Label: ASPIRIN LOW DOSE- aspirin tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 49638-001-01, 49638-001-02 - Packager: America Medic
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 16, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
Do not use this product if you are allergic to Aspirin or any other pain/fever product.
If pregnant or breastfeeding contact a doctor before using.
Contact a doctor or pharmacist if you are taking a prescription drug for-Gout, Diabetes or Arthritis.
Contact a doctor right away if: An allergic reaction occurs.
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ASPIRIN LOW DOSE
aspirin tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49638-001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 81 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) METHACRYLIC ACID - METHYL METHACRYLATE COPOLYMER (1:1) (UNII: 74G4R6TH13) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color yellow (yellow ) Score no score Shape ROUND (tablet) Size 6mm Flavor Imprint Code C3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49638-001-02 1 in 1 CARTON 1 NDC:49638-001-01 120 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part343 05/15/2012 Labeler - America Medic (071065464) Establishment Name Address ID/FEI Business Operations America Medic 071065464 relabel, repack Establishment Name Address ID/FEI Business Operations Cispharma Inc. 833171445 manufacture

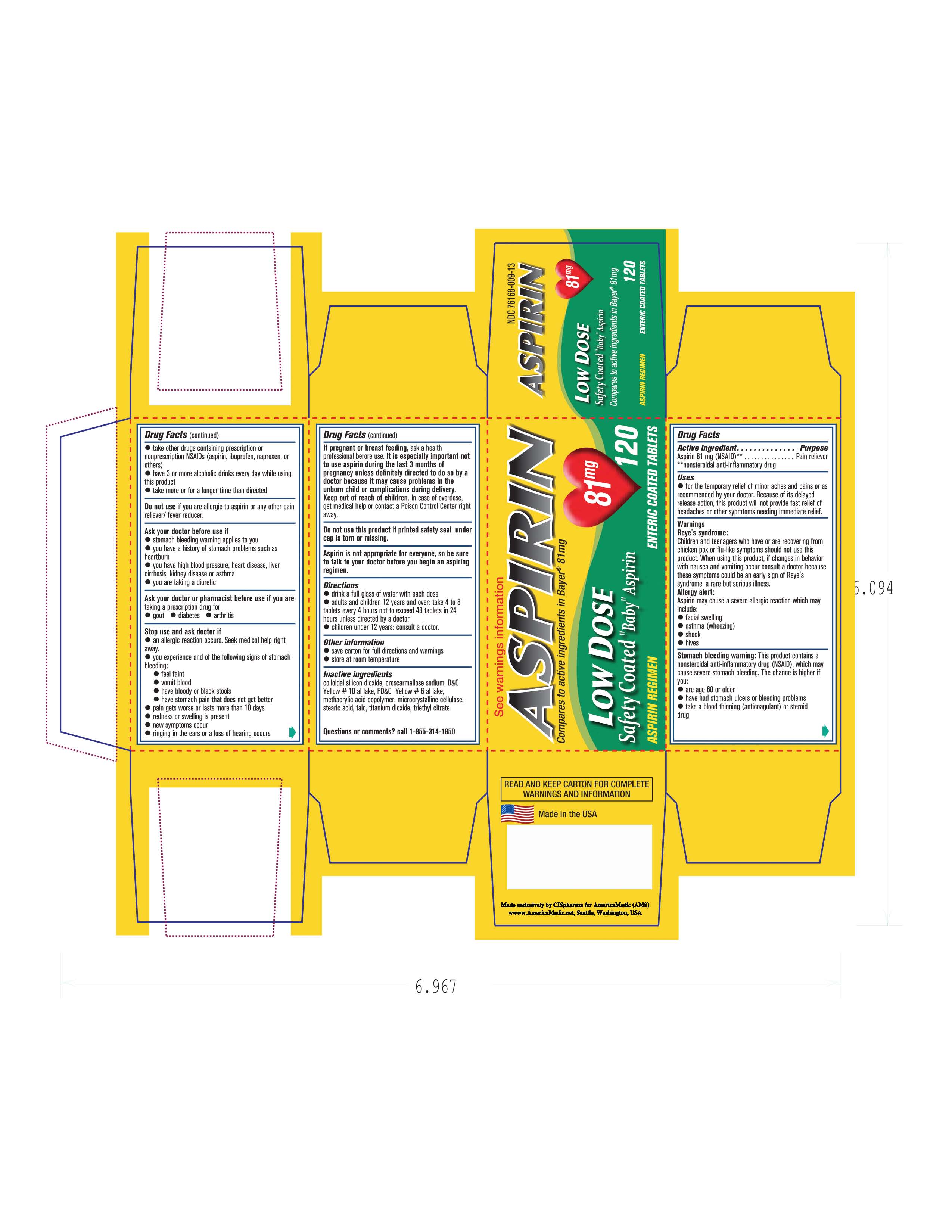
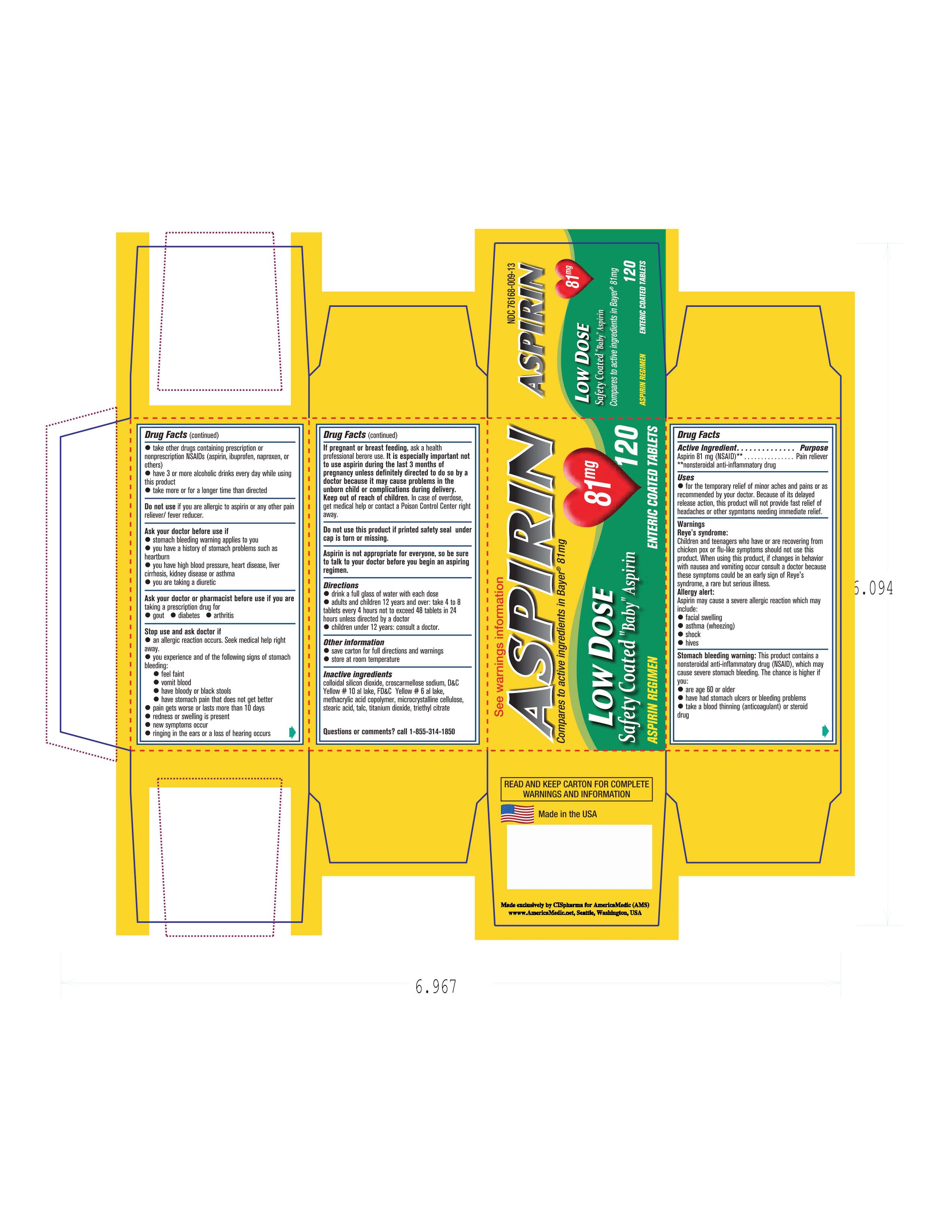 Image of carton
Image of carton