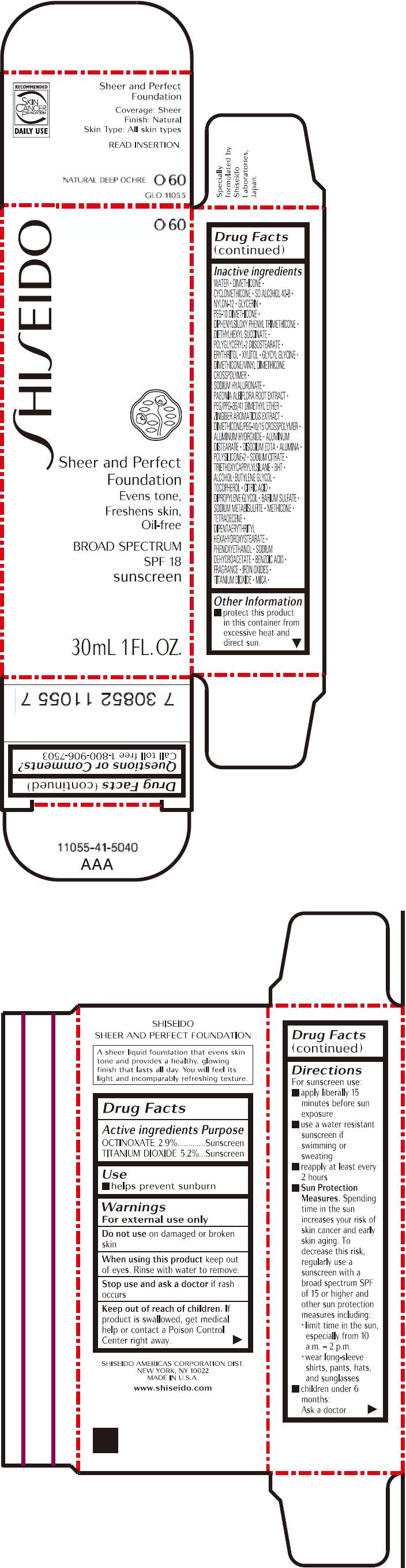
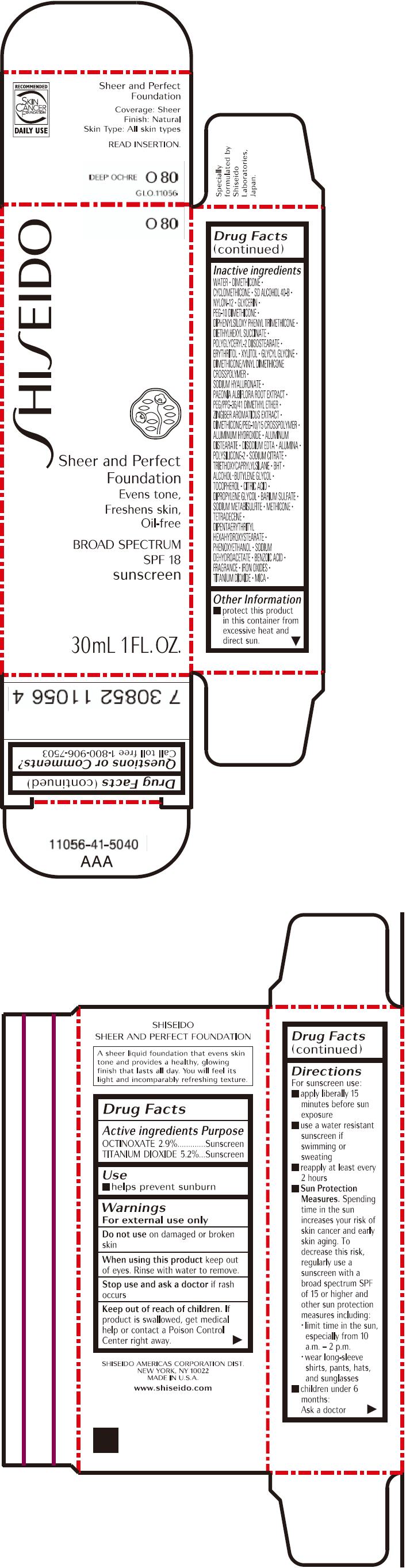
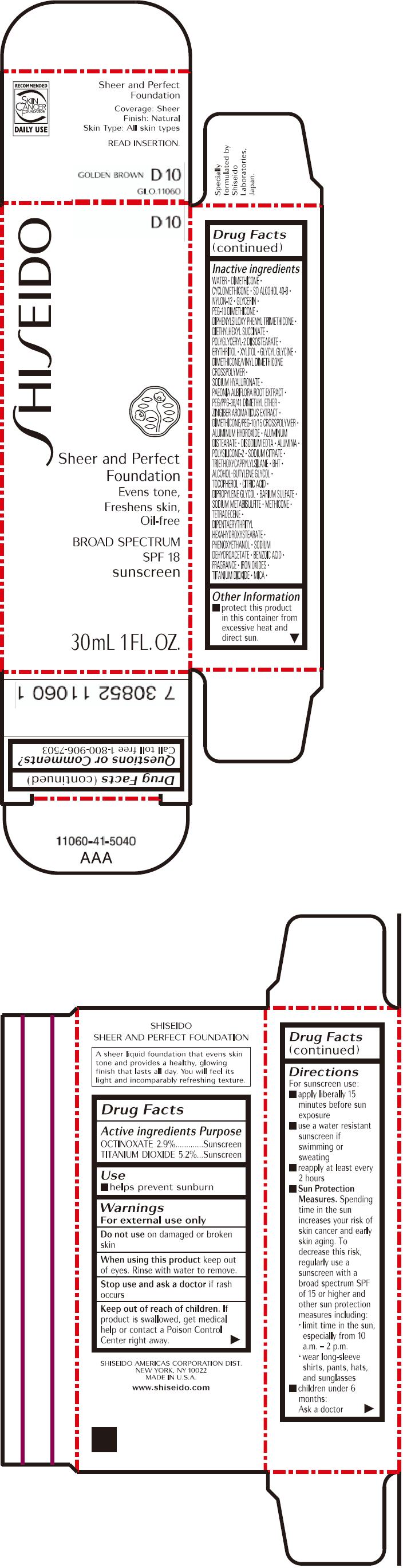
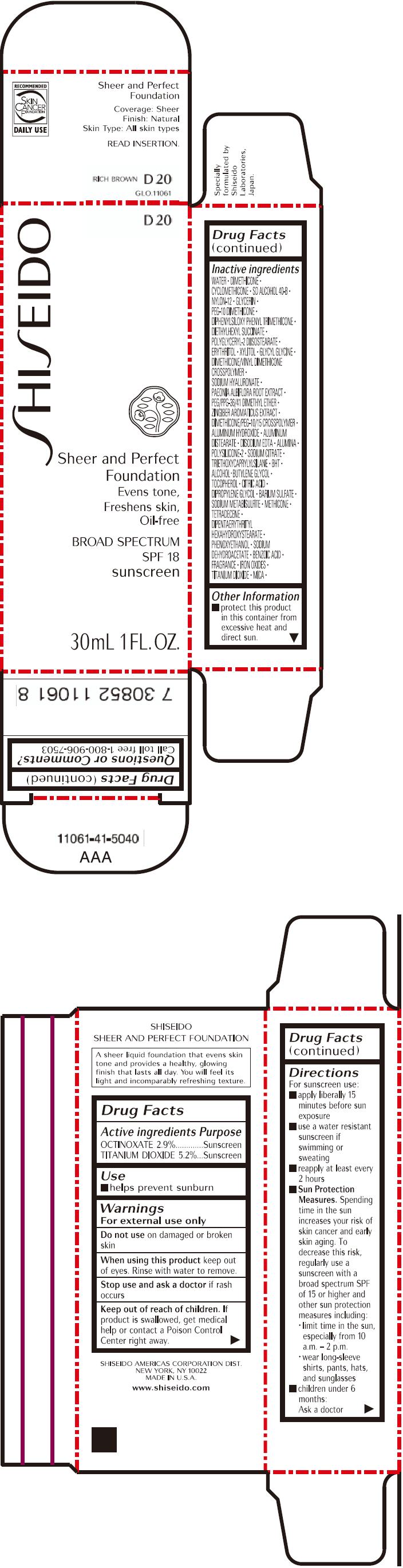
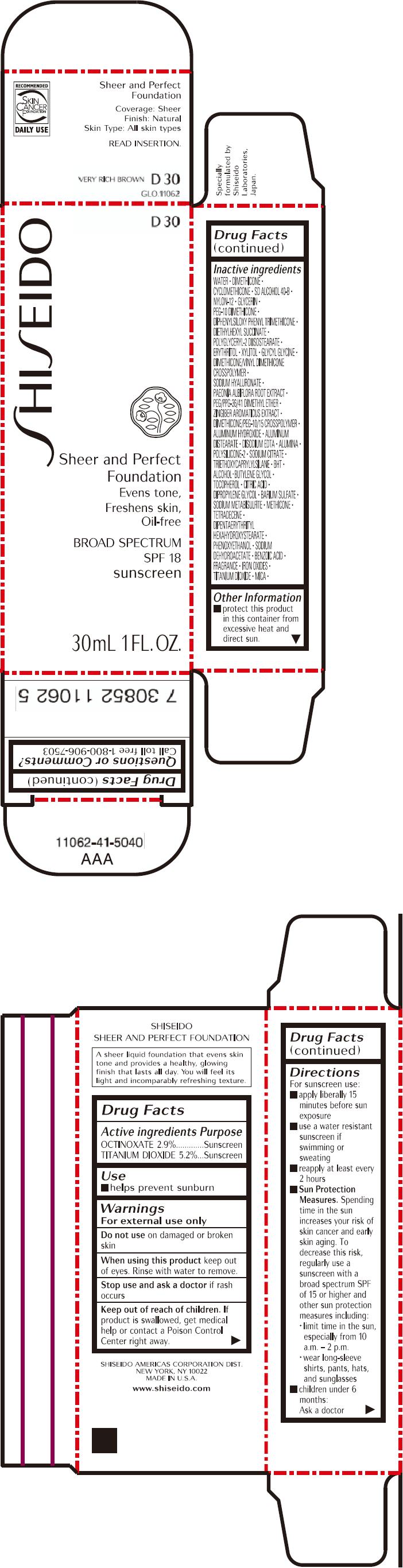
| ACTIVE INGREDIENTS: | Purpose |
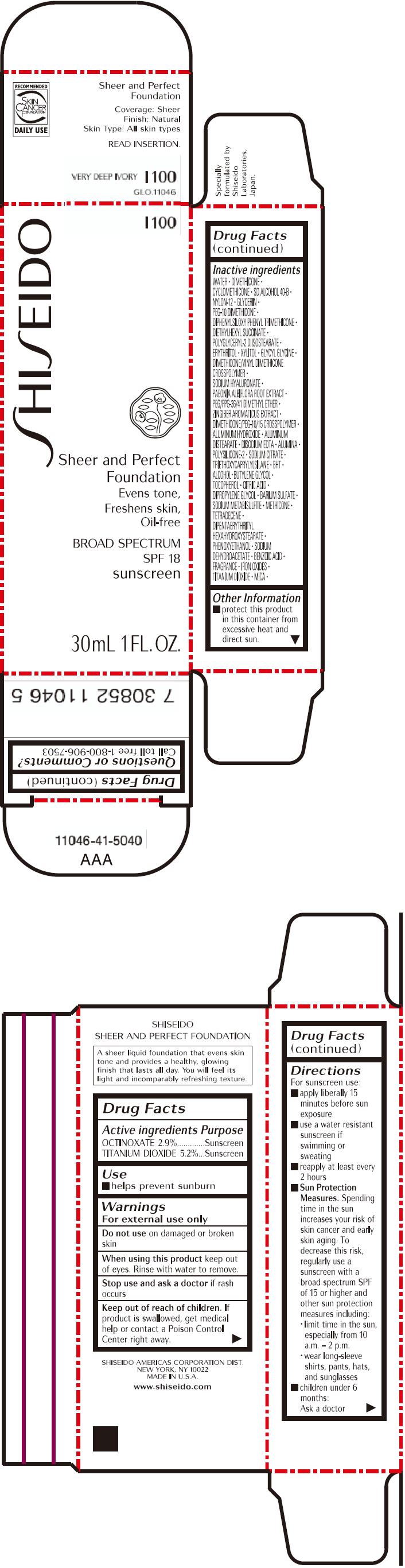
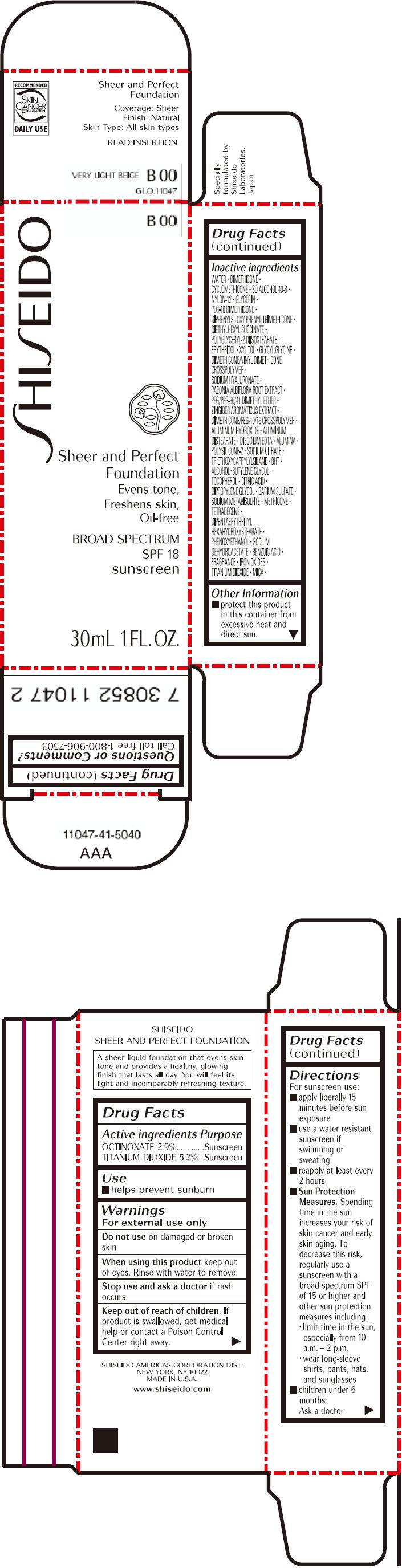
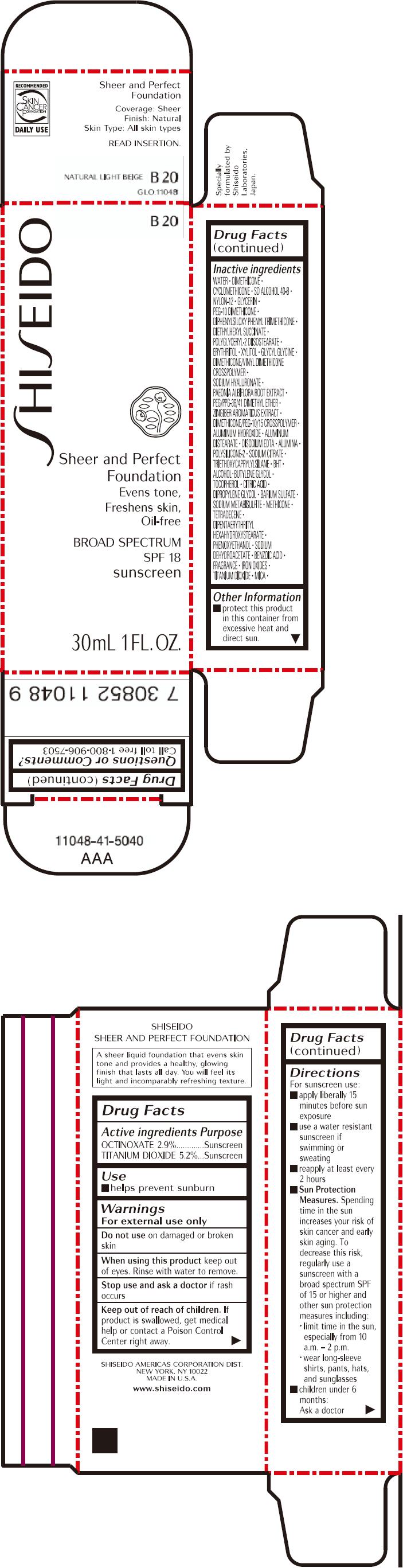
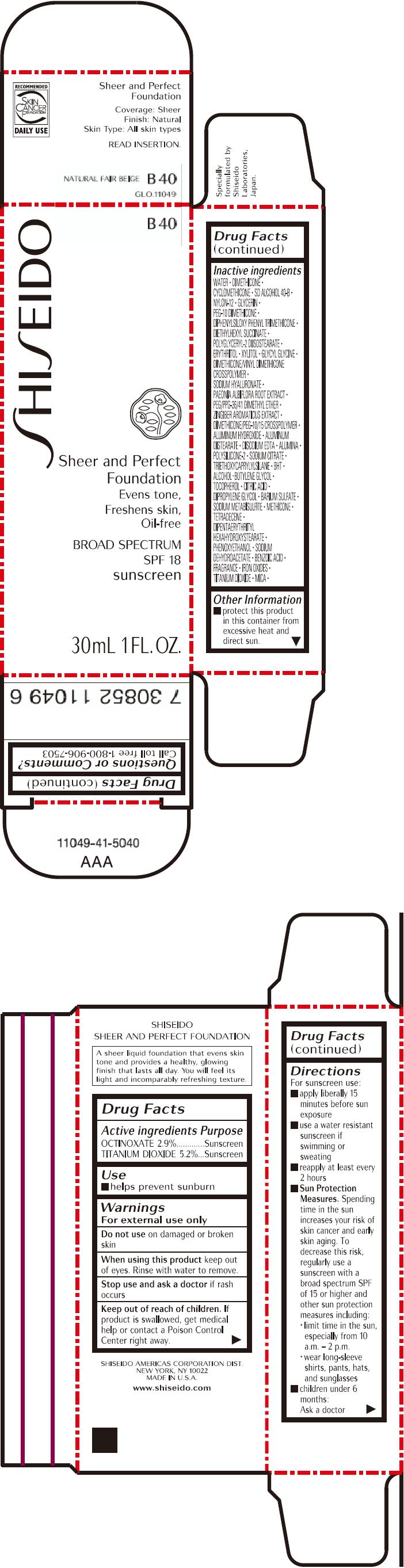
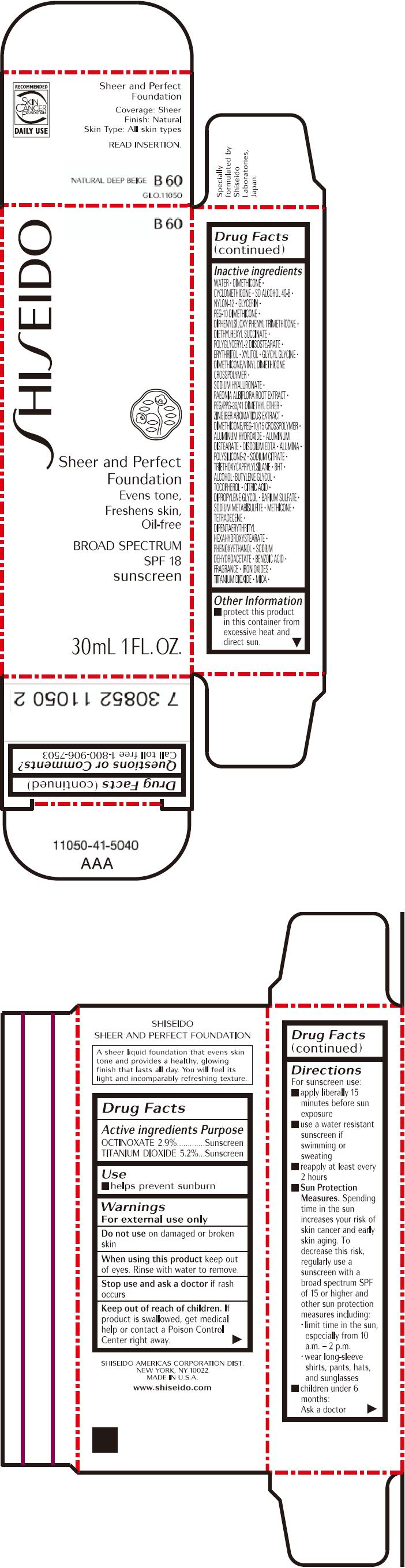
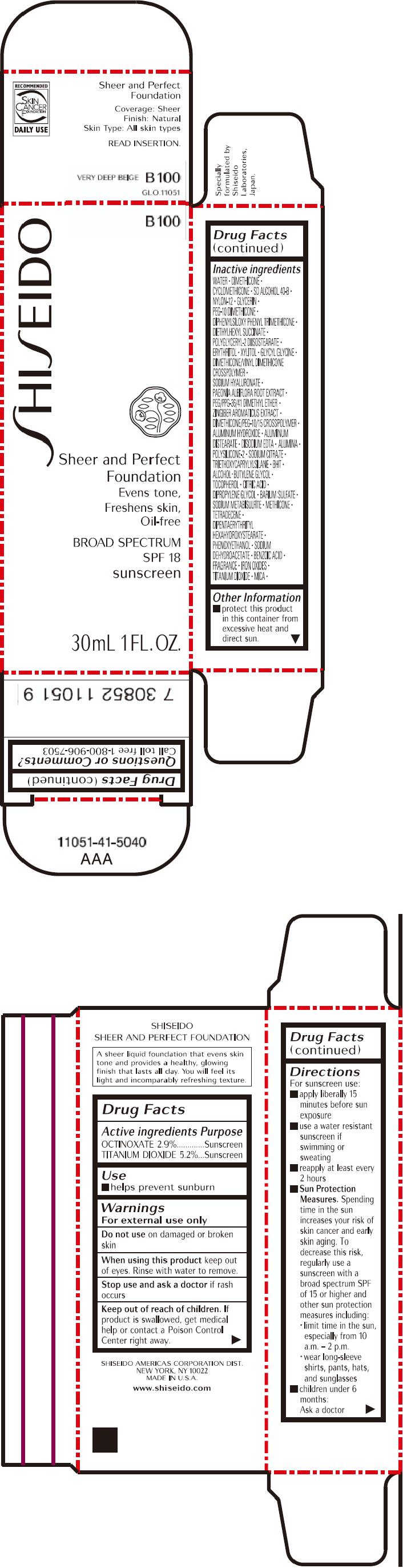
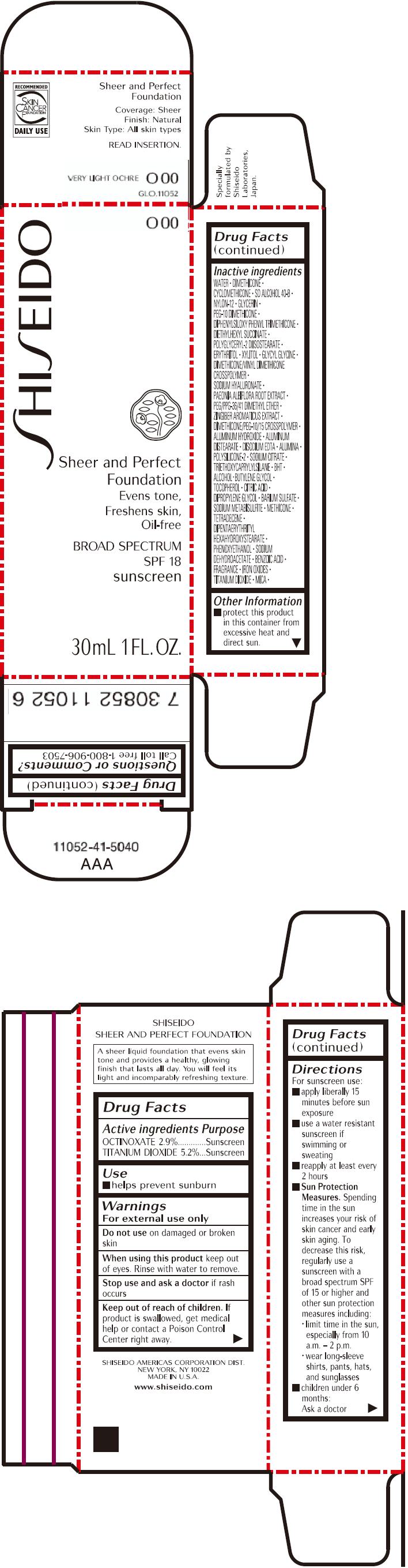
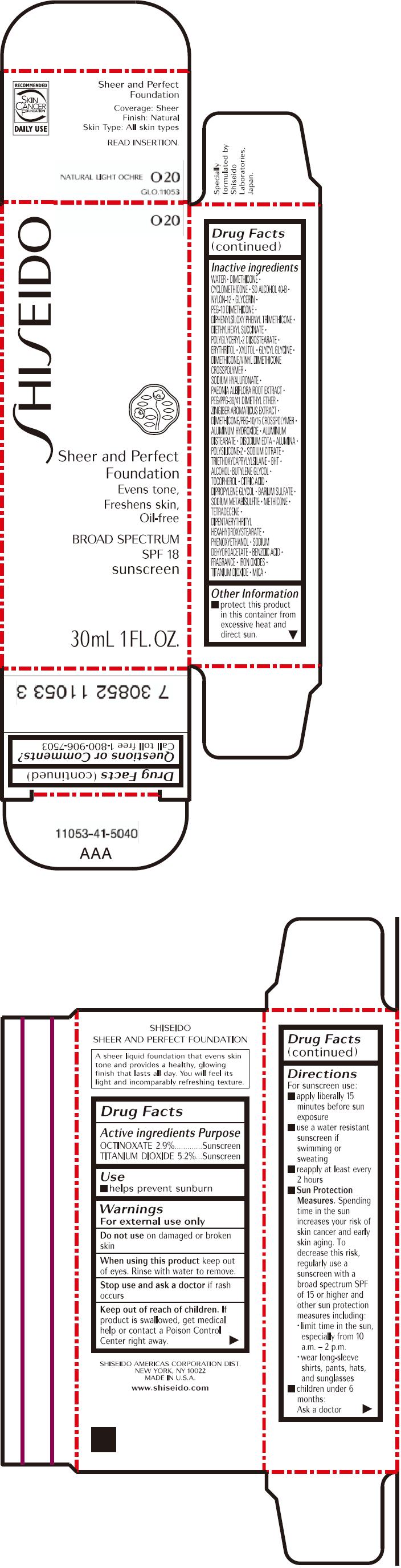
| OCTINOXATE 2.9% | Sunscreen |
| TITANIUM DIOXIDE 5.2% | Sunscreen |
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
Inactive Ingredients
WATER, DIMETHICONE, CYCLOMETHICONE, SD ALCOHOL 40-B, NYLON-12, GLYCERIN, PEG-10 DIMETHICONE, DIPHENYLSILOXY PHENYL TRIMETHICONE, DIETHYLHEXYL SUCCINATE, POLYGLYCERYL-2 DIISOSTEARATE, ERYTHRITOL, XYLITOL, GLYCYL GLYCINE, DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER, SODIUM HYALURONATE, PAEONIA ALBIFLORA ROOT EXTRACT, PEG/PPG-36/41 DIMETHYL ETHER, ZINGIBER AROMATICUS EXTRACT, DIMETHICONE/PEG-10/15 CROSSPOLYMER, ALUMINUM HYDROXIDE, ALUMINUM DISTEARATE, DISODIUM EDTA, ALUMINA, POLYSILICONE-2, SODIUM CITRATE, TRIETHOXYCAPRYLYLSILANE, BHT, ALCOHOL, BUTYLENE GLYCOL, TOCOPHEROL, CITRIC ACID, DIPROPYLENE GLYCOL, BARIUM SULFATE, SODIUM METABISULFITE, METHICONE, TETRADECENE, DIPENTAERYTHRITYL HEXAHYDROXYSTEARATE, PHENOXYETHANOL, SODIUM DEHYDROACETATE, BENZOIC ACID, FRAGRANCE, IRON OXIDES, TITANIUM DIOXIDE, MICA,
Other information
- protect this product in this container from excessive heat and direct sun.
Questions or comments?
Call toll free 1-800-906-7503
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - I00
SHISEIDO
I00
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - I20
SHISEIDO
I20
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - I40
SHISEIDO
I40
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - I60
SHISEIDO
I60
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - I100
SHISEIDO
I100
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - B00
SHISEIDO
B00
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - B20
SHISEIDO
B20
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - B40
SHISEIDO
B40
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - B60
SHISEIDO
B60
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - B100
SHISEIDO
B100
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - O00
SHISEIDO
O00
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - O20
SHISEIDO
O20
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - O40
SHISEIDO
O40
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - O60
SHISEIDO
O60
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - O80
SHISEIDO
O80
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - D10
SHISEIDO
D10
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - D20
SHISEIDO
D20
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.
PRINCIPAL DISPLAY PANEL - 30mL Bottle Carton - D30
SHISEIDO
D30
Sheer and Perfect
Foundation
Evens tone,
Freshens skin,
Oil-free
BROAD SPECTRUM
SPF 18
sunscreen
30mL 1 FL. OZ.