DIPROLENE AF- betamethasone dipropionate cream
Merck Sharp & Dohme Corp.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use DIPROLENE AF Cream safely and effectively. See full prescribing information for DIPROLENE AF Cream.
DIPROLENE® AF (augmented betamethasone dipropionate) cream, for topical use Initial U.S. Approval: 1983 INDICATIONS AND USAGEDIPROLENE AF Cream is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 13 years of age and older. (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 6/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
DIPROLENE® AF Cream is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 13 years of age or older.
2 DOSAGE AND ADMINISTRATION
Apply a thin film of DIPROLENE AF Cream to the affected skin areas once or twice daily.
Therapy should be discontinued when control is achieved. DIPROLENE AF Cream is a high-potency corticosteroid. Treatment with DIPROLENE AF Cream should not exceed 50 g per week because of the potential for the drug to suppress the hypothalamic-pituitary-adrenal (HPA) axis [see Warnings and Precautions (5.1)].
DIPROLENE AF Cream should not be used with occlusive dressings unless directed by a physician.
Avoid contact with eyes. Wash hands after each application.
Avoid use on the face, groin, or axillae, or if skin atrophy is present at the treatment site.
DIPROLENE AF Cream is for topical use only. It is not for oral, ophthalmic, or intravaginal use.
3 DOSAGE FORMS AND STRENGTHS
Cream, 0.05%. Each gram of DIPROLENE AF Cream, 0.05% contains 0.643 mg betamethasone dipropionate (equivalent to 0.5 mg betamethasone) in a white cream base.
4 CONTRAINDICATIONS
DIPROLENE AF Cream is contraindicated in patients who are hypersensitive to betamethasone dipropionate, to other corticosteroids, or to any ingredient in this preparation.
5 WARNINGS AND PRECAUTIONS
5.1 Effects on Endocrine System
DIPROLENE AF Cream can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency. This may occur during treatment or after withdrawal of treatment. Factors that predispose to HPA axis suppression include the use of high-potency steroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age. Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test.
DIPROLENE AF Cream 0.05% was applied once daily at 7 grams per day for 1 week to diseased skin, in adult subjects with psoriasis or atopic dermatitis, to study its effects on the HPA axis. The results suggested that the drug lowered adrenal corticosteroid secretion, although plasma cortisol levels did not go below the lower limit of the normal range.
In an open-label pediatric trial of 60 evaluable subjects (3 months to 12 years of age), 19 subjects showed evidence of HPA axis suppression. Four (4) subjects were tested 2 weeks after discontinuation of DIPROLENE AF Cream 0.05%, and 3 of the 4 (75%) had complete recovery of HPA axis function. The proportion of subjects with adrenal suppression in this trial was progressively greater, the younger the age group.
If HPA axis suppression is documented, gradually withdraw the drug, reduce the frequency of application, or substitute with a less potent corticosteroid. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids.
Cushing's syndrome and hyperglycemia may also occur with topical corticosteroids. These events are rare and generally occur after prolonged exposure to excessively large doses, especially of high-potency topical corticosteroids.
Pediatric patients may be more susceptible to systemic toxicity due to their larger skin surface to body mass ratios [see Use in Specific Populations (8.4)].
5.2 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including DIPROLENE AF Cream, may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported postmarketing with the use of topical corticosteroid products, including DIPROLENE AF Cream [see Adverse Reactions (6.2)].
Avoid contact of DIPROLENE AF Cream with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
5.3 Allergic Contact Dermatitis
Allergic contact dermatitis with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation. Such an observation should be corroborated with appropriate diagnostic patch testing. If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In controlled clinical trials, involving 242 adult subjects, the adverse reaction associated with the use of DIPROLENE AF Cream reported at a frequency of 0.4% was stinging. It occurred in 1 subject.
In a controlled clinical trial involving 67 pediatric subjects from 3 months to 12 years of age, the adverse reactions associated with the use of DIPROLENE AF Cream occurred in 7 of 67 (10%) subjects. Reported reactions included signs of skin atrophy (telangiectasia, bruising, shininess).
6.2 Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Postmarketing reports for local adverse reactions to topical corticosteroids may also include: burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, hypertrichosis, skin atrophy, striae, and miliaria.
Hypersensitivity reactions, consisting of predominantly skin signs and symptoms, e.g., contact dermatitis, pruritus, bullous dermatitis, and erythematous rash have been reported.
Ophthalmic adverse reactions of cataracts, glaucoma, increased intraocular pressure, and central serous chorioretinopathy have been reported with the use of topical corticosteroids, including topical betamethasone products.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on DIPROLENE AF Cream use in pregnant women to identify a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Observational studies suggest an increased risk of low birthweight infants with the use of greater than 300 grams of potent or very potent topical corticosteroid during a pregnancy. Advise pregnant women that DIPROLENE AF Cream may increase the risk of having a low birthweight infant and to use DIPROLENE AF Cream on the smallest area of skin and for the shortest duration possible.
In animal reproduction studies, increased malformations, including umbilical hernias, cephalocele, and cleft palate, were observed after intramuscular administration of betamethasone dipropionate to pregnant rabbits. The available data do not allow the calculation of relevant comparisons between the systemic exposure of betamethasone dipropionate in animal studies to the systemic exposure that would be expected in humans after topical use of DIPROLENE AF Cream (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data regarding the excretion of betamethasone dipropionate in breast milk, the effects on the breastfed infant, or the effects on milk production after topical application of DIPROLENE AF Cream to women who are breastfeeding.
It is possible that topical administration of large amounts of betamethasone dipropionate could result in sufficient systemic absorption to produce detectable quantities in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for DIPROLENE AF Cream and any potential adverse effects on the breastfed infant from DIPROLENE AF Cream or from the underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the breastfed infant via breast milk, use DIPROLENE AF Cream on the smallest area of skin and for the shortest duration possible while breastfeeding. Advise breastfeeding women not to apply DIPROLENE AF Cream directly to the nipple and areola to avoid direct infant exposure [see Use in Specific Populations (8.4)].
8.4 Pediatric Use
Use of DIPROLENE AF Cream in pediatric patients younger than 13 years of age is not recommended due to the potential for HPA axis suppression [see Warnings and Precautions (5.1)].
In an open-label HPA axis safety trial in subjects 3 months to 12 years of age with atopic dermatitis, DIPROLENE AF Cream 0.05% was applied twice daily for 2 to 3 weeks over a mean body surface area of 58% (range 35% to 95%). In 19 of 60 (32%) evaluable subjects, adrenal suppression was indicated by either a ≤5 mcg/dL pre-stimulation cortisol, or a cosyntropin post-stimulation cortisol ≤18 mcg/dL and/or an increase of <7 mcg/dL from the baseline cortisol. Out of the 19 subjects with HPA axis suppression, 4 subjects were tested 2 weeks after discontinuation of DIPROLENE AF Cream, and 3 of the 4 (75%) had complete recovery of HPA axis function. The proportion of subjects with adrenal suppression in this trial was progressively greater, the younger the age group.
Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of systemic toxicity when treated with topical drugs. They are, therefore, also at greater risk of HPA axis suppression and adrenal insufficiency upon the use of topical corticosteroids.
Rare systemic effects such as Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients, especially those with prolonged exposure to large doses of high potency topical corticosteroids.
Local adverse reactions including skin atrophy have also been reported with use of topical corticosteroids in pediatric patients.
Avoid use of DIPROLENE AF Cream in the treatment of diaper dermatitis.
8.5 Geriatric Use
Clinical trials of DIPROLENE AF Cream included 104 subjects who were 65 years of age and over and 8 subjects who were 75 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out.
11 DESCRIPTION
DIPROLENE AF (augmented betamethasone dipropionate) Cream 0.05% contains betamethasone dipropionate USP, a synthetic adrenocorticosteroid, for topical use in a cream base. Betamethasone, an analog of prednisolone, has a high degree of corticosteroid activity and a slight degree of mineralocorticoid activity. Betamethasone dipropionate is the 17,21-dipropionate ester of betamethasone.
Chemically, betamethasone dipropionate is 9-fluoro-11β,17,21-trihydroxy-16β-methylpregna-1,4-diene-3,20-dione 17,21-dipropionate, with the empirical formula C28H37FO7, a molecular weight of 504.6, and the following structural formula:
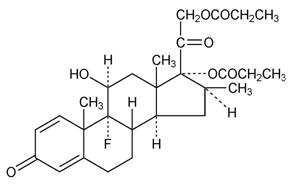
Betamethasone dipropionate is a white to creamy white, odorless crystalline powder, insoluble in water.
Each gram of DIPROLENE AF Cream 0.05% contains: 0.643 mg betamethasone dipropionate USP (equivalent to 0.5 mg betamethasone) in a white cream base of carbomer 940; ceteareth-30; chlorocresol; cyclomethicone; glyceryl oleate/propylene glycol; propylene glycol; purified water; sodium hydroxide; sorbitol solution; white petrolatum; and white wax.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action of DIPROLENE AF Cream in corticosteroid responsive dermatoses is unknown.
12.2 Pharmacodynamics
Vasoconstrictor Assay
Trials performed with DIPROLENE AF Cream, 0.05% indicate that it is in the high range of potency as demonstrated in vasoconstrictor trials in healthy subjects when compared with other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
12.3 Pharmacokinetics
No pharmacokinetics trials have been conducted with DIPROLENE AF Cream 0.05%. The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
Topical corticosteroids can be absorbed through normal intact skin. Inflammation and/or other disease processes in the skin may increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids [see Dosage and Administration (2)].
Once absorbed through the skin, topical corticosteroids enter pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees, are metabolized primarily in the liver, and excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of betamethasone dipropionate.
Betamethasone was negative in the bacterial mutagenicity assay (Salmonella typhimurium and Escherichia coli), and in the mammalian cell mutagenicity assay (CHO/HGPRT). It was positive in the in vitro human lymphocyte chromosome aberration assay, and equivocal in the in vivo mouse bone marrow micronucleus assay.
Studies in rabbits, mice, and rats using intramuscular doses up to 1, 33, and 2 mg/kg, respectively, resulted in dose-related increases in fetal resorptions in rabbits and mice.
14 CLINICAL STUDIES
The safety and efficacy of DIPROLENE AF Cream for the treatment of corticosteroid-responsive dermatoses have been established in two randomized and active controlled trials in subjects with chronic plaque psoriasis. A total of 81 subjects who received DIPROLENE AF Cream were included in these trials. These trials evaluated DIPROLENE AF Cream applied once or twice daily for 14 and 21 days, respectively, on bilateral paired psoriatic lesions. DIPROLENE AF Cream was shown to be effective in relieving the signs and symptoms of chronic plaque psoriasis.
16 HOW SUPPLIED/STORAGE AND HANDLING
DIPROLENE AF Cream 0.05% is a white cream supplied in 15-g (NDC 0085-0517-01) and 50-g (NDC 0085-0517-04) tubes.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Inform patients of the following:
- Discontinue therapy when control is achieved, unless directed otherwise by the physician.
- Use no more than 50 grams per week.
- Avoid contact with the eyes.
- Advise patients to report any visual symptoms to their healthcare providers.
- Avoid use of DIPROLENE AF Cream on the face, underarms, or groin areas unless directed by the physician.
- Do not occlude the treatment area with bandage or other covering, unless directed by the physician.
- Note that local reactions and skin atrophy are more likely to occur with occlusive use, prolonged use or use of higher potency corticosteroids.
- Advise a woman to use DIPROLENE AF Cream on the smallest area of skin and for the shortest duration possible while pregnant or breastfeeding. Advise breastfeeding women not to apply DIPROLENE AF Cream directly to the nipple and areola to avoid direct infant exposure.
Distributed by: Merck Sharp & Dohme Corp., a subsidiary of
MERCK & CO., INC., Whitehouse Station, NJ 08889, USA
For patent information: www.merck.com/product/patent/home.html
Copyright © 1987-2020 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.
All rights reserved.
uspi-mk1460-cr-2006r007
| Patient Information DIPROLENE® AF (DIH-pro-leen) (augmented betamethasone dipropionate) Cream |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 05/2019 |
| Important information: DIPROLENE AF Cream is for use on skin only. Do not use DIPROLENE AF Cream in your eyes, mouth, or vagina. | |
| What is DIPROLENE AF Cream? | |
DIPROLENE AF Cream is a prescription corticosteroid medicine used on the skin (topical) for the relief of redness, swelling, heat, pain (inflammation) and itching, caused by certain skin problems in people 13 years of age and older.
|
|
| Do not use DIPROLENE AF Cream if you are allergic to betamethasone dipropionate or any of the ingredients in DIPROLENE AF Cream. See the end of this leaflet for a complete list of ingredients in DIPROLENE AF Cream. | |
Before using DIPROLENE AF Cream, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
| Especially tell your healthcare provider if you take other corticosteroid medicines by mouth or injection or use other products on your skin or scalp that contain corticosteroids. | |
| Do not use other products containing a steroid medicine with DIPROLENE AF Cream without talking to your healthcare provider first. | |
How should I use DIPROLENE AF Cream?
|
|
| What are the possible side effects of DIPROLENE AF Cream? | |
DIPROLENE AF Cream may cause serious side effects, including:
|
|
| Your healthcare provider may do certain blood tests to check for side effects. | |
| The most common side effect of DIPROLENE AF Cream in adults includes stinging. | |
| The most common side effects of DIPROLENE AF Cream in children include thinning of the skin (atrophy), tiny red lines or blood vessels showing under the skin (telangiectasia), bruising and shininess. | |
| These are not all of the possible side effects of DIPROLENE AF Cream. | |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |
How should I store DIPROLENE AF Cream?
|
|
| General information about the safe and effective use of DIPROLENE AF Cream. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DIPROLENE AF Cream for a condition for which it was not prescribed. Do not give DIPROLENE AF Cream to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DIPROLENE AF Cream that is written for health professionals. | |
| What are the ingredients in DIPROLENE AF Cream? | |
| Active ingredient: betamethasone dipropionate | |
| Inactive ingredients: carbomer 940; ceteareth-30; chlorocresol; cyclomethicone; glyceryl oleate/propylene glycol; propylene glycol; purified water; sodium hydroxide; sorbitol solution; white petrolatum; and white wax. | |
| Distributed by: Merck Sharp & Dohme Corp., a subsidiary of MERCK & CO., INC., Whitehouse Station, NJ 08889, USA |
|
| For patent information: www.merck.com/product/patent/home.html Copyright © 1987-2019 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. All rights reserved. usppi-mk1460-cr-1905r000 |
|
| DIPROLENE AF
betamethasone dipropionate cream |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Merck Sharp & Dohme Corp. (001317601) |
