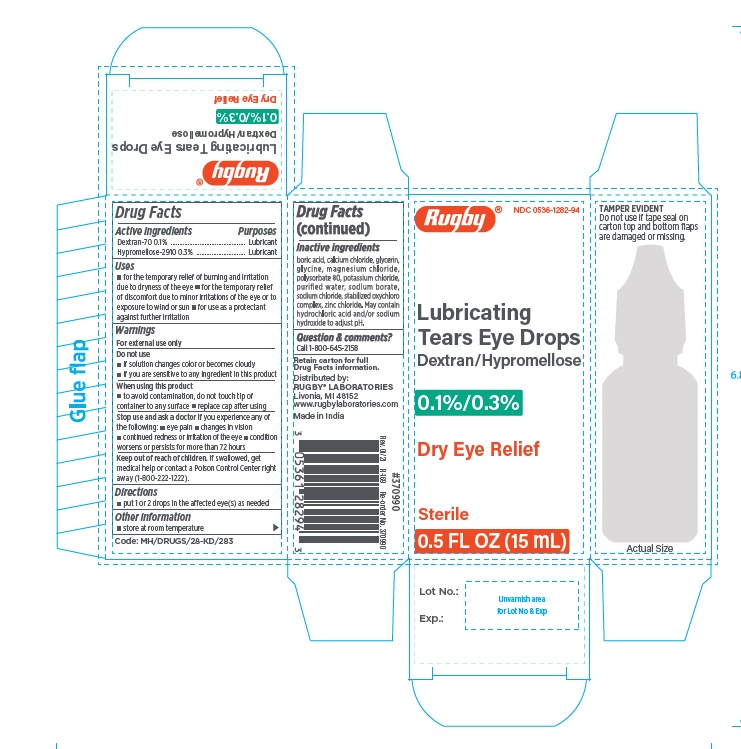
LUBRICATING EYE DROPS- dextran 70 and hypromellose 2910 solution/ drops
Rugby Laboratories
----------
Lubricating Tears Eyedrops
Uses
- temporary relief of burning and irritation due to dryness of the eye
- temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
- as a protectant against further irritation
Do not use
- if solution changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
When using this product
- to avoid contamination, do not touch tip of container to any surface
- replace cap after using
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
| LUBRICATING EYE DROPS
dextran 70 and hypromellose 2910 solution/ drops |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Rugby Laboratories (079246066) |
| Registrant - Velocity Pharma LLC (962198409) |
Revised: 1/2024
Document Id: 0fa853a8-61aa-5e6d-e063-6294a90a38ee
Set id: f5712fe5-fb2b-4c7d-865e-d3984bb5c4e4
Version: 6
Effective Time: 20240123
Rugby Laboratories