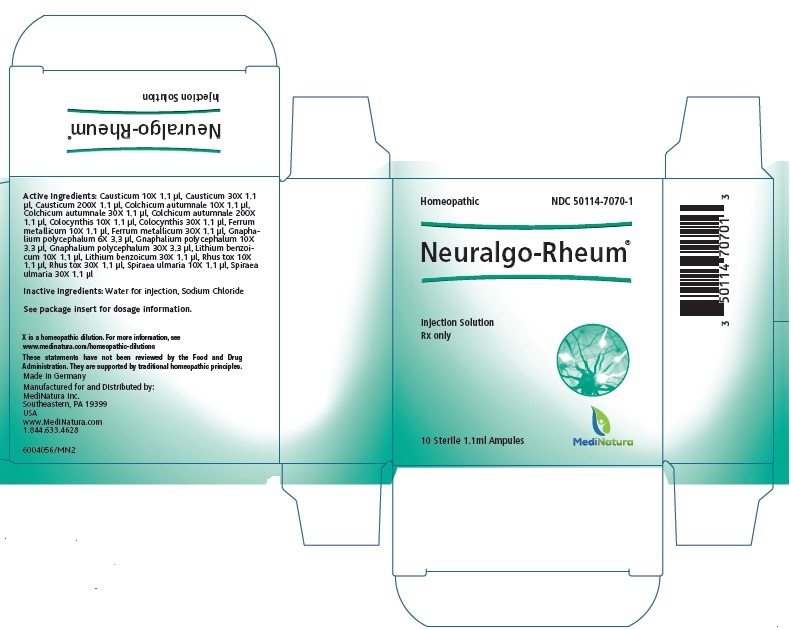
NEURALGO RHEUM- pseudognaphalium obtusifolium, causticum, colchicum autumnale bulb, citrullus colocynthis fruit pulp, iron, lithium benzoate, toxicodendron pubescens leaf and filipendula ulmaria root injection
MediNatura Inc
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
Neuralgo-Rheum 1.1ml Injection
DESCRIPTION
| Each 1.1 ml solution for injection ampule contains: | |||
| Active Ingredients: | |||
| Ingredient name | Potency | Quantity | Final dilution |
| Causticum | 10X | 1.1 µl | 12.99 |
| Causticum | 30X | 1.1 µl | 32.99 |
| Causticum | 200X | 1.1 µl | 203.00 |
| Colchicum autumnale | 10X | 1.1 µl | 12.99 |
| Colchicum autumnale | 30X | 1.1 µl | 32.99 |
| Colchicum autumnale | 200X | 1.1 µl | 203.00 |
| Colocynthis | 10X | 1.1 µl | 12.99 |
| Colocynthis | 30X | 1.1 µl | 32.99 |
| Ferrum metallicum | 10X | 1.1 µl | 12.99 |
| Ferrum metallicum | 30X | 1.1 µl | 32.99 |
| Gnaphalium polycephalum | 6X | 3.3 µl | 8.52 |
| Gnaphalium polycephalum | 10X | 3.3 µl | 12.52 |
| Gnaphalium polycephalum | 30X | 3.3 µl | 32.52 |
| Lithium benzoicum | 10X | 1.1 µl | 12.99 |
| Lithium benzoicum | 30X | 1.1 µl | 32.99 |
| Rhus toxicodendron | 10X | 1.1 µl | 12.99 |
| Rhus toxicodendron | 30X | 1.1 µl | 32.99 |
| Spiraea ulmaria | 10X | 1.1 µl | 12.99 |
| Spiraea ulmaria | 30X | 1.1 µl | 32.99 |
List of Excipients:
Water for injection 1,072.5 μl
Sodium Chloride 10.3 μl
INDICATIONS AND USAGE
Neuralgo-Rheum® Injection Solution is a homeopathic drug product indicated for the treatment of nerve pain, soft tissue rheumatism and symptoms of disc protrusion.
DOSAGE AND ADMINISTRATION
General Considerations
The dosage schedules listed below can be used as a general guide for the administration of Neuralgo-Rheum Injection Solution.
• Neuralgo-Rheum Injection Solution may be administered s.c., i.d., i.m., or i.v..
• If co-administration with a local anesthetic is desired, Neuralgo-Rheum Injection Solution may be mixed with lidocaine or similar agents at the discretion of the physician.
• The interval between injections is left to the discretion of the HCP, but should not exceed 1 ampule in 24 hours.
• Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
• Draw up required dose into syringe.
• Discard any unused ampule contents. Do not reuse ampule.
• Only licensed practitioners with sufficient expertise in injecting drugs, including the respective route of administration, should administer the product.
Standard Dosage:
Adults and children 12 years and older: 1 ml 1 to 3 times per 7 days.
Children 6 to 11 years: 0.7 ml 1 to 3 times per 7 days.
Children 2 to 5 years: 0.5 ml 1 to 3 times per 7 days.
Acute Dosage:
Adults and children 12 years and older: 1 ml daily, and then continue with standard dosage.
Children 6 to 11 years: 0.7 ml daily, and then continue with standard dosage.
Children 2 to 5 years: 0.5 ml daily, and then continue with standard dosage.
CONTRAINDICATIONS
Neuralgo-Rheum Injection Solution is contraindicated in patients with known hypersensitivity to Neuralgo-Rheum or any of its ingredients.
ADVERSE REACTIONS
Post-marketing Experience
No adverse events have been reported with a causal relationship to Neuralgo-Rheum Injection Solution.
To report SUSPECTED ADVERSE REACTIONS, contact MediNatura. at 1.844.633.4628 or info@medinatura.com or FDA at 1-800-FDA-1088
OVERDOSAGE
No negative effects of an overdose have been reported and none are expected due to the homeopathic dilutions.
CLINICAL PHARMACOLOGY
Mechanism of Action
The exact mechanism of Neuralgo-Rheum Injection Solution is not fully understood.
Pharmacodynamics
Not applicable for homeopathic medicinal products.
| NEURALGO RHEUM
pseudognaphalium obtusifolium, causticum, colchicum autumnale bulb, citrullus colocynthis fruit pulp, iron, lithium benzoate, toxicodendron pubescens leaf and filipendula ulmaria root injection |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - MediNatura Inc (102783016) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Biologische Heilmittel Heel | 315635359 | manufacture(50114-7070) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Siegfried Hameln GmbH | 315869123 | manufacture(50114-7070) | |