STERILE WATER- sterile water injection
VetTek
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
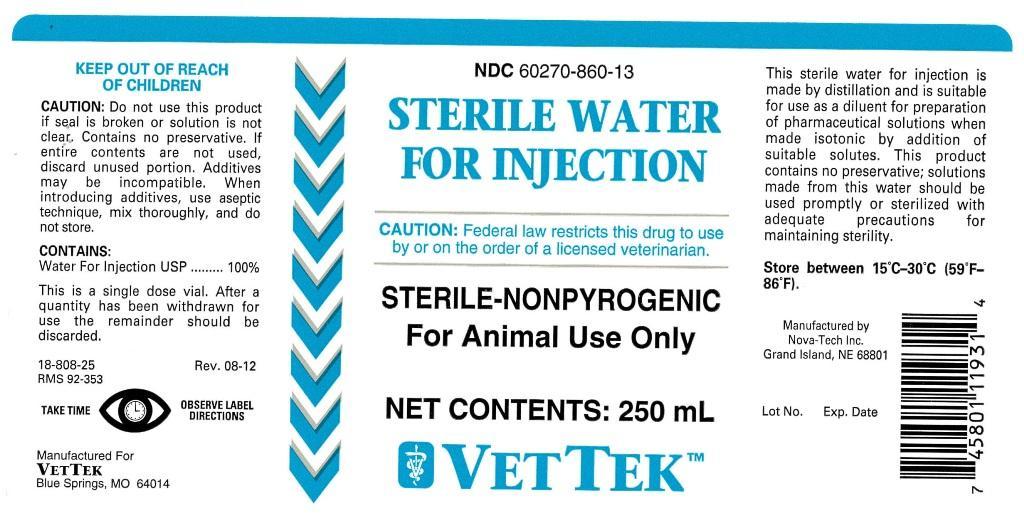
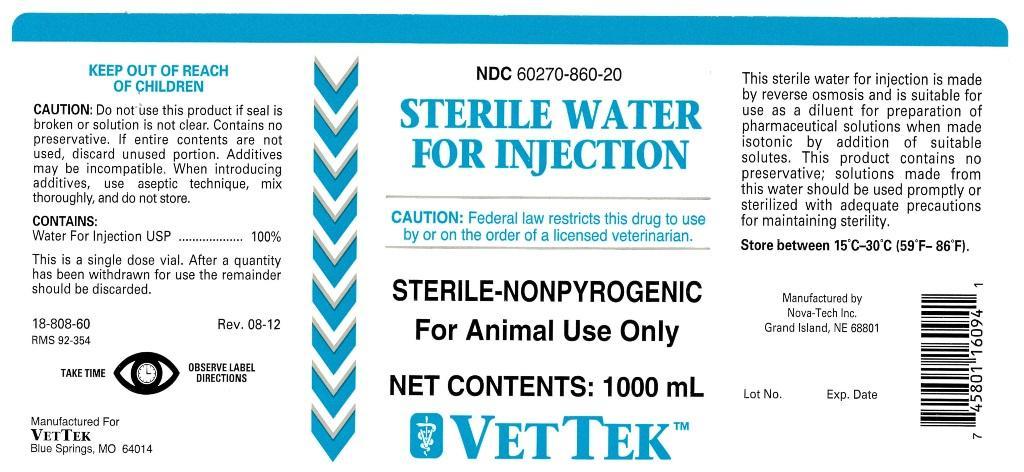
STERILE WATER FOR INJECTION
CAUTION:
Do not use this product if seal is broken or solution is not clear. Contains no preservative. If entire contents are not used, discard unused portion. Additives may be incomplatible. When introducing additives, use aseptic technique, mix thoroughly, and do not store.
CONTAINS:
Water For Injection USP.............100%
This is a single dose vial. After a quantity has been withdrawn for use the remainder should be discarded.
18-808-50 Rev. 08-12
RMS 92-352
Manufactured For
VETTEK
Blue Springs, MO 64014
STERILE-NONPYROGENIC
NET CONTENTS: 500 mL
Manufactured by
Nova-Tech Inc.
Grand Island, NE 68801
Lot No. Exp. Date
This sterile water for injection is made by reverse osmosis and is suitable for use as a diluent for preparation of pharmaceutical solutions when made isotonic by addition of suitable solutes. This product contains no preservative; solutions made from this water should be used promptly or sterilized with adequate pre-cautions for maintaining sterility.
| STERILE WATER
sterile water injection |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - VetTek (056387798) |
| Registrant - VetTek (056387798) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Nova-Tech, Inc. | 196078976 | manufacture, api manufacture | |