Label: DUETACT- pioglitazone and glimepiride tablet
- NDC Code(s): 64764-302-30, 64764-302-90, 64764-304-30, 64764-304-90
- Packager: Takeda Pharmaceuticals America, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated October 24, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DUETACT safely and effectively. See full prescribing information for DUETACT.
DUETACT (pioglitazone and glimepiride) tablets for oral use
Initial U.S. Approval: 2006WARNING: CONGESTIVE HEART FAILURE
See full prescribing information for complete boxed warning.
- •
- Thiazolidinediones, including pioglitazone, which is a component of DUETACT, cause or exacerbate congestive heart failure in some patients. (5.1)
- •
- After initiation of DUETACT, and after dose increases, monitor patients carefully for signs and symptoms of heart failure (e.g., excessive, rapid weight gain, dyspnea, and/or edema). If heart failure develops, it should be managed according to current standards of care and discontinuation or dose reduction of DUETACT must be considered. (5.1)
- •
- DUETACT is not recommended in patients with symptomatic heart failure. (5.1)
- •
- Initiation of DUETACT in patients with established New York Heart Association (NYHA) Class III or IV heart failure is contraindicated. (4, 5.1)
RECENT MAJOR CHANGES
Warnings and Precautions Urinary bladder tumors (5.6) 12/2016 INDICATIONS AND USAGE
DUETACT is a thiazolidinedione and a sulfonylurea combination product indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus when treatment with both pioglitazone and glimepiride is appropriate. (1)
Important Limitations of Use:
- •
- Not for treatment of type 1 diabetes or diabetic ketoacidosis. (1)
DOSAGE AND ADMINISTRATION
- •
- Individualize the starting dose of DUETACT based on the patient's current regimen. (2.1)
- •
- May adjust the dosing based on effectiveness and tolerability while not exceeding the maximum recommended daily dose of pioglitazone 45 mg and glimepiride 8 mg. (2.1)
- •
- DUETACT should be given in a single dose once daily with meals. (2.1)
- •
- Obtain liver tests before starting DUETACT. If abnormal, use caution when treating with DUETACT, investigate the probable cause, treat (if possible) and follow appropriately. Monitoring liver tests while on DUETACT is not recommended in patients without liver disease. (5.5)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
- •
- Initiation in patients with established New York Heart Association (NYHA) Class III or IV heart failure [see Boxed Warning]. (4)
- •
- Use in patients with known hypersensitivity to pioglitazone, glimepiride or any other component of DUETACT. (4)
- •
- Hypersensitivity to sulfonamide derivatives. (4)
WARNINGS AND PRECAUTIONS
- •
- Congestive heart failure: Fluid retention may occur and can exacerbate or lead to congestive heart failure. Combination use with insulin and use in congestive heart failure NYHA Class I and II may increase risk. Monitor patients for signs and symptoms. (5.1)
- •
- Hypoglycemia: May be severe. When insulin or an insulin secretagogue is used with pioglitazone, a lower dose of the insulin or insulin secretagogue may be needed to reduce the risk of hypoglycemia. (5.2)
- •
- Hypersensitivity Reactions: Postmarketing reports for glimepiride, a component of DUETACT, include anaphylaxis, angioedema and Stevens-Johnson Syndrome. Promptly discontinue DUETACT, assess for other cases, institute appropriate monitoring and treatment, and initiate alternative treatment for diabetes. (5.3)
- •
- Potential increased risk of cardiovascular mortality with sulfonylureas: Inform patients of risk, benefits, and treatment alternatives. (5.4)
- •
- Hepatic effects: Postmarketing reports of hepatic failure, sometimes fatal. Causality cannot be excluded. If liver injury is detected, promptly interrupt DUETACT and assess patient for probable cause, then treat cause if possible, to resolution or stabilization. Do not restart DUETACT if liver injury is confirmed and no alternate etiology can be found. (5.5)
- •
- Bladder cancer: May increase the risk of bladder cancer. Do not use in patients with active bladder cancer. Use caution when using in patients with a prior history of bladder cancer (5.6)
- •
- Edema: Dose-related edema may occur. (5.7)
- •
- Fractures: Increased incidence in female patients. Apply current standards of care for assessing and maintaining bone health. (5.8)
- •
- Hemolytic anemia: Can occur if glucose 6-phophate dehydrogenase (GP6D) deficient. Use with caution in patients with GP6D deficiency. (5.9)
- •
- Macular edema: Postmarketing reports. Recommend regular eye exams in all patients with diabetes according to current standards of care with prompt evaluation for acute visual changes. (5.10)
- •
- Macrovascular outcomes: There have been no clinical studies establishing conclusive evidence of macrovascular risk reduction with DUETACT. (5.11)
ADVERSE REACTIONS
Most common adverse reactions (≥5%) are upper respiratory tract infection, accidental injury, and combined edema/peripheral edema. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals America, Inc. at 1-877-825-3327 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Strong CYP2C8 inhibitors (e.g., gemfibrozil) increase pioglitazone concentrations. Limit pioglitazone dose to 15 mg daily. (2.3, 7.1)
- •
- CYP2C8 inducers (e.g., rifampin) may decrease pioglitazone concentrations. (7.2)
- •
- Topiramate may decrease pioglitazone concentrations. (7.3)
- •
- Certain medications may affect glucose metabolism, requiring DUETACT dose adjustment and close monitoring of blood glucose (7.4).
- •
- Miconazole: Severe hypoglycemia can occur when DUETACT and oral miconazole are used concomitantly. (7.5)
- •
- CYP2C9 interactions: Inhibitors and inducers may affect glycemic control by altering glimepiride plasma concentrations. (7.6)
- •
- Colesevelam: Coadministration may reduce glimepiride absorption. DUETACT should be administered at least 4 hours prior to colesevelam. (2.4, 7.7)
USE IN SPECIFIC POPULATIONS
- •
- Females and Males of Reproductive Potential: Advise premenopausal females of the potential for an unintended pregnancy. (8.3)
- •
- Pediatrics: Not recommended for use in pediatric patients. (8.4)
- •
- Geriatric or renally impaired patients: At risk for hypoglycemia with DUETACT. Use caution in dose selection and titration, and monitor closely. (8.5, 8.6)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 6/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: CONGESTIVE HEART FAILURE
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommendations for All Patients
2.2 Concomitant Use with an Insulin Secretagogue or Insulin
2.3 Concomitant Use with Strong CYP2C8 Inhibitors
2.4 Concomitant Use with Colesevelam
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Congestive Heart Failure
5.2 Hypoglycemia
5.3 Hypersensitivity Reactions
5.4 Potential Increased Risk of Cardiovascular Mortality with Sulfonylureas
5.5 Hepatic Effects
5.6 Urinary Bladder Tumors
5.7 Edema
5.8 Fractures
5.9 Hemolytic Anemia
5.10 Macular Edema
5.11 Macrovascular Outcomes
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Strong CYP2C8 Inhibitors
7.2 CYP2C8 Inducers
7.3 Topiramate
7.4 Drugs Affecting Glucose Metabolism
7.5 Miconazole
7.6 CYP2C9 Interactions
7.7 Concomitant Administration of Colesevelam
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: CONGESTIVE HEART FAILURE
- •
- Thiazolidinediones, including pioglitazone, which is a component of DUETACT, cause or exacerbate congestive heart failure in some patients [see Warnings and Precautions (5.1)].
- •
- After initiation of DUETACT and after dose increases, monitor patients carefully for signs and symptoms of heart failure (e.g., excessive, rapid weight gain, dyspnea, and/or edema). If heart failure develops, it should be managed according to current standards of care and discontinuation or dose reduction of DUETACT must be considered [see Warnings and Precautions (5.1)].
- •
- DUETACT is not recommended in patients with symptomatic heart failure [see Warnings and Precautions (5.1)].
- •
- Initiation of DUETACT in patients with established New York Heart Association (NYHA) Class III or IV heart failure is contraindicated [see Contraindications (4) and Warnings and Precautions (5.1)].
-
1 INDICATIONS AND USAGE
DUETACT is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus who are already treated with a thiazolidinedione and sulfonylurea or who have inadequate glycemic control on a thiazolidinedione alone or a sulfonylurea alone [see Clinical Studies (14)].
Important Limitations of Use
Pioglitazone exerts its antihyperglycemic effect only in the presence of endogenous insulin. DUETACT should not be used to treat type 1 diabetes or diabetic ketoacidosis, as it would not be effective in these settings.
Use caution in patients with liver disease [see Warnings and Precautions (5.5)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommendations for All Patients
DUETACT should be taken once daily with the first main meal.
DUETACT tablets are available as a 30 mg pioglitazone plus 2 mg glimepiride or a 30 mg pioglitazone plus 4 mg glimepiride tablet. If therapy with a combination tablet containing pioglitazone and glimepiride is considered appropriate the recommended starting dose is:
- •
- 30 mg/2 mg or 30 mg/4 mg once daily and gradually titrated, as needed, after assessing adequacy of therapeutic response and tolerability,
- •
- for patients inadequately controlled on glimepiride monotherapy: 30 mg/2 mg or 30 mg/4 mg once daily and gradually titrated, as needed, after assessing adequacy of therapeutic response and tolerability,
- •
- for patients inadequately controlled on pioglitazone monotherapy: 30 mg/2 mg once daily and gradually titrated, as needed, after assessing adequacy of therapeutic response and tolerability,
- •
- for patients who are changing from combination therapy of pioglitazone plus glimepiride as separate tablets: DUETACT should be taken at doses that are as close as possible to the dose of pioglitazone and glimepiride already being taken,
- •
- for patients currently on a different sulfonylurea monotherapy or switching from combination therapy of pioglitazone plus a different sulfonylurea (e.g., glyburide, glipizide, chlorpropamide, tolbutamide, acetohexamide): 30 mg/2 mg once daily and adjusted after assessing adequacy of therapeutic response. Observe for hypoglycemia for one to two weeks due to the potential overlapping drug effect.
- •
- for patients with systolic dysfunction, the lowest approved dose of DUETACT should be prescribed only after titration from 15 mg to 30 mg of pioglitazone has been safely tolerated.
After initiation of DUETACT or with dose increase, monitor patients carefully for hypoglycemia and adverse reactions related to fluid retention such as weight gain, edema, and signs and symptoms of congestive heart failure [see Boxed Warning and Warnings and Precautions (5.7)].
Liver tests (serum alanine and aspartate aminotransferases, alkaline phosphatase, and total bilirubin) should be obtained prior to initiating DUETACT. Routine periodic monitoring of liver tests during treatment with DUETACT is not recommended in patients without liver disease. Patients who have liver test abnormalities prior to initiation of DUETACT or who are found to have abnormal liver tests while taking DUETACT should be managed as described under Warnings and Precautions [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.3)].
2.2 Concomitant Use with an Insulin Secretagogue or Insulin
If hypoglycemia occurs in a patient coadministered DUETACT and an insulin secretagogue, the dose of the insulin secretagogue should be reduced.
If hypoglycemia occurs in a patient coadministered DUETACT and insulin, the dose of insulin should be decreased by 10% to 25%. Further adjustments to the insulin dose should be individualized based on glycemic response.
2.3 Concomitant Use with Strong CYP2C8 Inhibitors
Coadministration of pioglitazone and gemfibrozil, a strong CYP2C8 inhibitor, increases pioglitazone exposure approximately 3-fold. Therefore, the maximum recommended dose of pioglitazone is 15 mg daily when used in combination with gemfibrozil or other strong CYP2C8 inhibitors. If gemfibrozil or other CYP2C8 inhibitors need to co-administered, patients should switch to individual components of DUETACT because the minimum dose of pioglitazone in DUETACT exceeds 15 mg [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
2.4 Concomitant Use with Colesevelam
When colesevelam is coadministered with glimepiride, maximum plasma concentration and total exposure to glimepiride is reduced. Therefore, DUETACT should be administered at least four hours prior to colesevelam [see Drug Interactions (7.7) and Clinical Pharmacology (12.3)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
- •
- Initiation in patients with established NYHA Class III or IV heart failure [see Boxed Warning].
- •
- Use in patients with known hypersensitivity to pioglitazone, glimepiride or any other component of DUETACT [see Warnings and Precautions (5.3)].
- •
- Use in patients with known history of an allergic reaction to sulfonamide derivatives.
Reported hypersensitivity reactions with glimepiride include cutaneous eruptions with or without pruritus as well as more serious reactions (e.g., anaphylaxis, angioedema, Stevens-Johnson Syndrome, dyspnea) [see Warnings and Precautions (5.3) and Adverse Reactions (6.2)]
-
5 WARNINGS AND PRECAUTIONS
5.1 Congestive Heart Failure
Pioglitazone
Pioglitazone, like other thiazolidinediones, can cause dose-related fluid retention when used alone or in combination with other antidiabetic medications and is most common when DUETACT is used in combination with insulin. Fluid retention may lead to or exacerbate congestive heart failure. Patients should be observed for signs and symptoms of congestive heart failure. If congestive heart failure develops, it should be managed according to current standards of care and discontinuation or dose reduction of DUETACT must be considered [see Boxed Warning, Contraindications (4) and Adverse Reactions (6.1)].
5.2 Hypoglycemia
Glimepiride
All sulfonylureas, including glimepiride, a component of DUETACT, can cause severe hypoglycemia [see Adverse Reactions (6.1)]. The patient's ability to concentrate and react may be impaired as a result of hypoglycemia. These impairments may present a risk in situations where these abilities are especially important, such as driving or operating other machinery. Severe hypoglycemia can lead to unconsciousness or convulsions and may result in temporary or permanent impairment of brain function or death.
Patients must be educated to recognize and manage hypoglycemia. Use caution when initiating and increasing DUETACT doses in patients who may be predisposed to hypoglycemia (e.g., the elderly, patients with renal impairment, patients on other antidiabetic medications). Debilitated or malnourished patients and those with adrenal, pituitary, or hepatic impairment are particularly susceptible to the hypoglycemic action of glucose-lowering medications. Hypoglycemia is also more likely to occur when caloric intake is deficient, after severe or prolonged exercise, or when alcohol is ingested.
Early warning symptoms of hypoglycemia may be different or less pronounced in patients with autonomic neuropathy, the elderly, and in patients who are taking beta-adrenergic blocking medications or other sympatholytic agents. These situations may result in severe hypoglycemia before the patient is aware of the hypoglycemia.
5.3 Hypersensitivity Reactions
Glimepiride
There have been postmarketing reports of hypersensitivity reactions in patients treated with glimepiride, a component of DUETACT, including serious reactions such as anaphylaxis, angioedema, and Stevens-Johnson Syndrome. If a hypersensitivity reaction is suspected, promptly discontinue DUETACT, assess for other potential causes for the reaction, and institute alternative treatment for diabetes.
5.4 Potential Increased Risk of Cardiovascular Mortality with Sulfonylureas
Glimepiride
The administration of oral hypoglycemic drugs has been reported to be associated with increased cardiovascular mortality as compared to treatment with diet alone or diet plus insulin. This warning is based on the study conducted by the University Group Diabetes Program (UGDP), a long-term, prospective clinical trial designed to evaluate the effectiveness of glucose-lowering drugs in preventing or delaying vascular complications in patients with non-insulin-dependent diabetes. The study involved 823 patients who were randomly assigned to one of four treatment groups.
UGDP reported that patients treated for 5 to 8 years with diet plus a fixed dose of tolbutamide (1.5 grams per day) had a rate of cardiovascular mortality approximately 2.5 times that of patients treated with diet alone. A significant increase in total mortality was not observed, but the use of tolbutamide was discontinued based on the increase in cardiovascular mortality, thus limiting the opportunity for the study to show an increase in overall mortality. Despite controversy regarding the interpretation of these results, the findings of the UGDP study provide an adequate basis for this warning. The patient should be informed of the potential risks and advantages of glimepiride tablets and of alternative modes of therapy.
Although only one drug in the sulfonylurea class (tolbutamide) was included in this study, it is prudent from a safety standpoint to consider that this warning may also apply to other oral hypoglycemic drugs in this class, in view of their close similarities in mode of action and chemical structure.
5.5 Hepatic Effects
Pioglitazone
There have been postmarketing reports of fatal and non-fatal hepatic failure in patients taking pioglitazone, although the reports contain insufficient information necessary to establish the probable cause. There has been no evidence of drug-induced hepatotoxicity in the pioglitazone-controlled clinical trial database to date [see Adverse Reactions (6.1)].
Patients with type 2 diabetes may have fatty liver disease or cardiac disease with episodic congestive heart failure, both of which may cause liver test abnormalities, and they may also have other forms of liver disease, many of which can be treated or managed. Therefore, obtaining a liver test panel (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase, and total bilirubin) and assessing the patient is recommended before initiating DUETACT therapy. In patients with abnormal liver tests, DUETACT should be initiated with caution.
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient is found to have abnormal liver tests (ALT greater than 3 times the upper limit of the reference range), DUETACT treatment should be interrupted and investigation done to establish the probable cause. DUETACT should not be restarted in these patients without another explanation for the liver test abnormalities.
Patients who have serum ALT greater than three times the reference range with serum total bilirubin greater than two times the reference range without alternative etiologies are at risk for severe drug-induced liver injury and should not be restarted on DUETACT. For patients with lesser elevations of serum ALT or bilirubin and with an alternate probable cause, treatment with DUETACT can be used with caution.
5.6 Urinary Bladder Tumors
Pioglitazone
Tumors were observed in the urinary bladder of male rats in the two-year carcinogenicity study [see Nonclinical Toxicology (13.1)]. In addition, during the three year PROactive clinical trial, 14 patients out of 2605 (0.54%) randomized to pioglitazone and 5 out of 2633 (0.19%) randomized to placebo were diagnosed with bladder cancer. After excluding patients in whom exposure to study drug was less than one year at the time of diagnosis of bladder cancer, there were 6 (0.23%) cases on pioglitazone and two (0.08%) cases on placebo. After completion of the trial, a large subset of patients was observed for up to 10 additional years, with little additional exposure to pioglitazone. During the 13 years of both PROactive and observational follow-up, the occurrence of bladder cancer did not differ between patients randomized to pioglitazone or placebo (HR =1.00; [95% CI: 0.59–1.72]).
Findings regarding the risk of bladder cancer in patients exposed to pioglitazone vary among observational studies; some did not find an increased risk of bladder cancer associated with pioglitazone, while others did.
A large prospective 10-year observational cohort study conducted in the United States found no statistically significant increase in the risk of bladder cancer in diabetic patients ever exposed to pioglitazone, compared to those never exposed to pioglitazone (HR =1.06 [95% CI 0.89–1.26]).
A retrospective cohort study conducted with data from the United Kingdom found a statistically significant association between ever exposure to pioglitazone and bladder cancer (HR: 1.63; [95% CI: 1.22–2.19]).
Associations between cumulative dose or cumulative duration of exposure to pioglitazone and bladder cancer were not detected in some studies including the 10-year observational study in the U.S., but were in others. Inconsistent findings and limitations inherent in these and other studies preclude conclusive interpretations of the observational data.
Pioglitazone may be associated with an increase in the risk of urinary bladder tumors. There are insufficient data to determine whether pioglitazone is a tumor promoter for urinary bladder tumors.
Consequently, DUETACT should not be used in patients with active bladder cancer and the benefits of glycemic control versus unknown risks for cancer recurrence with DUETACT should be considered in patients with a prior history of bladder cancer.
5.7 Edema
Pioglitazone
In controlled clinical trials, edema was reported more frequently in patients treated with pioglitazone than in placebo-treated patients and is dose-related [see Adverse Reactions (6.1)]. In postmarketing experience, reports of new onset or worsening edema have been received.
DUETACT should be used with caution in patients with edema. Because thiazolidinediones, including pioglitazone, can cause fluid retention, which can exacerbate or lead to congestive heart failure, DUETACT should be used with caution in patients at risk for congestive heart failure. Patients treated with DUETACT should be monitored for signs and symptoms of congestive heart failure [see Boxed Warning, Warnings and Precautions (5.1), and Patient Counseling Information (17)].
5.8 Fractures
Pioglitazone
In PROactive (the Prospective Pioglitazone Clinical Trial in Macrovascular Events), 5238 patients with type 2 diabetes and a history of macrovascular disease were randomized to pioglitazone (N=2605), force-titrated up to 45 mg daily or placebo (N=2633) in addition to standard of care. During a mean follow-up of 34.5 months, the incidence of bone fracture in females was 5.1% (44/870) for pioglitazone versus 2.5% (23/905) for placebo. This difference was noted after the first year of treatment and persisted during the course of the study. The majority of fractures observed in female patients were nonvertebral fractures including lower limb and distal upper limb. No increase in the incidence of fracture was observed in men treated with pioglitazone (1.7%) versus placebo (2.1%). The risk of fracture should be considered in the care of patients, especially female patients, treated with DUETACT and attention should be given to assessing and maintaining bone health according to current standards of care.
5.9 Hemolytic Anemia
Glimepiride
Sulfonylureas can cause hemolytic anemia in patients with glucose 6-phosphate dehydrogenase (G6PD) deficiency. Because DUETACT contains glimepiride, which belongs to the class of sulfonylurea agents, use caution in patients with G6PD deficiency and consider the use of a nonsulfonylurea alternative. There are also postmarketing reports of hemolytic anemia in patients receiving glimepiride who did not have known G6PD deficiency [see Adverse Reactions (6.2)].
5.10 Macular Edema
Pioglitazone
Macular edema has been reported in postmarketing experience in diabetic patients who were taking pioglitazone or another thiazolidinedione. Some patients presented with blurred vision or decreased visual acuity, but others were diagnosed on routine ophthalmologic examination.
Most patients had peripheral edema at the time macular edema was diagnosed. Some patients had improvement in their macular edema after discontinuation of the thiazolidinedione.
Patients with diabetes should have regular eye exams by an ophthalmologist according to current standards of care. Patients with diabetes who report any visual symptoms should be promptly referred to an ophthalmologist, regardless of the patient's underlying medications or other physical findings [see Adverse Reactions (6.1)].
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed elsewhere in the labeling:
- •
- Congestive Heart Failure [see Boxed Warning and Warnings and Precautions (5.1)]
- •
- Hypoglycemia [see Warnings and Precautions (5.2)]
- •
- Edema [see Warnings and Precautions (5.7)]
- •
- Fractures [see Warnings and Precautions (5.8)]
- •
- Hemolytic Anemia [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The adverse events reported in at least 5% of patients in the controlled 16-week clinical studies between placebo plus a sulfonylurea and pioglitazone (15 mg and 30 mg combined) plus sulfonylurea treatment arms were upper respiratory tract infection (15.5% and 16.6%), accidental injury (8.6% and 3.5%), and combined edema/peripheral edema (2.1% and 7.2%), respectively.
The incidence and type of adverse events reported in at least 5% of patients in any combined treatment group from the 24-week study comparing pioglitazone 30 mg plus a sulfonylurea and pioglitazone 45 mg plus a sulfonylurea are shown in Table 1; the rate of adverse events resulting in study discontinuation between the two treatment groups was 6% and 9.7%, respectively.
Table 1. Adverse Events that Occurred in ≥5% of Patients in Any Treatment Group During the 24-Week Study
Adverse Event
Pioglitazone 30 mg
+ Sulfonylurea
N=351
n (%)Pioglitazone 45 mg
+ Sulfonylurea
N=351
n (%)Hypoglycemia
47 (13.4)
55 (15.7)
Upper Respiratory Tract Infection
43 (12.3)
52 (14.8)
Weight Increased
32 (9.1)
47 (13.4)
Edema Lower Limb
20 (5.7)
43 (12.3)
Headache
25 (7.1)
14 (4.0)
Urinary Tract Infection
20 (5.7)
24 (6.8)
Diarrhea
21 (6.0)
15 (4.3)
Nausea
18 (5.1)
14 (4.0)
Pain in Limb
19 (5.4)
14 (4.0)
In US double-blind studies, anemia was reported in ≤2% of patients treated with pioglitazone plus a sulfonylurea [see Warnings and Precautions (5.9)].
Pioglitazone
Over 8500 patients with type 2 diabetes have been treated with pioglitazone in randomized, double-blind, controlled clinical trials, including 2605 patients with type 2 diabetes and macrovascular disease treated with pioglitazone in the PROactive clinical trial. In these trials, over 6000 patients have been treated with pioglitazone for six months or longer, over 4500 patients have been treated with pioglitazone for one year or longer, and over 3000 patients have been treated with pioglitazone for at least two years.
In six pooled 16- to 26-week placebo-controlled monotherapy and 16- to 24-week add-on combination therapy trials, the incidence of withdrawals due to adverse events was 4.5% for patients treated with pioglitazone and 5.8% for comparator-treated patients. The most common adverse events leading to withdrawal were related to inadequate glycemic control, although the incidence of these events was lower (1.5%) with pioglitazone than with placebo (3.0%).
In the PROactive trial, the incidence of withdrawals due to adverse events was 9.0% for patients treated with pioglitazone and 7.7% for placebo-treated patients. Congestive heart failure was the most common serious adverse event leading to withdrawal occurring in 1.3% of patients treated with pioglitazone and 0.6% of patients treated with placebo.
Common Adverse Events: 16- to 26-Week Monotherapy Trials:
A summary of the incidence and type of common adverse events reported in three pooled 16- to 26-week placebo-controlled monotherapy trials of pioglitazone is provided in Table 2. Terms that are reported represent those that occurred at an incidence of >5% and more commonly in patients treated with pioglitazone than in patients who received placebo. None of these adverse events were related to the pioglitazone dose.
Table 2. Three Pooled 16- to 26-Week Placebo-Controlled Clinical Trials of Pioglitazone Monotherapy: Adverse Events Reported at an Incidence >5% and More Commonly in Patients Treated with Pioglitazone than in Patients Treated with Placebo
% of Patients
Placebo
N=259Pioglitazone
N=606Upper Respiratory Tract Infection
8.5
13.2
Headache
6.9
9.1
Sinusitis
4.6
6.3
Myalgia
2.7
5.4
Pharyngitis
0.8
5.1
A summary of the overall incidence and types of common adverse events reported in the PROactive trial is provided in Table 3. Terms that are reported represent those that occurred at an incidence of >5% and more commonly in patients treated with pioglitazone than in patients who received placebo.
Mean duration of patient follow-up was 34.5 months. Table 3. PROactive Trial: Incidence and Types of Adverse Events Reported in >5% of Patients Treated with Pioglitazone and More Commonly than Placebo
% of Patients
Placebo
N=2633Pioglitazone
N=2605Hypoglycemia
18.8
27.3
Edema
15.3
26.7
Cardiac Failure
6.1
8.1
Pain in Extremity
5.7
6.4
Back Pain
5.1
5.5
Chest Pain
5.0
5.1
Congestive Heart Failure
A summary of the incidence of adverse events related to congestive heart failure is provided in Table 4 for the 16- to 24-week add-on to sulfonylurea trials, for the 16- to 24-week add-on to insulin trials, and for the 16- to 24-week add-on to metformin trials. None of the events were fatal.
Table 4. Treatment-Emergent Adverse Events of Congestive Heart Failure (CHF)
Patients Treated with Pioglitazone or Placebo Added on to a Sulfonylurea
Number (%) of Patients
Placebo-Controlled Trial
(16 weeks)Non-Controlled
Double Blind Trial
(24 weeks)Placebo
+ Sulfonylurea
N=187Pioglitazone
15 mg
+ Sulfonylurea
N=184Pioglitazone
30 mg
+ Sulfonylurea
N=189Pioglitazone
30 mg
+ Sulfonylurea
N=351Pioglitazone
45 mg
+ Sulfonylurea
N=351At least one congestive
heart failure event2 (1.1%)
0
0
1 (0.3%)
6 (1.7%)
Hospitalized
2 (1.1%)
0
0
0
2 (0.6%)
Patients Treated with Pioglitazone or Placebo Added on to Insulin
Number (%) of Patients
Placebo-Controlled Trial
(16 weeks)Non-Controlled
Double Blind Trial
(24 weeks)Placebo
+ Insulin
N=187Pioglitazone
15 mg
+ Insulin
N=191Pioglitazone
30 mg
+ Insulin
N=188Pioglitazone
30 mg
+ Insulin
N=345Pioglitazone
45 mg
+ Insulin
N=345At least one congestive
heart failure event0
2 (1.0%)
2 (1.1%)
3 (0.9%)
5 (1.4%)
Hospitalized
0
2 (1.0%)
1 (0.5%)
1 (0.3%)
3 (0.9%)
Patients Treated with Pioglitazone or Placebo Added on to Metformin
Number (%) of Patients
Placebo-Controlled Trial
(16 weeks)Non-Controlled
Double Blind Trial
(24 weeks)Placebo
+ Metformin
N=160Pioglitazone
30 mg
+ Metformin
N=168Pioglitazone
30 mg
+ Metformin
N=411Pioglitazone
45 mg
+ Metformin
N=416At least one congestive
heart failure event0
1 (0.6%)
0
1 (0.2%)
Hospitalized
0
1 (0.6%)
0
1 (0.2%)
Patients with type 2 diabetes and NYHA class II or early class III congestive heart failure were randomized to receive 24 weeks of double-blind treatment with either pioglitazone at daily doses of 30 mg to 45 mg (n=262) or glyburide at daily doses of 10 mg to 15 mg (n=256). A summary of the incidence of adverse events related to congestive heart failure reported in this study is provided in Table 5.
Table 5. Treatment-Emergent Adverse Events of Congestive Heart Failure (CHF) in Patients with NYHA Class II or III Congestive Heart Failure Treated with Pioglitazone or Glyburide
Number (%) of Subjects
Pioglitazone
N=262Glyburide
N=256Death due to cardiovascular causes (adjudicated)
5 (1.9%)
6 (2.3%)
Overnight hospitalization for worsening CHF (adjudicated)
26 (9.9%)
12 (4.7%)
Emergency room visit for CHF (adjudicated)
4 (1.5%)
3 (1.2%)
Patients experiencing CHF progression during study
35 (13.4%)
21 (8.2%)
Congestive heart failure events leading to hospitalization that occurred during the PROactive trial are summarized in Table 6.
Table 6. Treatment-Emergent Adverse Events of Congestive Heart Failure (CHF) in PROactiveTrial
Number (%) of Patients
Placebo
N=2633Pioglitazone
N=2605At least one hospitalized CHF event
108 (4.1%)
149 (5.7%)
Fatal
22 (0.8%)
25 (1%)
Hospitalized, nonfatal
86 (3.3%)
124 (4.7%)
Cardiovascular Safety
In the PROactive trial, 5238 patients with type 2 diabetes and a history of macrovascular disease were randomized to pioglitazone (N=2605), force-titrated up to 45 mg daily or placebo (N=2633) in addition to standard of care. Almost all patients (95%) were receiving cardiovascular medications (beta blockers, ACE inhibitors, angiotensin II receptor blockers, calcium channel blockers, nitrates, diuretics, aspirin, statins, and fibrates). At baseline, patients had a mean age of 62 years, mean duration of diabetes of 9.5 years, and mean HbA1c of 8.1%. Mean duration of follow-up was 34.5 months.
The primary objective of this trial was to examine the effect of pioglitazone on mortality and macrovascular morbidity in patients with type 2 diabetes mellitus who were at high risk for macrovascular events. The primary efficacy variable was the time to the first occurrence of any event in a cardiovascular composite endpoint that included all-cause mortality, nonfatal myocardial infarction (MI) including silent MI, stroke, acute coronary syndrome, cardiac intervention including coronary artery bypass grafting or percutaneous intervention, major leg amputation above the ankle, and bypass surgery or revascularization in the leg. A total of 514 (19.7%) patients treated with pioglitazone and 572 (21.7%) placebo-treated patients experienced at least one event from the primary composite endpoint (hazard ratio 0.90; 95% Confidence Interval: 0.80, 1.02; p=0.10).
Although there was no statistically significant difference between pioglitazone and placebo for the three-year incidence of a first event within this composite, there was no increase in mortality or in total macrovascular events with pioglitazone. The number of first occurrences and total individual events contributing to the primary composite endpoint is shown in Table 7.
Table 7. PROactive: Number of First and Total Events for Each Component within the Cardiovascular Composite Endpoint
Cardiovascular Events
Placebo
N=2633Pioglitazone
N=2605First
Events
n (%)Total
Events
nFirst
Events
n (%)Total
Events
nAny event
572 (21.7)
900
514 (19.7)
803
All-cause mortality
122 (4.6)
186
110 (4.2)
177
Non-fatal myocardial infarction (MI)
118 (4.5)
157
105 (4.0)
131
Stroke
96 (3.6)
119
76 (2.9)
92
Acute coronary syndrome
63 (2.4)
78
42 (1.6)
65
Cardiac intervention (CABG/PCI)
101 (3.8)
240
101 (3.9)
195
Major leg amputation
15 (0.6)
28
9 (0.3)
28
Leg revascularization
57 (2.2)
92
71 (2.7)
115
CABG = coronary artery bypass grafting; PCI = percutaneous intervention
Weight Gain
Dose-related weight gain occurs when pioglitazone is used alone or in combination with other antidiabetic medications. The mechanism of weight gain is unclear but probably involves a combination of fluid retention and fat accumulation.
Tables 8 and 9 summarize the changes in body weight with pioglitazone and placebo in the 16- to 26-week randomized, double-blind monotherapy and 16- to 24-week combination add-on therapy trials and in the PROactive trial.
Table 8. Weight Changes (kg) from Baseline during Randomized, Double-Blind Clinical Trials
Control Group
(Placebo)Pioglitazone
15 mgPioglitazone
30 mgPioglitazone
45 mgMedian
(25th/75th
percentile)Median
(25th/75th
percentile)Median
(25th/75th
percentile)Median
(25th/75th
percentile)Monotherapy (16 to 26 weeks)
-1.4 (-2.7/0.0)
N=2560.9 (-0.5/3.4)
N=791.0 (-0.9/3.4)
N=1882.6 (0.2/5.4)
N=79Combination
Therapy
(16 to 24 weeks)Sulfonylurea
-0.5 (-1.8/0.7)
N=1872.0 (0.2/3.2)
N=1833.1 (1.1/5.4)
N=5284.1 (1.8/7.3)
N=333Metformin
-1.4 (-3.2/0.3)
N=160N/A
0.9 (-1.3/3.2)
N=5671.8 (-0.9/5.0)
N=407Insulin
0.2 (-1.4/1.4)
N=1822.3 (0.5/4.3)
N=1903.3 (0.9/6.3)
N=5224.1 (1.4/6.8)
N=338Table 9. Median Change in Body Weight in Patients Treated with Pioglitazone vs Patients Treated with Placebo During the Double-Blind Treatment Period in the PROactive Trial
Placebo
Pioglitazone
Median
(25th/75th
percentile)Median
(25th/75th
percentile)Change from baseline to final visit (kg)
-0.5 (-3.3, 2.0)
N=2581+3.6 (0.0, 7.5)
N=2560Note: Median exposure for both Pioglitazone and Placebo was 2.7 years
Edema
Edema induced from taking pioglitazone is reversible when pioglitazone is discontinued. The edema usually does not require hospitalization unless there is coexisting congestive heart failure. A summary of the frequency and types of edema adverse events occurring in clinical investigations of pioglitazone is provided in Table 10.
Table 10. Adverse Events of Edema in Patients Treated with PioglitazoneNumber (%) of Patients
Placebo
Pioglitazone
15 mgPioglitazone
30 mgPioglitazone
45 mgMonotherapy (16 to 26 weeks)
3 (1.2%)
N=2592 (2.5%)
N= 8113 (4.7%)
N= 27511 (6.5%)
N=169Combined Therapy
(16 to 24 weeks)Sulfonylurea
4 (2.1%)
N=1873 (1.6%)
N=18461 (11.3%)
N=54081 (23.1%)
N=351Metformin
4 (2.5%)
N=160N/A
34 (5.9%)
N=57958 (13.9%)
N=416Insulin
13 (7.0%)
N=18724 (12.6%)
N=191109 (20.5%)
N=53390 (26.1%)
N=345Note: The preferred terms of edema peripheral, generalized edema, pitting edema and fluid retention were combined to form the aggregate term of "edema."
Table 11. Adverse Events of Edema in Patients in the PROactive Trial
Number (%) of Patients
Placebo
N=2633Pioglitazone
N=2605419 (15.9%)
712 (27.3%)
Note: The preferred terms of edema peripheral, generalized edema, pitting edema and fluid retention were combined to form the aggregate term of "edema."
Hepatic Effects
There has been no evidence of pioglitazone-induced hepatotoxicity in the pioglitazone-controlled clinical trial database to date. One randomized, double-blind, 3-year trial comparing pioglitazone to glyburide as add-on to metformin and insulin therapy was specifically designed to evaluate the incidence of serum ALT elevation to greater than three times the upper limit of the reference range, measured every eight weeks for the first 48 weeks of the trial then every 12 weeks thereafter. A total of 3/1051 (0.3%) patients treated with pioglitazone and 9/1046 (0.9%) patients treated with glyburide developed ALT values greater than three times the upper limit of the reference range. None of the patients treated with pioglitazone in the pioglitazone-controlled clinical trial database to date have had a serum ALT greater than three times the upper limit of the reference range and a corresponding total bilirubin greater than two times the upper limit of the reference range, a combination predictive of the potential for severe drug-induced liver injury.
Hypoglycemia
In the pioglitazone clinical trials, adverse events of hypoglycemia were reported based on clinical judgment of the investigators and did not require confirmation with fingerstick glucose testing.
In the 16-week add-on to sulfonylurea trial, the incidence of reported hypoglycemia was 3.7% with pioglitazone 30 mg and 0.5% with placebo. In the 16-week add-on to insulin trial, the incidence of reported hypoglycemia was 7.9% with pioglitazone 15 mg, 15.4% with pioglitazone 30 mg, and 4.8% with placebo.
The incidence of reported hypoglycemia was higher with pioglitazone 45 mg compared to pioglitazone 30 mg in both the 24-week add-on to sulfonylurea trial (15.7% versus 13.4%) and in the 24-week add-on to insulin trial (47.8% versus 43.5%).
Three patients in these four trials were hospitalized due to hypoglycemia. All three patients were receiving pioglitazone 30 mg (0.9%) in the 24-week add-on to insulin trial. An additional 14 patients reported severe hypoglycemia (defined as causing considerable interference with patient's usual activities) that did not require hospitalization. These patients were receiving pioglitazone 45 mg in combination with sulfonylurea (N=2) or pioglitazone 30 mg or 45 mg in combination with insulin (N=12).
Urinary Bladder Tumors
Tumors were observed in the urinary bladder of male rats in the two-year carcinogenicity study [see Nonclinical Toxicology (13.1)]. During the three year PROactive clinical trial, 14 patients out of 2605 (0.54%) randomized to pioglitazone and 5 out of 2633 (0.19%) randomized to placebo were diagnosed with bladder cancer. After excluding patients in whom exposure to study drug was less than one year at the time of diagnosis of bladder cancer, there were 6 (0.23%) cases on pioglitazone and two (0.08%) cases on placebo. After completion of the trial, a large subset of patients was observed for up to 10 additional years, with little additional exposure to pioglitazone. During the 13 years of both PROactive and observational follow-up, the occurrence of bladder cancer did not differ between patients randomized to pioglitazone or placebo (HR =1.00; 95% CI: 0.59-1.72) [see Warnings and Precautions (5.6)].
Glimepiride
Adverse events that occurred in controlled clinical trials with placebo and glimepiride monotherapy, other than hypoglycemia, included: headache (7.8% and 8.2%), accidental injury (3.4% and 5.8%), flu syndrome (4.4% and 5.4%), nausea (3.4% and 5.0%) and dizziness (2.4% and 5.0%), respectively.
Hypoglycemia
In a randomized, double-blind, placebo-controlled monotherapy trial of 14 weeks duration, patients already on sulfonylurea therapy underwent a 3-week washout period then were randomized to glimepiride 1 mg, 4 mg, 8 mg or placebo. Patients randomized to glimepiride 4 mg or 8 mg underwent forced-titration from an initial dose of 1 mg to these final doses, as tolerated. The overall incidence of possible hypoglycemia (defined by the presence of at least one symptom that the investigator believed might be related to hypoglycemia; a concurrent glucose measurement was not required) was 4% for glimepiride 1 mg, 17% for glimepiride 4 mg, 16% for glimepiride 8 mg and 0% for placebo. All of these events were self-treated.
In a randomized, double-blind, placebo-controlled monotherapy trial of 22 weeks duration, patients received a starting dose of either 1 mg glimepiride or placebo daily. The dose of glimepiride was titrated to a target fasting plasma glucose of 90 −150 mg/dL. Final daily doses of glimepiride were 1, 2, 3, 4, 6 or 8 mg. The overall incidence of possible hypoglycemia (as defined above for the 14-week trial) for glimepiride versus placebo was 19.7% vs. 3.2%. All of these events were self-treated.
Weight Gain
Glimepiride, like all sulfonylureas, can cause weight gain.
Allergic Reactions
In clinical trials, allergic reactions, such as pruritus, erythema, urticaria, and morbilliform or maculopapular eruptions, occurred in less than 1% of glimepiride-treated patients. These may resolve despite continued treatment with glimepiride. There are postmarketing reports of more serious allergic reactions (e.g., dyspnea, hypotension, shock) [see Warnings and Precautions (5.3)].
Laboratory Tests
Elevated Serum Alanine Aminotransferase (ALT)
In 11 pooled placebo-controlled trials of glimepiride, 1.9% of glimepiride-treated patients and 0.8% of placebo-treated patients developed serum ALT greater than two times the upper limit of the reference range.
Laboratory Abnormalities
Pioglitazone
Hematologic Effects
Pioglitazone may cause decreases in hemoglobin and hematocrit. In placebo-controlled monotherapy trials, mean hemoglobin values declined by 2% to 4% in patients treated with pioglitazone compared with a mean change in hemoglobin of -1% to +1% in placebo-treated patients. These changes primarily occurred within the first 4 to 12 weeks of therapy and remained relatively constant thereafter. These changes may be related to increased plasma volume associated with pioglitazone therapy and are not likely to be associated with any clinically significant hematologic effects.
Creatine Phosphokinase
During protocol-specified measurement of serum creatine phosphokinase (CPK) in pioglitazone clinical trials, an isolated elevation in CPK to greater than 10 times the upper limit of the reference range was noted in nine (0.2%) patients treated with pioglitazone (values of 2150 to 11400 IU/L) and in no comparator-treated patients. Six of these nine patients continued to receive pioglitazone, two patients were noted to have the CPK elevation on the last day of dosing and one patient discontinued pioglitazone due to the elevation. These elevations resolved without any apparent clinical sequelae. The relationship of these events to pioglitazone therapy is unknown.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of pioglitazone and glimepiride. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Pioglitazone
- •
- New onset or worsening diabetic macular edema with decreased visual acuity [see Warnings and Precautions (5.10)].
- •
- Fatal and nonfatal hepatic failure [see Warnings and Precautions (5.5)].
Postmarketing reports of congestive heart failure have been reported in patients treated with pioglitazone, both with and without previously known heart disease and both with and without concomitant insulin administration.
In postmarketing experience, there have been reports of unusually rapid increases in weight and increases in excess of that generally observed in clinical trials. Patients who experience such increases should be assessed for fluid accumulation and volume-related events such as excessive edema and congestive heart failure [see Boxed Warning and Warnings and Precautions (5.1)].
Glimepiride
- •
- Serious hypersensitivity reactions, including anaphylaxis, angioedema, and Stevens-Johnson Syndrome [see Warnings and Precautions (5.3)]
- •
- Hemolytic anemia in patients with and without G6PD deficiency [see Warnings and Precautions (5.9)]
- •
- Impairment of liver function (e.g. with cholestasis and jaundice), as well as hepatitis, which may progress to liver failure.
- •
- Porphyria cutanea tarda, photosensitivity reactions and allergic vasculitis
- •
- Leukopenia, agranulocytosis, aplastic anemia, and pancytopenia
- •
- Thrombocytopenia (including severe cases with platelet count less than 10,000/mcL) and thrombocytopenic purpura
- •
- Hepatic porphyria reactions and disulfiram-like reactions
- •
- Hyponatremia and syndrome of inappropriate antidiuretic hormone secretion (SIADH), most often in patients who are on other medications or who have medical conditions known to cause hyponatremia or increase release of antidiuretic hormone
-
7 DRUG INTERACTIONS
7.1 Strong CYP2C8 Inhibitors
Pioglitazone
An inhibitor of CYP2C8 (e.g., gemfibrozil) significantly increases the exposure (area under the serum concentration-time curve or AUC) and half-life (t½) of pioglitazone. Therefore, the maximum recommended dose of pioglitazone is 15 mg daily if used in combination with gemfibrozil or other strong CYP2C8 inhibitors. Since the minimum dose of pioglitazone in DUETACT exceeds 15 mg, patients taking concomitant strong CYP2C8 inhibitors should switch to individual components of DUETACT, unless the prescribing health care provider determines that the benefit of DUETACT clearly outweighs the risk of increased pioglitazone exposure [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
7.2 CYP2C8 Inducers
Pioglitazone
An inducer of CYP2C8 (e.g., rifampin) may significantly decrease the exposure (AUC) of pioglitazone. Therefore, if an inducer of CYP2C8 is started or stopped during treatment with pioglitazone, changes in diabetes treatment may be needed based on clinical response without exceeding the maximum recommended daily dose of 45 mg for pioglitazone [see Clinical Pharmacology (12.3)].
7.3 Topiramate
Pioglitazone
A decrease in the exposure of pioglitazone and its active metabolites were noted with concomitant administration of pioglitazone and topiramate [see Clinical Pharmacology (12.3)]. The clinical relevance of this decrease is unknown; however, when DUETACT and topiramate are used concomitantly, monitor patients for adequate glycemic control.
7.4 Drugs Affecting Glucose Metabolism
Glimepiride
A number of medications affect glucose metabolism and may require DUETACT dose adjustment and particularly close monitoring for hypoglycemia or worsening glycemic control.
The following are examples of medications that may increase the glucose-lowering effect of sulfonylureas including glimepiride, a component of DUETACT, increasing the susceptibility to and/or intensity of hypoglycemia: oral anti-diabetic medications, pramlintide acetate, insulin, angiotensin converting enzyme (ACE) inhibitors, H2 receptor antagonists, fibrates, propoxyphene, pentoxifylline, somatostatin analogs, anabolic steroids and androgens, cyclophosphamide, phenyramidol, guanethidine, fluconazole, sulfinpyrazone, tetracyclines, clarithromycin, disopyramide, quinolones, and those drugs that are highly protein-bound, such as fluoxetine, nonsteroidal anti-inflammatory drugs, salicylates, sulfonamides, chloramphenicol, coumarins, probenecid and monoamine oxidase inhibitors. When these medications are administered to a patient receiving DUETACT, monitor the patient closely for hypoglycemia. When these medications are withdrawn from a patient receiving DUETACT, monitor the patient closely for worsening glycemic control.
The following are examples of medications that may reduce the glucose-lowering effect of sulfonylureas including glimepiride, leading to worsening glycemic control: danazol, glucagon, somatropin, protease inhibitors, atypical antipsychotic medications (e.g., olanzapine and clozapine), barbiturates, diazoxide, laxatives, rifampin, thiazides and other diuretics, corticosteroids, phenothiazines, thyroid hormones, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics (e.g., epinephrine, albuterol, terbutaline), and isoniazid. When these medications are administered to a patient receiving DUETACT, monitor the patient closely for worsening glycemic control. When these medications are withdrawn from a patient receiving DUETACT, monitor the patient closely for hypoglycemia.
Beta-blockers, clonidine, and reserpine may lead to either potentiation or weakening of DUETACT's glucose-lowering effect.
Both acute and chronic alcohol intake may potentiate or weaken the glucose-lowering action of DUETACT in an unpredictable fashion.
The signs of hypoglycemia may be reduced or absent in patients taking sympatholytic drugs such as beta-blockers, clonidine, guanethidine, and reserpine.
7.5 Miconazole
Glimepiride
A potential interaction between oral miconazole and sulfonylureas leading to severe hypoglycemia has been reported. Whether this interaction also occurs with other dosage forms of miconazole is not known.
7.6 CYP2C9 Interactions
Glimepiride
There may be an interaction between glimepiride and inhibitors (e.g., fluconazole) and inducers (e.g., rifampin) of CYP2C9. Fluconazole may inhibit the metabolism of glimepiride, causing increased plasma concentrations of glimepiride which may lead to hypoglycemia. Rifampin may induce the metabolism of glimepiride, causing decreased plasma concentrations of glimepiride which may lead to worsening glycemic control.
7.7 Concomitant Administration of Colesevelam
Glimepiride
Colesevelam can reduce the maximum plasma concentrations and total exposure of glimepiride when the two are coadministered. However, absorption is not reduced when glimepiride is administered four hours prior to colesevelam. Therefore, DUETACT should be administered at least four hours prior to colesevelam [see Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Limited data with DUETACT or pioglitazone in pregnant women are not sufficient to determine a drug-associated risk for major birth defects or miscarriage. There are clinical considerations related to fetal and neonatal adverse reactions and drug discontinuation if glimepiride is used during pregnancy. There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy [see Clinical Considerations].
No adverse developmental effects were observed when pioglitazone was administered to pregnant rats and rabbits during organogenesis at exposures up to 5- and 35- times the 45 mg clinical dose, respectively, based on the body surface area. Administration of glimepiride to pregnant rats and rabbits during organogenesis induced maternal hypoglycemia and also increased fetal mortality at doses 50 (rats) and 0.1-times (rabbits) the 8 mg clinical dose, respectively, based on body surface area [see Data].
The estimated background risk of major birth defects is 6-10% in women with pre-gestational diabetes with a HbA1c >7 and has been reported to be as high as 20-25% in women with a HbA1c >10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20% respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, still birth and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, still birth, and macrosomia related morbidity.
Fetal/Neonatal Adverse Reaction
Neonates of women with gestational diabetes, who are treated with sulfonylureas during pregnancy, may be at increased risk for neonatal intensive care unit admission, and may develop respiratory distress, hypoglycemia, birth injury, and be large for gestational age. Prolonged severe hypoglycemia, lasting 4-10 days, has been reported in neonates born to mothers receiving a sulfonylurea at the time of delivery and has been reported with the use of agents with a prolonged half-life. Observe newborns for symptoms of hypoglycemia and respiratory distress and manage accordingly.
Dose adjustments during pregnancy and the postpartum period
Due to reports of prolonged severe hypoglycemia in neonates born to mothers receiving a sulfonylurea at the time of delivery, DUETACT should be discontinued at least two weeks before expected delivery [see Fetal/Neonatal Adverse Reaction].
Data
Animal Data
Pioglitazone and Glimepiride
Animal reproduction studies were not conducted with the combined products in DUETACT. The following data are based on studies conducted with the individual components of DUETACT.
Pioglitazone
Pioglitazone administered to pregnant rats during organogenesis did not cause adverse developmental effects at a dose of 20 mg/kg (~5-times the 45 mg clinical dose), but delayed parturition and reduced embryofetal viability at 40 and 80 mg/kg, or ≥9-times the 45 mg clinical dose, by body surface area. In pregnant rabbits administered pioglitazone during organogenesis, no adverse developmental effects were observed at 80 mg/kg (~35-times the 45 mg clinical dose), but reduced embryofetal viability at 160 mg/kg, or ~69-times the 45 mg clinical dose, by body surface area. When pregnant rats received pioglitazone during late gestation and lactation, delayed postnatal development, attributed to decreased body weight occurred in offspring at maternal doses of 10 mg/kg and above or ≥2 times the 45 mg clinical dose, by body surface area.
Glimepiride
Fetal deaths occurred in rats and rabbits administered glimepiride during the period of organogenesis at doses 50-times (rats) and 0.1-times (rabbits) the 8 mg clinical dose, based on body surface area. This fetotoxicity, observed only at doses inducing maternal hypoglycemia, is believed to be directly related to the pharmacologic (hypoglycemic) action of glimepiride and has been similarly noted with other sulfonylureas.
8.2 Lactation
Risk Summary
There is no information regarding the presence of pioglitazone or glimepiride in human milk, the effects on the breastfed infant, or the effects on milk production. Pioglitazone and glimepiride are present in rat milk; however, due to species-specific differences in lactation physiology, animal data may not reliably predict drug levels in human milk [see Data].
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for DUETACT and any potential adverse effects on the breastfed infant from DUETACT or from the underlying maternal condition.
Data
During pre- and postnatal studies in rats, glimepiride was present in lactational milk and in serum of nursing rat pups. Offspring exposed to high levels of glimepiride during lactation developed skeletal abnormalities (shortening, thickening and bending of the humerus) during the postnatal period.
8.3 Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as therapy with pioglitazone, like other thiazolidinediones, may result in ovulation in some anovulatory women.
8.4 Pediatric Use
Safety and effectiveness of DUETACT in pediatric patients have not been established.
DUETACT is not recommended for use in pediatric patients based on adverse effects observed in adults, including fluid retention and congestive heart failure, fractures, and urinary bladder tumors [see Warnings and Precautions (5.1, 5.6, 5.7, 5.8)].
Glimepiride
The pharmacokinetics, efficacy and safety of glimepiride have been evaluated in pediatric patients with type 2 diabetes as described below. Glimepiride is not recommended in pediatric patients because of its adverse effects on body weight and hypoglycemia.
The pharmacokinetics of a 1 mg single dose of glimepiride was evaluated in 30 patients with type 2 diabetes (male = 7; female = 23) between ages 10 and 17 years. The mean (±SD) AUC (0-last) (339±203 ng•hr/mL), Cmax (102±48 ng/mL) and t1/2 (3.1±1.7 hours) for glimepiride were comparable to historical data from adults (AUC (0-last) 315±96 ng•hr/mL, Cmax 103±34 ng/mL and t1/2 5.3±4.1 hours).
The safety and efficacy of glimepiride in pediatric patients was evaluated in a single-blind, 24-week trial that randomized 272 patients (8 to 17 years of age) with type 2 diabetes to glimepiride (n=135) or metformin (n=137). Both treatment-naïve patients (those treated with only diet and exercise for at least two weeks prior to randomization) and previously treated patients (those previously treated or currently treated with other oral antidiabetic medications for at least three months) were eligible to participate. Patients who were receiving oral antidiabetic agents at the time of study entry discontinued these medications before randomization without a washout period. Glimepiride was initiated at 1 mg, and then titrated up to 2, 4 or 8 mg (mean last dose 4 mg) through Week 12, targeting a self monitored fasting fingerstick blood glucose <126 mg/dL. Metformin was initiated at 500 mg twice daily and titrated at Week 12 up to 1000 mg twice daily (mean last dose 1365 mg).
After 24 weeks, the overall mean treatment difference in HbA1c between glimepiride and metformin was 0.2%, favoring metformin (95% confidence interval -0.3% to +0.6%).
Based on these results, the trial did not meet its primary objective of showing a similar reduction in HbA1c with glimepiride compared to metformin.
The profile of adverse reactions in pediatric patients treated with glimepiride was similar to that observed in adults.
Hypoglycemic events documented by blood glucose values <36 mg/dL were observed in 4% of pediatric patients treated with glimepiride and in 1% of pediatric patients treated with metformin. One patient in each treatment group experienced a severe hypoglycemic episode (severity was determined by the investigator based on observed signs and symptoms).
8.5 Geriatric Use
To minimize the risk of hypoglycemia, the initial dosing, dose increments, and maintenance dosage of DUETACT should be conservative. During initiation of DUETACT therapy and any subsequent dose adjustments, geriatric patients should be observed carefully for hypoglycemia.
Pioglitazone
A total of 92 patients (15.2%) treated with pioglitazone in the three pooled 16- to 26-week double-blind, placebo-controlled, monotherapy trials were ≥65 years old and two patients (0.3%) were ≥75 years old. In the two pooled 16- to 24-week add-on to sulfonylurea trials, 201 patients (18.7%) treated with pioglitazone were ≥65 years old and 19 (1.8%) were ≥75 years old. In the two pooled 16- to 24-week add-on to metformin trials, 155 patients (15.5%) treated with pioglitazone were ≥65 years old and 19 (1.9%) were ≥75 years old. In the two pooled 16- to 24-week add-on to insulin trials, 272 patients (25.4%) treated with pioglitazone were ≥65 years old and 22 (2.1%) were ≥75 years old.
In PROactive, 1068 patients (41.0%) treated with pioglitazone were ≥65 years old and 42 (1.6%) were ≥75 years old.
In pharmacokinetic studies with pioglitazone, no significant differences were observed in pharmacokinetic parameters between elderly and younger patients [see Clinical Pharmacology (12.3)].
Although clinical experiences have not identified differences in effectiveness and safety between the elderly (≥65 years) and younger patients, these conclusions are limited by small sample sizes for patients ≥75 years old.
Glimepiride
In clinical trials of glimepiride, 1053 of 3491 patients (30%) were ≥65 years of age. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
There were no significant differences in glimepiride pharmacokinetics between patients with type 2 diabetes ≤65 years (n=49) and those >65 years (n=42) [see Clinical Pharmacology (12.3)].
Glimepiride is substantially excreted by the kidney. Elderly patients are more likely to have renal impairment. In addition, hypoglycemia may be difficult to recognize in the elderly [see Dosage and Administration (2.1) and Warnings and Precautions (5.2)]. Use caution when initiating DUETACT and increasing the dose of DUETACT in this patient population.
8.6 Renal Impairment
To minimize the risk of hypoglycemia, the initial dosing, dose increments and maintenance dosage of DUETACT should be conservative. During initiation of DUETACT therapy and any subsequent dose adjustments, these patients should be observed carefully for hypoglycemia.
A multiple-dose titration study was conducted in 16 patients with type 2 diabetes and renal impairment using doses ranging from 1 mg to 8 mg daily for three months. Baseline creatinine clearance ranged from 10 to 60 mL/min. The pharmacokinetics of glimepiride were evaluated in the multiple-dose titration study and the results were consistent with those observed in patients enrolled in a single-dose study. In both studies, the relative total clearance of glimepiride increased when kidney function was impaired. Both studies also demonstrated that the elimination of the two major metabolites was reduced in patients with renal impairment [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Pioglitazone
During controlled clinical trials, one case of overdose with pioglitazone was reported. A male patient took 120 mg per day for four days, then 180 mg per day for seven days. The patient denied any clinical symptoms during this period.
In the event of overdosage, appropriate supportive treatment should be initiated according to the patient's clinical signs and symptoms.
Glimepiride
An overdosage of glimepiride, as with other sulfonylureas, can produce severe hypoglycemia. Mild episodes of hypoglycemia can be treated with oral glucose. Severe hypoglycemic reactions constitute medical emergencies requiring immediate treatment. Severe hypoglycemia with coma, seizure, or neurological impairment can be treated with glucagon or intravenous glucose. Continued observation and additional carbohydrate intake may be necessary because hypoglycemia may recur after apparent clinical recovery [see Warnings and Precautions (5.2)].
-
11 DESCRIPTION
DUETACT tablets are a thiazolidinedione and a sulfonylurea combination product that contains two oral antihyperglycemic agents: pioglitazone and glimepiride. The concomitant use of pioglitazone and a sulfonylurea, the class of drugs that includes glimepiride, has been previously approved based on clinical trials in patients with type 2 diabetes inadequately controlled on a sulfonylurea. Additional efficacy and safety information about pioglitazone and glimepiride monotherapies may be found in the prescribing information for each individual drug.
Pioglitazone is an oral antidiabetic medication.
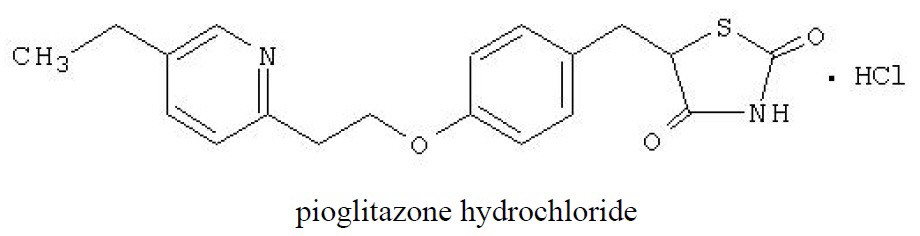
Pioglitazone [(±)-5-[[4-[2-(5-ethyl-2-pyridinyl)ethoxy]phenyl]methyl]-2,4-] thiazolidinedione monohydrochloride contains one asymmetric carbon, and the compound is synthesized and used as the racemic mixture. The two enantiomers of pioglitazone interconvert in vivo. No differences were found in the pharmacologic activity between the two enantiomers. The structural formula is as shown:
Pioglitazone hydrochloride is an odorless, white crystalline powder that has a molecular formula of C19H20N2O3S•HCl and a molecular weight of 392.90 daltons. It is soluble in N,N‑dimethylformamide, slightly soluble in anhydrous ethanol, very slightly soluble in acetone and acetonitrile, practically insoluble in water, and insoluble in ether.
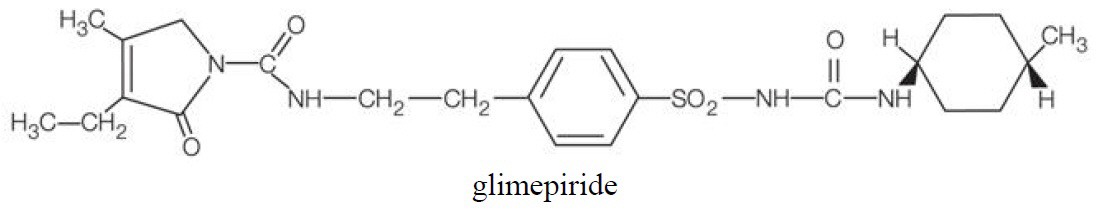
Glimepiride is an oral sulfonylurea chemically identified as 1-[[p-[2-(3-ethyl-4-methyl-2-oxo-3-pyrroline-1-carboxamido)ethyl]phenyl]sulfonyl]-3-(trans-4-methylcyclohexyl)-urea (C24H34N4O5S) with a molecular weight of 490.62. Glimepiride is a white to yellowish-white, crystalline, odorless to practically odorless powder and is practically insoluble in water. The structural formula is:
DUETACT is available as a tablet for oral administration containing 30 mg pioglitazone (as the base) with 2 mg glimepiride (30 mg/2 mg) or 30 mg pioglitazone (as the base) with 4 mg glimepiride (30 mg/4 mg) formulated with the following excipients: croscarmellose sodium NF, lactose monohydrate NF, magnesium stearate NF, hydroxypropyl cellulose NF, polysorbate 80 NF, and microcrystalline cellulose NF.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
DUETACT combines 2 antihyperglycemic agents with different mechanisms of action to improve glycemic control in patients with type 2 diabetes: pioglitazone, a member of the thiazolidinedione class, and glimepiride, a member of the sulfonylurea class. Thiazolidinediones are insulin-sensitizing agents that act primarily by enhancing peripheral glucose utilization, whereas sulfonylureas are insulin secretagogues that act primarily by stimulating release of insulin from functioning pancreatic beta cells.
Pioglitazone
Pioglitazone is a thiazolidinedione that depends on the presence of insulin for its mechanism of action. Pioglitazone decreases insulin resistance in the periphery and in the liver resulting in increased insulin-dependent glucose disposal and decreased hepatic glucose output. Pioglitazone is not an insulin secretagogue. Pioglitazone is an agonist for peroxisome proliferator-activated receptor-gamma (PPARγ). PPAR receptors are found in tissues important for insulin action such as adipose tissue, skeletal muscle, and liver. Activation of PPARγ nuclear receptors modulates the transcription of a number of insulin responsive genes involved in the control of glucose and lipid metabolism.
In animal models of diabetes, pioglitazone reduces the hyperglycemia, hyperinsulinemia, and hypertriglyceridemia characteristic of insulin-resistant states such as type 2 diabetes. The metabolic changes produced by pioglitazone result in increased responsiveness of insulin-dependent tissues and are observed in numerous animal models of insulin resistance.
Because pioglitazone enhances the effects of circulating insulin (by decreasing insulin resistance), it does not lower blood glucose in animal models that lack endogenous insulin.
Glimepiride
Glimepiride primarily lowers blood glucose by stimulating the release of insulin from pancreatic beta cells. Sulfonylureas bind to the sulfonylurea receptor in the pancreatic beta cell plasma membrane, leading to closure of the ATP-sensitive potassium channel, thereby stimulating the release of insulin.
12.2 Pharmacodynamics
Pioglitazone
Clinical studies demonstrate that pioglitazone improves insulin sensitivity in insulin-resistant patients. Pioglitazone enhances cellular responsiveness to insulin, increases insulin-dependent glucose disposal and improves hepatic sensitivity to insulin. In patients with type 2 diabetes, the decreased insulin resistance produced by pioglitazone results in lower plasma glucose concentrations, lower plasma insulin concentrations, and lower HbA1c values. In controlled clinical trials, pioglitazone had an additive effect on glycemic control when used in combination with a sulfonylurea, metformin, or insulin [see Clinical Studies (14)].
Patients with lipid abnormalities were included in clinical trials with pioglitazone. Overall, patients treated with pioglitazone had mean decreases in serum triglycerides, mean increases in HDL cholesterol, and no consistent mean changes in LDL and total cholesterol. There is no conclusive evidence of macrovascular benefit with pioglitazone [see Warnings and Precautions (5.11) and Adverse Reactions (6.1)].
In a 26-week, placebo-controlled, dose-ranging monotherapy study, mean serum triglycerides decreased in the 15 mg, 30 mg, and 45 mg pioglitazone dose groups compared to a mean increase in the placebo group. Mean HDL cholesterol increased to a greater extent in patients treated with pioglitazone than in the placebo-treated patients. There were no consistent differences for LDL and total cholesterol in patients treated with pioglitazone compared to placebo (Table 12).
*Adjusted for baseline, pooled center, and pooled center by treatment interaction
†p <0.05 versus placeboTable 12. Lipids in a 26-Week Placebo-Controlled Monotherapy Dose-Ranging Study
Placebo
Pioglitazone
15 mg
Once
DailyPioglitazone
30 mg
Once
DailyPioglitazone
45 mg
Once
DailyTriglycerides (mg/dL)
N=79
N=79
N=84
N=77
Baseline (mean)
263
284
261
260
Percent change from baseline (adjusted mean*)
4.8%
-9.0%†
-9.6%†
-9.3%†
HDL Cholesterol (mg/dL)
N=79
N=79
N=83
N=77
Baseline (mean)
42
40
41
41
Percent change from baseline (adjusted mean*)
8.1%
14.1%†
12.2%
19.1%†
LDL Cholesterol (mg/dL)
N=65
N=63
N=74
N=62
Baseline (mean)
139
132
136
127
Percent change from baseline (adjusted mean*)
4.8%
7.2%
5.2%
6.0%
Total Cholesterol (mg/dL)
N=79
N=79
N=84
N=77
Baseline (mean)
225
220
223
214
Percent change from baseline (adjusted mean*)
4.4%
4.6%
3.3%
6.4%
In the two other monotherapy studies (16 weeks and 24 weeks) and in combination therapy studies with sulfonylurea (16 weeks and 24 weeks), metformin (16 weeks and 24 weeks) or insulin (16 weeks and 24 weeks), the results were generally consistent with the data above.
Glimepiride
In healthy subjects, the time to reach maximal effect (minimum blood glucose concentrations) was approximately by two to three hours after single oral doses of glimepiride. The effects of HbA1C, fasting plasma glucose, and post-prandial glucose have been assessed in clinical trials.
12.3 Pharmacokinetics
Absorption and Bioavailability:
DUETACT
Bioequivalence studies were conducted following a single dose of the DUETACT 30 mg/2 mg and 30 mg/4 mg tablets and concomitant administration of pioglitazone (30 mg) and glimepiride (2 mg or 4 mg) under fasting conditions in healthy subjects.
Based on the area under the curve (AUC) and maximum concentration (Cmax) of both pioglitazone and glimepiride, DUETACT 30 mg/2 mg and 30 mg/4 mg were bioequivalent to pioglitazone 30 mg concomitantly administered with glimepiride (2 mg or 4 mg, respectively).
Food did not change the systemic exposures of glimepiride or pioglitazone following administration of DUETACT. The presence of food did not significantly alter the time to peak serum concentration (Tmax) of glimepiride or pioglitazone and Cmax of pioglitazone. However, for glimepiride, there was a 22% increase in Cmax when DUETACT was administered with food.
Pioglitazone
Following once-daily administration of pioglitazone, steady-state serum concentrations of both pioglitazone and its major active metabolites, M-III (keto derivative of pioglitazone) and M-IV (hydroxyl derivative of pioglitazone), are achieved within seven days. At steady-state, M-III and M-IV reach serum concentrations equal to or greater than that of pioglitazone. At steady-state, in both healthy volunteers and patients with type 2 diabetes, pioglitazone comprises approximately 30% to 50% of the peak total pioglitazone serum concentrations (pioglitazone plus active metabolites) and 20% to 25% of the total AUC.
Cmax, AUC, and trough serum concentrations (Cmin) for pioglitazone and M-III and M-IV, increased proportionally with administered doses of 15 mg and 30 mg per day.
Following oral administration of pioglitazone, Tmax of pioglitazone was within two hours. Food delays Tmax to three to four hours but does not alter the extent of absorption (AUC).
Glimepiride
Studies with single oral doses of glimepiride in healthy subjects and with multiple oral doses in patients with type 2 diabetes showed peak drug concentrations (Cmax) two to three hours post-dose. When glimepiride was given with meals, the mean Cmax and AUC were decreased by 8% and 9%, respectively.
Glimepiride does not accumulate in serum following multiple dosing. The pharmacokinetics of glimepiride does not differ between healthy subjects and patients with type 2 diabetes. Clearance (CL/F) of glimepiride after oral administration does not change over the 1 mg to 8 mg dose range, indicating linear pharmacokinetics.
In healthy subjects, the intra- and inter-individual variabilities of glimepiride pharmacokinetic parameters were 15% to 23% and 24% to 29%, respectively.
Distribution
Pioglitazone
The mean apparent volume of distribution (Vd/F) of pioglitazone following single-dose administration is 0.63 ± 0.41 (mean ± SD) L/kg of body weight. Pioglitazone is extensively protein bound (>99%) in human serum, principally to serum albumin. Pioglitazone also binds to other serum proteins, but with lower affinity. M-III and M-IV are also extensively bound (>98%) to serum albumin.
Glimepiride
After intravenous (IV) dosing in healthy subjects, Vd/F was 8.8 L (113 mL/kg), and the total body clearance (CL) was 47.8 mL/min. Protein binding was greater than 99.5%.
Metabolism
Pioglitazone
Pioglitazone is extensively metabolized by hydroxylation and oxidation; the metabolites also partly convert to glucuronide or sulfate conjugates. Metabolites M-III and M-IV are the major circulating active metabolites in humans.
In vitro data demonstrate that multiple CYP isoforms are involved in the metabolism of pioglitazone which include CYP2C8 and, to a lesser degree, CYP3A4 with additional contributions from a variety of other isoforms including the mainly extrahepatic CYP1A1. In vivo study of pioglitazone in combination with gemfibrozil, a strong CYP2C8 inhibitor, showed that pioglitazone is a CYP2C8 substrate [see Dosage and Administration (2.3) and Drug Interactions (7.1)]. Urinary 6ß-hydroxycortisol/cortisol ratios measured in patients treated with pioglitazone showed that pioglitazone is not a strong CYP3A4 enzyme inducer.
Glimepiride
Glimepiride is completely metabolized by oxidative biotransformation after either an IV or oral dose. The major metabolites are the cyclohexyl hydroxy methyl derivative (M1) and the carboxyl derivative (M2). CYP2C9 is involved in the biotransformation of glimepiride to M1. M1 is further metabolized to M2 by one or several cytosolic enzymes. In animals, M1 possesses about one-third of the pharmacological activity of glimepiride, but it is unclear whether M1 results in clinically meaningful effects on blood glucose in humans. M2 is inactive.
Excretion and Elimination
Pioglitazone
Following oral administration, approximately 15% to 30% of the pioglitazone dose is recovered in the urine. Renal elimination of pioglitazone is negligible and the drug is excreted primarily as metabolites and their conjugates. It is presumed that most of the oral dose is excreted into the bile either unchanged or as metabolites and eliminated in the feces.
The mean serum half-life (t1/2) of pioglitazone and its metabolites (M-III and M-IV) range from three to seven hours and 16 to 24 hours, respectively. Pioglitazone has an apparent clearance, CL/F, calculated to be five to seven L/hr.
Glimepiride
When 14C-glimepiride was given orally to three healthy male subjects, approximately 60% of the total radioactivity was recovered in the urine in seven days. M1 and M2 accounted for 80% to 90% of the radioactivity recovered in the urine. The ratio of M1 to M2 in the urine was approximately 3:2 in two subjects and 4:1 in one subject. Approximately 40% of the total radioactivity was recovered in feces. M1 and M2 accounted for approximately 70% (ratio of M1 to M2 was 1:3) of the radioactivity recovered in feces. No parent drug was recovered from urine or feces. After IV dosing in patients, no significant biliary excretion of glimepiride or its M1 metabolite was observed.
Renal Impairment
Pioglitazone
The serum elimination half-life of pioglitazone, M-III, and M-IV remains unchanged in patients with moderate [creatinine clearance (CLcr) 30 to 50 mL/min] and severe (CLcr <30 mL/min) renal impairment when compared to subjects with normal renal function. Therefore, no dose adjustment in patients with renal impairment is required.
Glimepiride
In a single-dose, open-label study glimepiride 3 mg was administered to patients with mild, moderate and severe renal impairment as estimated by CLcr: Group I consisted of five patients with mild renal impairment (CLcr >50 mL/min), Group II consisted of 3 patients with moderate renal impairment (CLcr = 20 to 50 mL/min) and Group III consisted of seven patients with severe renal impairment (CLcr <20 mL/min). Although, glimepiride serum concentrations decreased with decreasing renal function, Group III had a 2.3-fold higher mean AUC for M1 and an 8.6-fold higher mean AUC for M2 compared to corresponding mean AUCs in Group I. The t½ for glimepiride did not change, while the t½ for M1 and M2 increased as renal function decreased. Mean urinary excretion of M1 plus M2 as a percentage of dose decreased from 44.4% for Group I to 21.9% for Group II and 9.3% for Group III.
Hepatic Impairment
Pioglitazone
Compared with healthy controls, subjects with impaired hepatic function (Child-Turcotte-Pugh Grade B/C) have an approximate 45% reduction in pioglitazone and total pioglitazone (pioglitazone, M-III, and M-IV) mean Cmax but no change in the mean AUC values. Therefore, no dose adjustment in patients with hepatic impairment is required.
There are postmarketing reports of liver failure with pioglitazone and clinical trials have generally excluded patients with serum ALT >2.5 times the upper limit of the reference range. Use DUETACT with caution in patients with liver disease [see Warnings and Precautions (5.5)].
Glimepiride
It is unknown whether there is an effect of hepatic impairment on glimepiride pharmacokinetics because the pharmacokinetics of glimepiride has not been adequately evaluated in patients with hepatic impairment.
Geriatric Patients
Pioglitazone
In healthy elderly subjects, Cmax of pioglitazone was not significantly different, but AUC values were approximately 21% higher than those achieved in younger subjects. The mean t½ of pioglitazone was also prolonged in elderly subjects (about 10 hours) as compared to younger subjects (about seven hours). These changes were not of a magnitude that would be considered clinically relevant.
Glimepiride
A comparison of glimepiride pharmacokinetics in patients with type 2 diabetes ≤65 years and those >65 years was evaluated in a multiple-dose study using 6 mg daily dose. There were no significant differences in glimepiride pharmacokinetics between the two age groups. The mean AUC at steady state for the older patients was approximately 13% lower than that for the younger patients; the mean weight-adjusted clearance for the older patients was approximately 11% higher than that for the younger patients.
Pediatric Patients
No pharmacokinetic studies of DUETACT were performed in pediatric patients.
Pioglitazone
Safety and efficacy of pioglitazone in pediatric patients have not been established. DUETACT is not recommended for use in pediatric patients [see Use in Specific Populations (8.4)].
Gender
Pioglitazone
The mean Cmax and AUC values of pioglitazone were increased 20% to 60% in women compared to men. In controlled clinical trials, HbA1c decreases from baseline were generally greater for females than for males (average mean difference in HbA1c 0.5%). Because therapy should be individualized for each patient to achieve glycemic control, no dose adjustment is recommended based on gender alone.
Glimepiride
There were no differences between males and females in the pharmacokinetics of glimepiride when adjustment was made for differences in body weight.
Ethnicity
Pioglitazone
Pharmacokinetic data among various ethnic groups are not available.
Glimepiride
No studies have been conducted to assess the effects of race on glimepiride pharmacokinetics but in placebo-controlled trials of glimepiride in patients with type 2 diabetes, the reduction in HbA1c was comparable in Caucasians (n=536), blacks (n=63), and Hispanics (n=63).
Obese Patients
The pharmacokinetics of glimepiride and its metabolites were measured in a single-dose study involving 28 patients with type 2 diabetes who either had normal body weight or were morbidly obese. While the Tmax, CL/F, and Vd/F of glimepiride in the morbidly obese patients were similar to those in the normal weight group, the morbidly obese had lower Cmax and AUC than those of normal body weight. The mean Cmax, AUC0-24, AUC0-∞ values of glimepiride in normal vs. morbidly obese patients were 547 ± 218 ng/mL vs. 410 ± 124 ng/mL, 3210 ± 1030 hours∙ng/mL vs. 2820 ± 1110 hours∙ng/mL and 4000 ± 1320 hours∙ng/mL versus 3280 ± 1360 hours∙ng/mL, respectively.
Drug-Drug Interactions
Coadministration of pioglitazone (45 mg) and a sulfonylurea (5 mg glipizide) administered orally once daily for seven days did not alter the steady-state pharmacokinetics of glipizide. Glimepiride and glipizide have similar metabolic pathways and are mediated by CYP2C9; therefore, drug-drug interaction between pioglitazone and glimepiride is considered unlikely. Specific pharmacokinetic drug interaction studies with DUETACT have not been performed, although such studies have been conducted with the individual pioglitazone and glimepiride components.
Pioglitazone
*Daily for 7 days unless otherwise noted
†% change (with/without coadministered drug and no change = 0%); symbols of ↑ and ↓ indicate the exposure increase and decrease, respectively
‡Pioglitazone had no clinically significant effect on prothrombin timeTable 13. Effect of Pioglitazone Coadministration on Systemic Exposure of Other Drugs
Coadministered Drug
Pioglitazone
Dosage
Regimen (mg)*Name and Dose Regimens
Change
in AUC†Change
in Cmax†45 mg
(N=12)Warfarin‡
Daily loading then maintenance doses
based PT and INR values
Quick's Value = 35 ± 5%R-Warfarin
↓3%
R-Warfarin
↓2%
S-Warfarin
↓1%
S-Warfarin
↑1%
45 mg
(N=12)Digoxin
0.250 mg twice daily (loading dose) then
0.250 mg daily (maintenance dose, 7 days)↑15%
↑17%
45 mg daily
for 21 days
(N=35)Oral Contraceptive
[Ethinyl Estradiol (EE) 0.035 mg plus
Norethindrone (NE) 1 mg] for 21 daysEE
↓11%
EE
↓13%
NE
↑3%
NE
↓7%
45 mg
(N=23)Fexofenadine
60 mg twice daily for 7 days
↑30%
↑37%
45 mg
(N=14)Glipizide
5 mg daily for 7 days
↓3%
↓8%
45 mg daily
for 8 days
(N=16)Metformin
1000 mg single dose on Day 8
↓3%
↓5%
45 mg
(N=21)Midazolam
7.5 mg single dose on Day 15
↓26%
↓26%
45 mg
(N=24)Ranitidine
150 mg twice daily for 7 days
↑1%
↓1%
45 mg daily
for 4 days
(N=24)Nifedipine ER
30 mg daily for 4 days
↓13%
↓17%
45 mg
(N=25)Atorvastatin Ca
80 mg daily for 7 days
↓14%
↓23%
45 mg
(N=22)Theophylline
400 mg twice daily for 7 days
↑2%
↑5%
Table 14. Effect of Coadministered Drugs on Pioglitazone Systemic Exposure Coadministered Drug and Dosage Regimen Pioglitazone Dose Regimen
(mg)*Change in AUC† Change in Cmax† - *
- Daily for 7 days unless otherwise noted
- †
- Mean ratio (with/without coadministered drug and no change = 1-fold) % change (with/without coadministered drug and no change = 0%); symbols of ↑ and ↓ indicate the exposure increase and decrease, respectively
- ‡
- The half-life of pioglitazone increased from 8.3 hours to 22.7 hours in the presence of gemfibrozil [see Dosage and Administration (2.3) and Drug Interactions (7)]
- §
- Indicates duration of concomitant administration with highest twice-daily dose of topiramate from Day 14 onwards over the 22 days of study
- ¶
- Additional decrease in active metabolites; 60% for M-III and 16% for M-IV
Gemfibrozil 600 mg
twice daily for 2 days
(N=12)15 mg single dose ↑3.2-fold‡ ↑6% Ketoconazole 200 mg
twice daily for 7 days
(N=28)45 mg ↑34% ↑14% Rifampin 600 mg
daily for 5 days
(N=10)30 mg single dose ↓54% ↓5% Fexofenadine 60 mg
twice daily for 7 days
(N=23)45 mg ↑ 1% 0% Ranitidine 150 mg
twice daily for 4 days
(N=23)45 mg ↓ 13% ↓16% Nifedipine ER 30 mg
daily for 7 days
(N=23)45 mg ↑5% ↑4% Atorvastatin Ca 80 mg
daily for 7 days
(N = 24)45 mg ↓24% ↓31% Theophylline 400 mg
twice daily for 7 days
(N=22)45 mg ↓4% ↓2% Topiramate 96 mg
twice daily for 7 days§
(N=26)30 mg§ ↓15%¶ 0% Glimepiride
Aspirin
In a randomized, double-blind, two-period, crossover study, healthy subjects were given either placebo or aspirin 1 gram three times daily for a total treatment period of 5 days. On Day 4 of each study period, a single 1 mg dose of glimepiride was administered. The glimepiride doses were separated by a 14-day washout period. Coadministration of aspirin and glimepiride resulted in a 34% decrease in the mean glimepiride AUC and a 4% decrease in the mean glimepiride Cmax.
Cimetidine and Ranitidine
In a randomized, open-label, 3-way crossover study, healthy subjects received either a single 4 mg dose of glimepiride alone, glimepiride with ranitidine (150 mg twice daily for 4 days; glimepiride was administered on Day 3), or glimepiride with cimetidine (800 mg daily for 4 days; glimepiride was administered on Day 3). Coadministration of cimetidine or ranitidine with a single 4 mg oral dose of glimepiride did not significantly alter the absorption and disposition of glimepiride.
Propranolol
In a randomized, double-blind, two-period, crossover study, healthy subjects were given either placebo or propranolol 40 mg three times daily for a total treatment period of five days. On Day 4 or each study period, a single 2 mg dose of glimepiride was administered. The glimepiride doses were separated by a 14-day washout period. Concomitant administration of propranolol and glimepiride significantly increased glimepiride Cmax, AUC, and t1/2 by 23%, 22%, and 15%, respectively, and decreased glimepiride CL/F by 18%. The recovery of M1 and M2 from urine was not changed.
Warfarin
In an open-label, two-way, crossover study, healthy subjects received 4 mg of glimepiride daily for 10 days. Single 25 mg doses of warfarin were administered six days before starting glimepiride and on Day 4 of glimepiride administration. The concomitant administration of glimepiride did not alter the pharmacokinetics of R- and S-warfarin enantiomers. No changes were observed in warfarin plasma protein binding. Glimepiride resulted in a statistically significant decrease in the pharmacodynamic response to warfarin. The reductions in mean area under the prothrombin time (PT) curve and maximum PT values during glimepiride treatment were 3.3% and 9.9%, respectively, and are unlikely to be clinically relevant.
Colesevelam
Concomitant administration of colesevelam and glimepiride resulted in reductions in glimepiride AUC0-∞ and Cmax of 18% and 8%, respectively. When glimepiride was administered 4 hours prior to colesevelam, there was not significant change in glimepiride AUC0-∞ and Cmax, -6% and 3%, respectively [see Dosage and Administration (2.4) and Drug Interactions (7.7)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies have been conducted with DUETACT. The following data are based on findings in studies performed with pioglitazone or glimepiride individually.
Pioglitazone
A two-year carcinogenicity study was conducted in male and female rats at oral doses up to 63 mg/kg (approximately 14 times the maximum recommended human oral dose of 45 mg based on mg/m2). Drug-induced tumors were not observed in any organ except for the urinary bladder of male rats. Benign and/or malignant transitional cell neoplasms were observed in male rats at 4 mg/kg/day and above (approximately equal to the maximum recommended human oral dose based on mg/m2). Urinary calculi with subsequent irritation and hyperplasia were postulated as the mechanism for bladder tumors observed in male rats. A two-year mechanistic study in male rats utilizing dietary acidification to reduce calculi formation was completed in 2009. Dietary acidification decreased but did not abolish the hyperplastic changes in the bladder. The presence of calculi exacerbated the hyperplastic response to pioglitazone but was not considered the primary cause of the hyperplastic changes.
The relevance to humans of the bladder findings in the male rat cannot be excluded.
A two-year carcinogenicity study was also conducted in male and female mice at oral doses up to 100 mg/kg/day (approximately 11 times the maximum recommended human oral dose based on mg/m2). No drug-induced tumors were observed in any organ.
Pioglitazone hydrochloride was not mutagenic in a battery of genetic toxicology studies, including the Ames bacterial assay, a mammalian cell forward gene mutation assay (CHO/HPRT and AS52/XPRT), an in vitro cytogenetics assay using CHL cells, an unscheduled DNA synthesis assay, and an in vivo micronucleus assay.
No adverse effects upon fertility were observed in male and female rats at oral doses up to 40 mg/kg pioglitazone hydrochloride daily prior to and throughout mating and gestation (approximately nine times the maximum recommended human oral dose based on mg/m2).
Glimepiride
Studies in rats at doses of up to 5000 parts per million (ppm) in complete feed (approximately 340 times the maximum recommended human dose, based on surface area) for 30 months showed no evidence of carcinogenesis. In mice, administration of glimepiride for 24 months resulted in an increase in benign pancreatic adenoma formation that was dose-related and was thought to be the result of chronic pancreatic stimulation. No adenoma formation in mice was observed at a dose of 320 ppm in complete feed, or 46 − 54 mg/kg body weight/day. This is about 35 times the maximum human recommended dose of 8 mg once daily based on surface area.
Glimepiride was non-mutagenic in a battery of in vitro and in vivo mutagenicity studies (Ames test, somatic cell mutation, chromosomal aberration, unscheduled DNA synthesis and mouse micronucleus test).
There was no effect of glimepiride on male mouse fertility in animals exposed up to 2500 mg/kg body weight (>1,700 times the maximum recommended human dose based on surface area). Glimepiride had no effect on the fertility of male and female rats administered up to 4000 mg/kg body weight (approximately 4,000 times the maximum recommended human dose based on surface area).
13.2 Animal Toxicology and/or Pharmacology
Pioglitazone
Heart enlargement has been observed in mice (100 mg/kg), rats (4 mg/kg and above), and dogs (3 mg/kg) treated orally with the pioglitazone hydrochloride component of DUETACT (approximately 11, one, and two times the maximum recommended human oral dose for mice, rats, and dogs, respectively, based on mg/m2). In a one-year rat study, drug-related early death due to apparent heart dysfunction occurred at an oral dose of 160 mg/kg/day (approximately 35 times the maximum recommended human oral dose based on mg/m2). Heart enlargement was seen in a 13-week study in monkeys at oral doses of 8.9 mg/kg and above (approximately four times the maximum recommended human oral dose based on mg/m2), but not in a 52-week study at oral doses up to 32 mg/kg (approximately 13 times the maximum recommended human oral dose based on mg/m2).
-
14 CLINICAL STUDIES
There have been no clinical efficacy studies conducted with DUETACT. However, the efficacy and safety of the separate components have been previously established. The coadministration of pioglitazone and a sulfonylurea, including glimepiride, has been evaluated for efficacy and safety in two clinical studies. These clinical studies established an added benefit of pioglitazone in glycemic control of patients with inadequately controlled type 2 diabetes while on sulfonylurea therapy. Bioequivalence of DUETACT with coadministered pioglitazone and glimepiride tablets was demonstrated at the 30 mg/2 mg and 30 mg/4 mg dosage strengths [see Clinical Pharmacology (12.3)].
Two clinical trials were conducted with pioglitazone in combination with a sulfonylurea. Both studies included patients with type 2 diabetes on any dose of a sulfonylurea, either alone or in combination with another antidiabetic agent. All other antidiabetic agents were withdrawn at least three weeks prior to starting study treatment.
In the first study, 560 patients were randomized to receive 15 mg or 30 mg of pioglitazone or placebo once daily for 16 weeks in addition to their current sulfonylurea regimen. Treatment with pioglitazone as add-on to sulfonylurea produced statistically significant improvements in HbA1c and FGP at endpoint compared to placebo add-on to sulfonylurea (Table 15).
*Adjusted for baseline, pooled center, and pooled center by treatment interaction
†p ≤0.05 versus placebo + sulfonylureaTable 15. Glycemic Parameters in a 16-Week Placebo-Controlled, Add-on to Sulfonylurea Trial
Placebo
+ SulfonylureaPioglitazone 15 mg
+ SulfonylureaPioglitazone 30 mg
+ SulfonylureaTotal Population
HbA1c (%)
N=181
N=176
N=182
Baseline (mean)
9.9
10.0
9.9
Change from baseline (adjusted mean*)
0.1
-0.8
-1.2
Difference from placebo + sulfonylurea
(adjusted mean*) 95% Confidence Interval-0.9†
(-1.2, -0.6)-1.3†
(-1.6, -1.0)Fasting Plasma Glucose (mg/dL)
N=182
N=179
N=186
Baseline (mean)
236
247
239
Change from baseline (adjusted mean*)
6
-34
-52
Difference from placebo + sulfonylurea (adjusted mean*) 95% Confidence Interval
-39†
(-52, -27)-58†
(-70, -46)In the second trial, 702 patients were randomized to receive 30 mg or 45 mg of pioglitazone once daily for 24 weeks in addition to their current sulfonylurea regimen. The mean reduction from baseline at Week 24 in HbA1c was 1.6% for the 30 mg dose and 1.7% for the 45 mg dose (see Table 16). The mean reduction from baseline at Week 24 in FPG was 52 mg/dL for the 30 mg dose and 56 mg/dL for the 45 mg dose.
The therapeutic effect of pioglitazone in combination with sulfonylurea was observed in patients regardless of the sulfonylurea dose.
95% CI = 95% confidence interval
*Adjusted for baseline, pooled center, and pooled center by treatment interaction
Table 16. Glycemic Parameters in a 24-Week Add-on to Sulfonylurea TrialPioglitazone
30 mg
+ SulfonylureaPioglitazone
45 mg
+ SulfonylureaTotal Population
HbA1c (%)
N=340
N=332
Baseline (mean)
9.8
9.9
Change from baseline (adjusted mean*)
-1.6
-1.7
Difference from 30 mg daily pioglitazone + sulfonylurea (adjusted mean*) (95% CI)
-0.1
(-0.4, 0.1)Fasting Plasma Glucose (mg/dL)
N=338
N=329
Baseline (mean)
214
217
Change from baseline (adjusted mean*)
-52
-56
Difference from 30 mg daily pioglitazone + sulfonylurea (adjusted mean*) (95% CI)
-5
(-12, 3) -
16 HOW SUPPLIED/STORAGE AND HANDLING
DUETACT is available in 30 mg pioglitazone plus 2 mg glimepiride or 30 mg pioglitazone plus 4 mg glimepiride tablets as follows:
30 mg/2 mg tablet: white to off-white, round, convex tablets, debossed with 4833G on one side and 30/2 on the other, available in:
NDC 64764-302-30 Bottles of 30
NDC 64764-302-90 Bottles of 90
30 mg/4 mg tablet: white to off-white, round, convex tablets, debossed with 4833G on one side and 30/4 on the other, available in:
NDC 64764-304-30 Bottles of 30
NDC 64764-304-90 Bottles of 90
Storage
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Keep container tightly closed and protect from moisture and humidity.
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Medication Guide).
- •
- Inform patients that DUETACT is not recommended for patients with symptoms of heart failure.
- •
- Inform patients that patients with severe heart failure (NYHA Class III or IV) cannot start DUETACT as the risks exceed the benefits in such patients.
- •
- It is important to instruct patients to adhere to dietary instructions and to have blood glucose and glycosylated hemoglobin tested regularly. During periods of stress such as fever, trauma, infection, or surgery, medication requirements may change and patients should be reminded to seek medical advice promptly. Patients should also be informed of the potential risks and advantages of DUETACT and of alternative modes of therapy.
- •
- Tell patients to promptly report any sign of macroscopic hematuria or other symptoms such as dysuria or urinary urgency that develop or increase during treatment as these may be due to bladder cancer.
- •
- Prior to initiation of DUETACT therapy, the risks of hypoglycemia, its symptoms and treatment, and conditions that predispose to its development should be explained to patients and responsible family members [see Warnings and Precautions (5.2)]. Combination therapy of DUETACT with other antihyperglycemic agents may also cause hypoglycemia.
- •
- Patients who experience an unusually rapid increase in weight or edema or who develop shortness of breath or other symptoms of heart failure while on DUETACT should immediately report these symptoms to a physician.
- •
- Tell patients to promptly stop taking DUETACT and seek immediate medical advice if there is unexplained nausea, vomiting, abdominal pain, fatigue, anorexia, or dark urine as these symptoms may be due to hepatotoxicity.
- •
- Inform female patients that treatment with pioglitazone, like other thiazolidinediones may result in an unintended pregnancy in some premenopausal anovulatory females due to its effect on ovulation [see Use in Specific Populations (8.3)].
- •
- Patients should be told to take a single dose of DUETACT once daily with the first main meal and instructed that any change in dosing should be made only if directed by their physician [see Dosage and Administration (2)].
Distributed by:
Takeda Pharmaceuticals America, Inc.
Lexington, MA 02421DUETACT is a trademark of Takeda Pharmaceutical Company Limited registered with the U.S. Patent and Trademark Office and used under license by Takeda Pharmaceuticals America, Inc.
© 2009 - 2020 Takeda Pharmaceuticals America, Inc.
DTA007 R16
-
MEDICATION GUIDE
MEDICATION GUIDE
DUETACT (doo-et' -ăct)
(pioglitazone and glimepiride) tabletsRead this Medication Guide carefully before you start taking DUETACT and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about DUETACT, ask your doctor or pharmacist.
What is the most important information I should know about DUETACT?
DUETACT can cause serious side effects, including new or worse heart failure.- •
- Pioglitazone, one of the medicines in DUETACT, can cause your body to keep extra fluid (fluid retention), which leads to swelling (edema) and weight gain. Extra body fluid can make some heart problems worse or lead to heart failure. Heart failure means your heart does not pump blood well enough
- •
- Do not take DUETACT if you have severe heart failure
- •
- If you have heart failure with symptoms (such as shortness of breath or swelling), even if these symptoms are not severe, DUETACT may not be right for you
Call your doctor right away if you have any of the following:
- •
- swelling or fluid retention, especially in the ankles or legs
- •
- shortness of breath or trouble breathing, especially when you lie down
- •
- an unusually fast increase in weight
- •
- unusual tiredness
DUETACT can have other serious side effects. See "What are the possible side effects of DUETACT?"
What is DUETACT?
DUETACT is a prescription medicine used with diet and exercise to improve blood sugar (glucose) control in adults with type 2 diabetes.
DUETACT contains 2 prescription diabetes medicines called pioglitazone (ACTOS) and glimepiride, a sulfonylurea.
DUETACT is not for people with type 1 diabetes.
DUETACT is not for people with diabetic ketoacidosis (increased ketones in your blood or urine).
It is not known if DUETACT is safe and effective in children under the age of 18. DUETACT is not recommended for use in children.
Who should not take DUETACT?
See "What is the most important information I should know about DUETACT?"
Do not take DUETACT if you:
- •
- have severe heart failure
- •
- are allergic to any of the ingredients in DUETACT. See the end of this Medication Guide for a complete list of ingredients in DUETACT
- •
- have a condition called diabetic ketoacidosis. Diabetic ketoacidosis should be treated with insulin
Talk to your doctor before taking DUETACT if you have any of these conditions.
What should I tell my doctor before taking DUETACT?
Before you take DUETACT, tell your doctor if you:
- •
- have heart failure
- •
- have kidney problems
- •
- have type 1 ("juvenile") diabetes or had diabetic ketoacidosis
- •
- have a type of diabetic eye disease that causes swelling in the back of the eye (macular edema)
- •
- have liver problems
- •
- have or have had cancer of the bladder
- •
- are pregnant or plan to become pregnant. It is not known if DUETACT can harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant about the best way to control your blood glucose levels while pregnant
- •
- are a premenopausal woman (before the "change of life"), who does not have periods regularly or at all. DUETACT may increase your chance of becoming pregnant. Talk to your doctor about birth control choices while taking DUETACT. Tell your doctor right away if you become pregnant while taking DUETACT
- •
- are breastfeeding or plan to breastfeed. It is not known if DUETACT passes into your milk and if it can harm your baby. Talk to your doctor about the best way to control your blood glucose levels while breastfeeding
- •
- have G6PD deficiency (an inherited condition where you don't produce enough of the enzyme (G6PD). Taking glimepiride, one of the medicines in DUETACT, with this condition may cause your red blood cells to be destroyed too quickly (hemolytic anemia)
Tell your doctor about all the medicines you take including prescription and over the counter medicines, vitamins, and herbal supplements.
DUETACT and some of your other medicines can affect each other. You may need to have your dose of DUETACT or certain other medicines changed.
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist before you start a new medicine. They will tell you if it is okay to take DUETACT with other medicines.
How should I take DUETACT?
- •
- Take DUETACT exactly as your doctor tells you to take it
- •
- Your doctor may change your dose of DUETACT. Do not change your dose unless your doctor tells you to
- •
- DUETACT may be prescribed alone or with other diabetes medicines. This will depend on how well your blood sugar is controlled
- •
- Take DUETACT one time each day with the first main meal
- •
- If you take colesevelam, a medicine used to lower your cholesterol, take your DUETACT at least 4 hours before you take your colesevelam.
- •
- If you miss a dose of DUETACT, take your next dose as prescribed unless your doctor tells you differently. Do not take two doses at one time the next day
- •
- If you take too much DUETACT, call your doctor or go to the nearest hospital emergency room right away
- •
- If your body is under stress such as from a fever, infection, accident, or surgery, the dose of your diabetes medicines may need to be changed. Call your doctor right away
- •
- Stay on your diet and exercise programs and test your blood sugar regularly while taking DUETACT
- •
- Your doctor should do certain blood tests before you start and while you take DUETACT
- •
- Your doctor should also do hemoglobin A1C testing to check how well your blood sugar is controlled with DUETACT
- •
- Your doctor should check your eyes regularly while you take DUETACT
What are the possible side effects of DUETACT?
DUETACT may cause serious side effects including:
- •
- See "What is the most important information I should know about DUETACT?"
- •
- low blood sugar (hypoglycemia). This can happen if you skip meals, if you also use another medicine that lowers blood sugar, or if you have certain medical problems. Lightheadedness, dizziness, shakiness, or hunger may happen if your blood sugar is too low. Severe low blood sugar can cause unconsciousness (passing out), seizures, and death. Call your doctor if low blood sugar levels are a problem for you
- •
-
liver problems. Call your doctor right away if you have:
- nausea or vomiting
- stomach pain
- unusual or unexplained tiredness
- loss of appetite
- dark urine
- yellowing of your skin or the whites of your eyes
- •
-
bladder cancer. There may be an increased chance of having bladder cancer when you take DUETACT. You should not take DUETACT if you are receiving treatment for bladder cancer. Tell your doctor right away if you have any of the following symptoms of bladder cancer:
- blood or a red color in your urine
- an increased need to urinate
- pain while you urinate
- •
- broken bones (fractures). Usually in the hand, upper arm, or foot in women. Talk to your doctor for advice on how to keep your bones healthy.
- •
- diabetic eye disease with swelling in the back of the eye (macular edema). Tell your doctor right away if you have any changes in your vision. Your doctor should check your eyes regularly
- •
- release of an egg from an ovary in a woman (ovulation) leading to pregnancy. Ovulation may happen when premenopausal women who do not have regular monthly periods take DUETACT. This can increase your chance of getting pregnant
The most common side effects of DUETACT include:
- •
- cold-like symptoms (upper respiratory tract infection)
- •
- headache
- •
- sinus infection
- •
- diarrhea
- •
- nausea
- •
- muscle pain
- •
- sore throat
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the side effects of DUETACT. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store DUETACT?
- •
- Store DUETACT at 68°F to 77°F (20°C to 25°C). Keep DUETACT in the original container to protect from light
- •
- Keep the DUETACT bottle tightly closed and keep tablets dry
- •
- Keep DUETACT and all medicines out of the reach of children
General information about the safe and effective use of DUETACT
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use DUETACT for a condition for which it was not prescribed. Do not give DUETACT to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about DUETACT. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about DUETACT that is written for healthcare professionals. For more information, go to www.duetact.com or call 1-877-825-3327.
What are the ingredients in DUETACT?
Active ingredients: pioglitazone and glimepiride
Inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, hydroxypropyl cellulose, polysorbate 80, and microcrystalline cellulose
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Distributed by:
Takeda Pharmaceuticals America, Inc.
Lexington, MA 02421
Revised: December 2016
DUETACT and ACTOS are registered trademarks of Takeda Pharmaceutical Company Limited and used under license by Takeda Pharmaceuticals America, Inc.
© 2009 - 2020 Takeda Pharmaceuticals America, Inc.
DTA007 R16
- PRINCIPAL DISPLAY PANEL - 30 mg/2 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 30 mg/4 mg Tablet Bottle Label
-
INGREDIENTS AND APPEARANCE
DUETACT
pioglitazone and glimepiride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64764-302 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength pioglitazone hydrochloride (UNII: JQT35NPK6C) (pioglitazone - UNII:X4OV71U42S) pioglitazone 30 mg glimepiride (UNII: 6KY687524K) (glimepiride - UNII:6KY687524K) glimepiride 2 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) polysorbate 80 (UNII: 6OZP39ZG8H) microcrystalline cellulose (UNII: OP1R32D61U) Product Characteristics Color WHITE (white to off-white) Score no score Shape ROUND (convex) Size 8mm Flavor Imprint Code 30;2;4833G Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64764-302-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 07/28/2006 2 NDC:64764-302-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 07/28/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021925 07/28/2006 DUETACT
pioglitazone and glimepiride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64764-304 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength pioglitazone hydrochloride (UNII: JQT35NPK6C) (pioglitazone - UNII:X4OV71U42S) pioglitazone 30 mg glimepiride (UNII: 6KY687524K) (glimepiride - UNII:6KY687524K) glimepiride 4 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) polysorbate 80 (UNII: 6OZP39ZG8H) microcrystalline cellulose (UNII: OP1R32D61U) Product Characteristics Color WHITE (white to off-white) Score no score Shape ROUND (convex) Size 9mm Flavor Imprint Code 30;4;4833G Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64764-304-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 07/28/2006 2 NDC:64764-304-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 07/28/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021925 07/28/2006 Labeler - Takeda Pharmaceuticals America, Inc. (039997266) Establishment Name Address ID/FEI Business Operations Takeda GmbH 313270015 MANUFACTURE(64764-302, 64764-304) , ANALYSIS(64764-302, 64764-304)