FOSAMAX- alendronate sodium tablet
Merck Sharp & Dohme Corp.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use FOSAMAX safely and effectively. See full prescribing information for FOSAMAX.
FOSAMAX® (alendronate sodium) tablets, for oral use FOSAMAX® (alendronate sodium) oral solution Initial U.S. Approval: 1995 INDICATIONS AND USAGEFOSAMAX is a bisphosphonate indicated for:
Limitations of use: DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSTablets: 70 mg (3) CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (greater than or equal to 3%) are abdominal pain, acid regurgitation, constipation, diarrhea, dyspepsia, musculoskeletal pain, nausea. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSUSE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 8/2019 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Treatment of Osteoporosis in Postmenopausal Women
FOSAMAX® is indicated for the treatment of osteoporosis in postmenopausal women. In postmenopausal women, FOSAMAX increases bone mass and reduces the incidence of fractures, including those of the hip and spine (vertebral compression fractures). [See Clinical Studies (14.1).]
1.2 Prevention of Osteoporosis in Postmenopausal Women
FOSAMAX is indicated for the prevention of postmenopausal osteoporosis [see Clinical Studies (14.2)].
1.3 Treatment to Increase Bone Mass in Men with Osteoporosis
FOSAMAX is indicated for treatment to increase bone mass in men with osteoporosis [see Clinical Studies (14.3)].
1.4 Treatment of Glucocorticoid-Induced Osteoporosis
FOSAMAX is indicated for the treatment of glucocorticoid-induced osteoporosis in men and women receiving glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and who have low bone mineral density [see Clinical Studies (14.4)].
1.5 Treatment of Paget's Disease of Bone
FOSAMAX is indicated for the treatment of Paget's disease of bone in men and women. Treatment is indicated in patients with Paget's disease of bone who have alkaline phosphatase at least two times the upper limit of normal, or those who are symptomatic, or those at risk for future complications from their disease. [See Clinical Studies (14.5).]
1.6 Important Limitations of Use
The optimal duration of use has not been determined. The safety and effectiveness of FOSAMAX for the treatment of osteoporosis are based on clinical data of four years duration. All patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis. Patients at low-risk for fracture should be considered for drug discontinuation after 3 to 5 years of use. Patients who discontinue therapy should have their risk for fracture re-evaluated periodically.
2 DOSAGE AND ADMINISTRATION
Although alendronate tablets 5 mg, 10 mg, 35 mg, and 40 mg are available in the marketplace, FOSAMAX is no longer marketed in the 5 mg, 10 mg, 35 mg, and 40 mg strengths.
Although an oral solution of alendronate may be available in the marketplace, FOSAMAX oral solution is no longer marketed.
2.1 Treatment of Osteoporosis in Postmenopausal Women
The recommended dosage is:
- one 70 mg tablet once weekly
or - one bottle of 70 mg oral solution once weekly
or - one 10 mg tablet once daily
2.2 Prevention of Osteoporosis in Postmenopausal Women
The recommended dosage is:
- one 35 mg tablet once weekly
or - one 5 mg tablet once daily
2.3 Treatment to Increase Bone Mass in Men with Osteoporosis
The recommended dosage is:
- one 70 mg tablet once weekly
or - one bottle of 70 mg oral solution once weekly
or - one 10 mg tablet once daily
2.4 Treatment of Glucocorticoid-Induced Osteoporosis
The recommended dosage is one 5 mg tablet once daily, except for postmenopausal women not receiving estrogen, for whom the recommended dosage is one 10 mg tablet once daily.
2.5 Treatment of Paget's Disease of Bone
The recommended treatment regimen is 40 mg once a day for six months.
Re-treatment of Paget's Disease
Re-treatment with FOSAMAX may be considered, following a six-month post-treatment evaluation period in patients who have relapsed, based on increases in serum alkaline phosphatase, which should be measured periodically. Re-treatment may also be considered in those who failed to normalize their serum alkaline phosphatase.
2.6 Important Administration Instructions
Instruct patients to do the following:
- Take FOSAMAX at least one-half hour before the first food, beverage, or medication of the day with plain water only [see Patient Counseling Information (17.2)]. Other beverages (including mineral water), food, and some medications are likely to reduce the absorption of FOSAMAX [see Drug Interactions (7.1)]. Waiting less than 30 minutes, or taking FOSAMAX with food, beverages (other than plain water) or other medications will lessen the effect of FOSAMAX by decreasing its absorption into the body.
- Take FOSAMAX upon arising for the day. To facilitate delivery to the stomach and thus reduce the potential for esophageal irritation, a FOSAMAX tablet should be swallowed with a full glass of water (6-8 ounces). To facilitate gastric emptying FOSAMAX oral solution should be followed by at least 2 ounces (a quarter of a cup) of water. Patients should not lie down for at least 30 minutes and until after their first food of the day. FOSAMAX should not be taken at bedtime or before arising for the day. Failure to follow these instructions may increase the risk of esophageal adverse experiences [see Warnings and Precautions (5.1) and Patient Counseling Information (17.2)].
2.7 Recommendations for Calcium and Vitamin D Supplementation
Instruct patients to take supplemental calcium if dietary intake is inadequate [see Warnings and Precautions (5.2)]. Patients at increased risk for vitamin D insufficiency (e.g., over the age of 70 years, nursing home-bound, or chronically ill) may need vitamin D supplementation. Patients with gastrointestinal malabsorption syndromes may require higher doses of vitamin D supplementation and measurement of 25-hydroxyvitamin D should be considered.
Patients treated with glucocorticoids should receive adequate amounts of calcium and vitamin D.
2.8 Administration Instructions for Missed Doses
If a once-weekly dose of FOSAMAX is missed, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day.
3 DOSAGE FORMS AND STRENGTHS
- 70 mg tablets are white, oval, uncoated tablets with code 31 on one side and an outline of a bone image on the other.
4 CONTRAINDICATIONS
FOSAMAX is contraindicated in patients with the following conditions:
- Abnormalities of the esophagus which delay esophageal emptying such as stricture or achalasia [see Warnings and Precautions (5.1)]
- Inability to stand or sit upright for at least 30 minutes [see Dosage and Administration (2.6); Warnings and Precautions (5.1)]
- Do not administer FOSAMAX oral solution to patients at increased risk of aspiration.
- Hypocalcemia [see Warnings and Precautions (5.2)]
- Hypersensitivity to any component of this product. Hypersensitivity reactions including urticaria and angioedema have been reported [see Adverse Reactions (6.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Upper Gastrointestinal Adverse Reactions
FOSAMAX, like other bisphosphonates administered orally, may cause local irritation of the upper gastrointestinal mucosa. Because of these possible irritant effects and a potential for worsening of the underlying disease, caution should be used when FOSAMAX is given to patients with active upper gastrointestinal problems (such as known Barrett's esophagus, dysphagia, other esophageal diseases, gastritis, duodenitis, or ulcers).
Esophageal adverse experiences, such as esophagitis, esophageal ulcers and esophageal erosions, occasionally with bleeding and rarely followed by esophageal stricture or perforation, have been reported in patients receiving treatment with oral bisphosphonates including FOSAMAX. In some cases these have been severe and required hospitalization. Physicians should therefore be alert to any signs or symptoms signaling a possible esophageal reaction and patients should be instructed to discontinue FOSAMAX and seek medical attention if they develop dysphagia, odynophagia, retrosternal pain or new or worsening heartburn.
The risk of severe esophageal adverse experiences appears to be greater in patients who lie down after taking oral bisphosphonates including FOSAMAX and/or who fail to swallow oral bisphosphonates including FOSAMAX with the recommended full glass (6-8 ounces) of water, and/or who continue to take oral bisphosphonates including FOSAMAX after developing symptoms suggestive of esophageal irritation. Therefore, it is very important that the full dosing instructions are provided to, and understood by, the patient [see Dosage and Administration (2.6)]. In patients who cannot comply with dosing instructions due to mental disability, therapy with FOSAMAX should be used under appropriate supervision.
There have been post-marketing reports of gastric and duodenal ulcers with oral bisphosphonate use, some severe and with complications, although no increased risk was observed in controlled clinical trials [see Adverse Reactions (6.2)].
5.2 Mineral Metabolism
Hypocalcemia must be corrected before initiating therapy with FOSAMAX [see Contraindications (4)]. Other disorders affecting mineral metabolism (such as vitamin D deficiency) should also be effectively treated. In patients with these conditions, serum calcium and symptoms of hypocalcemia should be monitored during therapy with FOSAMAX.
Presumably due to the effects of FOSAMAX on increasing bone mineral, small, asymptomatic decreases in serum calcium and phosphate may occur, especially in patients with Paget's disease, in whom the pretreatment rate of bone turnover may be greatly elevated, and in patients receiving glucocorticoids, in whom calcium absorption may be decreased.
Ensuring adequate calcium and vitamin D intake is especially important in patients with Paget's disease of bone and in patients receiving glucocorticoids.
5.3 Musculoskeletal Pain
In post-marketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain has been reported in patients taking bisphosphonates that are approved for the prevention and treatment of osteoporosis [see Adverse Reactions (6.2)]. This category of drugs includes FOSAMAX (alendronate). Most of the patients were postmenopausal women. The time to onset of symptoms varied from one day to several months after starting the drug. Discontinue use if severe symptoms develop. Most patients had relief of symptoms after stopping. A subset had recurrence of symptoms when rechallenged with the same drug or another bisphosphonate.
In placebo-controlled clinical studies of FOSAMAX, the percentages of patients with these symptoms were similar in the FOSAMAX and placebo groups.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ), which can occur spontaneously, is generally associated with tooth extraction and/or local infection with delayed healing, and has been reported in patients taking bisphosphonates, including FOSAMAX. Known risk factors for osteonecrosis of the jaw include invasive dental procedures (e.g., tooth extraction, dental implants, boney surgery), diagnosis of cancer, concomitant therapies (e.g., chemotherapy, corticosteroids, angiogenesis inhibitors), poor oral hygiene, and co-morbid disorders (e.g., periodontal and/or other pre-existing dental disease, anemia, coagulopathy, infection, ill-fitting dentures). The risk of ONJ may increase with duration of exposure to bisphosphonates.
For patients requiring invasive dental procedures, discontinuation of bisphosphonate treatment may reduce the risk for ONJ. Clinical judgment of the treating physician and/or oral surgeon should guide the management plan of each patient based on individual benefit/risk assessment.
Patients who develop osteonecrosis of the jaw while on bisphosphonate therapy should receive care by an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Discontinuation of bisphosphonate therapy should be considered based on individual benefit/risk assessment.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical, low-energy, or low trauma fractures of the femoral shaft have been reported in bisphosphonate-treated patients. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with bisphosphonates.
Atypical femur fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
Any patient with a history of bisphosphonate exposure who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of bisphosphonate therapy should be considered, pending a risk/benefit assessment, on an individual basis.
5.6 Renal Impairment
FOSAMAX is not recommended for patients with creatinine clearance less than 35 mL/min.
5.7 Glucocorticoid-Induced Osteoporosis
The risk versus benefit of FOSAMAX for treatment at daily dosages of glucocorticoids less than 7.5 mg of prednisone or equivalent has not been established [see Indications and Usage (1.4)]. Before initiating treatment, the gonadal hormonal status of both men and women should be ascertained and appropriate replacement considered.
A bone mineral density measurement should be made at the initiation of therapy and repeated after 6 to 12 months of combined FOSAMAX and glucocorticoid treatment.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Treatment of Osteoporosis in Postmenopausal Women
Daily Dosing
The safety of FOSAMAX in the treatment of postmenopausal osteoporosis was assessed in four clinical trials that enrolled 7453 women aged 44-84 years. Study 1 and Study 2 were identically designed, three-year, placebo-controlled, double-blind, multicenter studies (United States and Multinational n=994); Study 3 was the three-year vertebral fracture cohort of the Fracture Intervention Trial [FIT] (n=2027) and Study 4 was the four-year clinical fracture cohort of FIT (n=4432). Overall, 3620 patients were exposed to placebo and 3432 patients exposed to FOSAMAX. Patients with pre-existing gastrointestinal disease and concomitant use of non-steroidal anti-inflammatory drugs were included in these clinical trials. In Study 1 and Study 2 all women received 500 mg elemental calcium as carbonate. In Study 3 and Study 4 all women with dietary calcium intake less than 1000 mg per day received 500 mg calcium and 250 international units Vitamin D per day.
Among patients treated with alendronate 10 mg or placebo in Study 1 and Study 2, and all patients in Study 3 and Study 4, the incidence of all-cause mortality was 1.8% in the placebo group and 1.8% in the FOSAMAX group. The incidence of serious adverse event was 30.7% in the placebo group and 30.9% in the FOSAMAX group. The percentage of patients who discontinued the study due to any clinical adverse event was 9.5% in the placebo group and 8.9% in the FOSAMAX group. Adverse reactions from these studies considered by the investigators as possibly, probably, or definitely drug related in greater than or equal to 1% of patients treated with either FOSAMAX or placebo are presented in Table 1.
| United States/Multinational Studies | Fracture Intervention Trial | |||
|---|---|---|---|---|
| FOSAMAX*
% (n=196) | Placebo % (n=397) | FOSAMAX†
% (n=3236) | Placebo % (n=3223) |
|
| Gastrointestinal | ||||
| abdominal pain | 6.6 | 4.8 | 1.5 | 1.5 |
| nausea | 3.6 | 4.0 | 1.1 | 1.5 |
| dyspepsia | 3.6 | 3.5 | 1.1 | 1.2 |
| constipation | 3.1 | 1.8 | 0.0 | 0.2 |
| diarrhea | 3.1 | 1.8 | 0.6 | 0.3 |
| flatulence | 2.6 | 0.5 | 0.2 | 0.3 |
| acid regurgitation | 2.0 | 4.3 | 1.1 | 0.9 |
| esophageal ulcer | 1.5 | 0.0 | 0.1 | 0.1 |
| vomiting | 1.0 | 1.5 | 0.2 | 0.3 |
| dysphagia | 1.0 | 0.0 | 0.1 | 0.1 |
| abdominal distention | 1.0 | 0.8 | 0.0 | 0.0 |
| gastritis | 0.5 | 1.3 | 0.6 | 0.7 |
| Musculoskeletal | ||||
| musculoskeletal (bone, muscle or joint) pain | 4.1 | 2.5 | 0.4 | 0.3 |
| muscle cramp | 0.0 | 1.0 | 0.2 | 0.1 |
| Nervous System/Psychiatric | ||||
| headache | 2.6 | 1.5 | 0.2 | 0.2 |
| dizziness | 0.0 | 1.0 | 0.0 | 0.1 |
| Special Senses | ||||
| taste perversion | 0.5 | 1.0 | 0.1 | 0.0 |
Rash and erythema have occurred.
Gastrointestinal Adverse Reactions: One patient treated with FOSAMAX (10 mg/day), who had a history of peptic ulcer disease and gastrectomy and who was taking concomitant aspirin, developed an anastomotic ulcer with mild hemorrhage, which was considered drug related. Aspirin and FOSAMAX were discontinued and the patient recovered. In the Study 1 and Study 2 populations, 49-54% had a history of gastrointestinal disorders at baseline and 54-89% used nonsteroidal anti-inflammatory drugs or aspirin at some time during the studies. [See Warnings and Precautions (5.1).]
Laboratory Test Findings: In double-blind, multicenter, controlled studies, asymptomatic, mild, and transient decreases in serum calcium and phosphate were observed in approximately 18% and 10%, respectively, of patients taking FOSAMAX versus approximately 12% and 3% of those taking placebo. However, the incidences of decreases in serum calcium to less than 8.0 mg/dL (2.0 mM) and serum phosphate to less than or equal to 2.0 mg/dL (0.65 mM) were similar in both treatment groups.
Weekly Dosing
The safety of FOSAMAX 70 mg once weekly for the treatment of postmenopausal osteoporosis was assessed in a one-year, double-blind, multicenter study comparing FOSAMAX 70 mg once weekly and FOSAMAX 10 mg daily. The overall safety and tolerability profiles of once weekly FOSAMAX 70 mg and FOSAMAX 10 mg daily were similar. The adverse reactions considered by the investigators as possibly, probably, or definitely drug related in greater than or equal to 1% of patients in either treatment group are presented in Table 2.
| Once Weekly FOSAMAX 70 mg % (n=519) | FOSAMAX 10 mg/day % (n=370) |
|
|---|---|---|
| Gastrointestinal | ||
| abdominal pain | 3.7 | 3.0 |
| dyspepsia | 2.7 | 2.2 |
| acid regurgitation | 1.9 | 2.4 |
| nausea | 1.9 | 2.4 |
| abdominal distention | 1.0 | 1.4 |
| constipation | 0.8 | 1.6 |
| flatulence | 0.4 | 1.6 |
| gastritis | 0.2 | 1.1 |
| gastric ulcer | 0.0 | 1.1 |
| Musculoskeletal | ||
| musculoskeletal (bone, muscle, joint) pain | 2.9 | 3.2 |
| muscle cramp | 0.2 | 1.1 |
Prevention of Osteoporosis in Postmenopausal Women
Daily Dosing
The safety of FOSAMAX 5 mg/day in postmenopausal women 40-60 years of age has been evaluated in three double-blind, placebo-controlled studies involving over 1,400 patients randomized to receive FOSAMAX for either two or three years. In these studies the overall safety profiles of FOSAMAX 5 mg/day and placebo were similar. Discontinuation of therapy due to any clinical adverse event occurred in 7.5% of 642 patients treated with FOSAMAX 5 mg/day and 5.7% of 648 patients treated with placebo.
Weekly Dosing
The safety of FOSAMAX 35 mg once weekly compared to FOSAMAX 5 mg daily was evaluated in a one-year, double-blind, multicenter study of 723 patients. The overall safety and tolerability profiles of once weekly FOSAMAX 35 mg and FOSAMAX 5 mg daily were similar.
The adverse reactions from these studies considered by the investigators as possibly, probably, or definitely drug related in greater than or equal to 1% of patients treated with either once weekly FOSAMAX 35 mg, FOSAMAX 5 mg/day or placebo are presented in Table 3.
| Two/Three-Year Studies | One-Year Study | |||
|---|---|---|---|---|
| FOSAMAX 5 mg/day % (n=642) | Placebo % (n=648) | FOSAMAX 5 mg/day % (n=361) | Once Weekly FOSAMAX 35 mg % (n=362) |
|
| Gastrointestinal | ||||
| dyspepsia | 1.9 | 1.4 | 2.2 | 1.7 |
| abdominal pain | 1.7 | 3.4 | 4.2 | 2.2 |
| acid regurgitation | 1.4 | 2.5 | 4.2 | 4.7 |
| nausea | 1.4 | 1.4 | 2.5 | 1.4 |
| diarrhea | 1.1 | 1.7 | 1.1 | 0.6 |
| constipation | 0.9 | 0.5 | 1.7 | 0.3 |
| abdominal distention | 0.2 | 0.3 | 1.4 | 1.1 |
| Musculoskeletal | ||||
| musculoskeletal (bone, muscle or joint) pain | 0.8 | 0.9 | 1.9 | 2.2 |
Concomitant Use with Estrogen/Hormone Replacement Therapy
In two studies (of one and two years' duration) of postmenopausal osteoporotic women (total: n=853), the safety and tolerability profile of combined treatment with FOSAMAX 10 mg once daily and estrogen ± progestin (n=354) was consistent with those of the individual treatments.
Osteoporosis in Men
In two placebo-controlled, double-blind, multicenter studies in men (a two-year study of FOSAMAX 10 mg/day and a one-year study of once weekly FOSAMAX 70 mg) the rates of discontinuation of therapy due to any clinical adverse event were 2.7% for FOSAMAX 10 mg/day vs. 10.5% for placebo, and 6.4% for once weekly FOSAMAX 70 mg vs. 8.6% for placebo. The adverse reactions considered by the investigators as possibly, probably, or definitely drug related in greater than or equal to 2% of patients treated with either FOSAMAX or placebo are presented in Table 4.
| Two-year Study | One-year Study | |||
|---|---|---|---|---|
|
FOSAMAX 10 mg/day % (n=146) | Placebo % (n=95) | Once Weekly FOSAMAX 70 mg % (n=109) |
Placebo % (n=58) |
|
| Gastrointestinal | ||||
| acid regurgitation | 4.1 | 3.2 | 0.0 | 0.0 |
| flatulence | 4.1 | 1.1 | 0.0 | 0.0 |
| gastroesophageal reflux disease | 0.7 | 3.2 | 2.8 | 0.0 |
| dyspepsia | 3.4 | 0.0 | 2.8 | 1.7 |
| diarrhea | 1.4 | 1.1 | 2.8 | 0.0 |
| abdominal pain | 2.1 | 1.1 | 0.9 | 3.4 |
| nausea | 2.1 | 0.0 | 0.0 | 0.0 |
Glucocorticoid-Induced Osteoporosis
In two, one-year, placebo-controlled, double-blind, multicenter studies in patients receiving glucocorticoid treatment, the overall safety and tolerability profiles of FOSAMAX 5 and 10 mg/day were generally similar to that of placebo. The adverse reactions considered by the investigators as possibly, probably, or definitely drug related in greater than or equal to 1% of patients treated with either FOSAMAX 5 or 10 mg/day or placebo are presented in Table 5.
| FOSAMAX 10 mg/day % (n=157) | FOSAMAX 5 mg/day % (n=161) | Placebo % (n=159) |
|
|---|---|---|---|
| Gastrointestinal | |||
| abdominal pain | 3.2 | 1.9 | 0.0 |
| acid regurgitation | 2.5 | 1.9 | 1.3 |
| constipation | 1.3 | 0.6 | 0.0 |
| melena | 1.3 | 0.0 | 0.0 |
| nausea | 0.6 | 1.2 | 0.6 |
| diarrhea | 0.0 | 0.0 | 1.3 |
| Nervous System/Psychiatric | |||
| headache | 0.6 | 0.0 | 1.3 |
The overall safety and tolerability profile in the glucocorticoid-induced osteoporosis population that continued therapy for the second year of the studies (FOSAMAX: n=147) was consistent with that observed in the first year.
Paget's Disease of Bone
In clinical studies (osteoporosis and Paget's disease), adverse events reported in 175 patients taking FOSAMAX 40 mg/day for 3-12 months were similar to those in postmenopausal women treated with FOSAMAX 10 mg/day. However, there was an apparent increased incidence of upper gastrointestinal adverse reactions in patients taking FOSAMAX 40 mg/day (17.7% FOSAMAX vs. 10.2% placebo). One case of esophagitis and two cases of gastritis resulted in discontinuation of treatment.
Additionally, musculoskeletal (bone, muscle or joint) pain, which has been described in patients with Paget's disease treated with other bisphosphonates, was considered by the investigators as possibly, probably, or definitely drug related in approximately 6% of patients treated with FOSAMAX 40 mg/day versus approximately 1% of patients treated with placebo, but rarely resulted in discontinuation of therapy. Discontinuation of therapy due to any clinical adverse events occurred in 6.4% of patients with Paget's disease treated with FOSAMAX 40 mg/day and 2.4% of patients treated with placebo.
6.2 Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of FOSAMAX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: hypersensitivity reactions including urticaria and angioedema. Transient symptoms of myalgia, malaise, asthenia and fever have been reported with FOSAMAX, typically in association with initiation of treatment. Symptomatic hypocalcemia has occurred, generally in association with predisposing conditions. Peripheral edema.
Gastrointestinal: esophagitis, esophageal erosions, esophageal ulcers, esophageal stricture or perforation, and oropharyngeal ulceration. Gastric or duodenal ulcers, some severe and with complications, have also been reported [see Dosage and Administration (2.6); Warnings and Precautions (5.1)].
Localized osteonecrosis of the jaw, generally associated with tooth extraction and/or local infection with delayed healing, has been reported [see Warnings and Precautions (5.4)].
Musculoskeletal: bone, joint, and/or muscle pain, occasionally severe, and incapacitating [see Warnings and Precautions (5.3)]; joint swelling; low-energy femoral shaft and subtrochanteric fractures [see Warnings and Precautions (5.5)].
Nervous System: dizziness and vertigo.
Pulmonary: acute asthma exacerbations.
Skin: rash (occasionally with photosensitivity), pruritus, alopecia, severe skin reactions, including Stevens-Johnson syndrome and toxic epidermal necrolysis.
Special Senses: uveitis, scleritis or episcleritis. Cholesteatoma of the external auditory canal (focal osteonecrosis).
7 DRUG INTERACTIONS
7.1 Calcium Supplements/Antacids
Co-administration of FOSAMAX and calcium, antacids, or oral medications containing multivalent cations will interfere with absorption of FOSAMAX. Therefore, instruct patients to wait at least one-half hour after taking FOSAMAX before taking any other oral medications.
7.2 Aspirin
In clinical studies, the incidence of upper gastrointestinal adverse events was increased in patients receiving concomitant therapy with daily doses of FOSAMAX greater than 10 mg and aspirin-containing products.
7.3 Nonsteroidal Anti-Inflammatory Drugs
FOSAMAX may be administered to patients taking nonsteroidal anti-inflammatory drugs (NSAIDs). In a 3-year, controlled, clinical study (n=2027) during which a majority of patients received concomitant NSAIDs, the incidence of upper gastrointestinal adverse events was similar in patients taking FOSAMAX 5 or 10 mg/day compared to those taking placebo. However, since NSAID use is associated with gastrointestinal irritation, caution should be used during concomitant use with FOSAMAX.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data on the use of FOSAMAX in pregnant women are insufficient to inform a drug-associated risk of adverse maternal or fetal outcomes. Discontinue FOSAMAX when pregnancy is recognized.
In animal reproduction studies, daily oral administration of alendronate to rats from before mating through the end of gestation or lactation showed decreased postimplantation survival and decreased pup body weight gain starting at doses equivalent to less than half of the highest recommended 40 mg clinical daily dose (based on body surface area, mg/m2). Oral administration of alendronate to rats during organogenesis resulted in reduced fetal ossification starting at doses 3 times the 40 mg clinical daily dose. No similar fetal effects were observed in pregnant rabbits dosed orally during organogenesis at doses equivalent to approximately 10 times the 40 mg clinical daily dose.
Delayed or failed delivery of offspring, protracted parturition, and late pregnancy maternal and fetal deaths due to maternal hypocalcemia occurred in rats at oral doses as low as one tenth the 40 mg clinical daily dose (see Data).
Bisphosphonates are incorporated into the bone matrix, from which they are gradually released over a period of years. The amount of bisphosphonate incorporated into adult bone and available for release into the systemic circulation is directly related to the dose and duration of bisphosphonate use. Consequently, based on the mechanism of action of bisphosphonates, there is a potential risk of fetal harm, predominantly skeletal, if a woman becomes pregnant after completing a course of bisphosphonate therapy. The impact of variables such as time between cessation of bisphosphonate therapy to conception, the particular bisphosphonate used, and the route of administration (intravenous versus oral) on the risk has not been studied.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Reproduction studies in rats dosed orally from before mating to the end of gestation or lactation showed decreased postimplantation survival starting at 2 mg/kg/day and decreased body weight gain starting at 1 mg/kg/day, doses equivalent to less than half the 40 mg clinical daily dose based on body surface area, mg/m2. Incidence of incomplete fetal ossification in vertebral, skull, and sternebral bones were increased in rats dosed orally during organogenesis starting at 10 mg/kg/day (approximately 3 times the 40 mg clinical daily dose). No similar fetal effects were observed in pregnant rabbits dosed orally during organogenesis at up to 35 mg/kg/day (equivalent to approximately 10 times the 40 mg clinical daily dose).
Both total and ionized calcium decreased in pregnant rats dosed orally with 15 mg/kg/day alendronate (approximately 4 times the 40 mg clinical daily dose) resulting in delays and failures of delivery. Protracted parturition due to maternal hypocalcemia was observed when rats were treated from before mating through gestation starting at 0.5 mg/kg/day (approximately one tenth the 40 mg clinical daily dose). Maternotoxicity (late pregnancy deaths) also occurred in female rats treated orally with 15 mg/kg/day (approximately 4 times the 40 mg clinical daily dose) for varying gestational time periods. These maternal deaths were lessened but not eliminated by cessation of treatment. Calcium supplementation in the drinking water or by subcutaneous minipump to rats dosed orally with 15 mg/kg/day alendronate could not ameliorate the hypocalcemia or prevent the dystocia-related maternal and neonatal deaths. However, intravenous calcium supplementation prevented maternal, but not neonatal deaths.
8.2 Lactation
Risk Summary
It is not known whether alendronate is present in human breast milk, affects human milk production, or has effects on the breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for FOSAMAX and any potential adverse effects on the breastfed child from FOSAMAX or from the underlying maternal condition.
8.4 Pediatric Use
FOSAMAX is not indicated for use in pediatric patients.
The safety and efficacy of FOSAMAX were examined in a randomized, double-blind, placebo-controlled two-year study of 139 pediatric patients, aged 4-18 years, with severe osteogenesis imperfecta (OI). One-hundred-and-nine patients were randomized to 5 mg FOSAMAX daily (weight less than 40 kg) or 10 mg FOSAMAX daily (weight greater than or equal to 40 kg) and 30 patients to placebo. The mean baseline lumbar spine BMD Z-score of the patients was -4.5. The mean change in lumbar spine BMD Z-score from baseline to Month 24 was 1.3 in the FOSAMAX-treated patients and 0.1 in the placebo-treated patients. Treatment with FOSAMAX did not reduce the risk of fracture. Sixteen percent of the FOSAMAX patients who sustained a radiologically-confirmed fracture by Month 12 of the study had delayed fracture healing (callus remodeling) or fracture non-union when assessed radiographically at Month 24 compared with 9% of the placebo-treated patients. In FOSAMAX-treated patients, bone histomorphometry data obtained at Month 24 demonstrated decreased bone turnover and delayed mineralization time; however, there were no mineralization defects. There were no statistically significant differences between the FOSAMAX and placebo groups in reduction of bone pain. The oral bioavailability in children was similar to that observed in adults.
The overall safety profile of FOSAMAX in osteogenesis imperfecta patients treated for up to 24 months was generally similar to that of adults with osteoporosis treated with FOSAMAX. However, there was an increased occurrence of vomiting in osteogenesis imperfecta patients treated with FOSAMAX compared to placebo. During the 24-month treatment period, vomiting was observed in 32 of 109 (29.4%) patients treated with FOSAMAX and 3 of 30 (10%) patients treated with placebo.
In a pharmacokinetic study, 6 of 24 pediatric osteogenesis imperfecta patients who received a single oral dose of FOSAMAX 35 or 70 mg developed fever, flu-like symptoms, and/or mild lymphocytopenia within 24 to 48 hours after administration. These events, lasting no more than 2 to 3 days and responding to acetaminophen, are consistent with an acute-phase response that has been reported in patients receiving bisphosphonates, including FOSAMAX. [See Adverse Reactions (6.2).]
8.5 Geriatric Use
Of the patients receiving FOSAMAX in the Fracture Intervention Trial (FIT), 71% (n=2302) were greater than or equal to 65 years of age and 17% (n=550) were greater than or equal to 75 years of age. Of the patients receiving FOSAMAX in the United States and Multinational osteoporosis treatment studies in women, osteoporosis studies in men, glucocorticoid-induced osteoporosis studies, and Paget's disease studies [see Clinical Studies (14.1), (14.3), (14.4), (14.5)], 45%, 54%, 37%, and 70%, respectively, were 65 years of age or over. No overall differences in efficacy or safety were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
FOSAMAX is not recommended for patients with creatinine clearance less than 35 mL/min. No dosage adjustment is necessary in patients with creatinine clearance values between 35-60 mL/min [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
As there is evidence that alendronate is not metabolized or excreted in the bile, no studies were conducted in patients with hepatic impairment. No dosage adjustment is necessary [see Clinical Pharmacology (12.3)].
10 OVERDOSAGE
Significant lethality after single oral doses was seen in female rats and mice at 552 mg/kg (3256 mg/m2) and 966 mg/kg (2898 mg/m2), respectively. In males, these values were slightly higher, 626 and 1280 mg/kg, respectively. There was no lethality in dogs at oral doses up to 200 mg/kg (4000 mg/m2).
No specific information is available on the treatment of overdosage with FOSAMAX. Hypocalcemia, hypophosphatemia, and upper gastrointestinal adverse events, such as upset stomach, heartburn, esophagitis, gastritis, or ulcer, may result from oral overdosage. Milk or antacids should be given to bind alendronate. Due to the risk of esophageal irritation, vomiting should not be induced and the patient should remain fully upright.
Dialysis would not be beneficial.
11 DESCRIPTION
FOSAMAX (alendronate sodium) is a bisphosphonate that acts as a specific inhibitor of osteoclast-mediated bone resorption. Bisphosphonates are synthetic analogs of pyrophosphate that bind to the hydroxyapatite found in bone.
Alendronate sodium is chemically described as (4-amino-1-hydroxybutylidene) bisphosphonic acid monosodium salt trihydrate.
The empirical formula of alendronate sodium is C4H12NNaO7P2•3H2O and its formula weight is 325.12. The structural formula is:
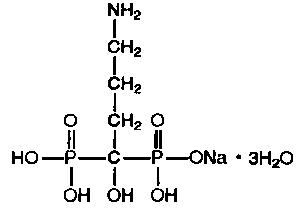
Alendronate sodium is a white, crystalline, nonhygroscopic powder. It is soluble in water, very slightly soluble in alcohol, and practically insoluble in chloroform.
FOSAMAX tablets for oral administration contain 91.37 mg of alendronate monosodium salt trihydrate, which is the molar equivalent of 70 mg of free acid, and the following inactive ingredients: microcrystalline cellulose, anhydrous lactose, croscarmellose sodium, and magnesium stearate.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Animal studies have indicated the following mode of action. At the cellular level, alendronate shows preferential localization to sites of bone resorption, specifically under osteoclasts. The osteoclasts adhere normally to the bone surface but lack the ruffled border that is indicative of active resorption. Alendronate does not interfere with osteoclast recruitment or attachment, but it does inhibit osteoclast activity. Studies in mice on the localization of radioactive [3H]alendronate in bone showed about 10-fold higher uptake on osteoclast surfaces than on osteoblast surfaces. Bones examined 6 and 49 days after [3H]alendronate administration in rats and mice, respectively, showed that normal bone was formed on top of the alendronate, which was incorporated inside the matrix. While incorporated in bone matrix, alendronate is not pharmacologically active. Thus, alendronate must be continuously administered to suppress osteoclasts on newly formed resorption surfaces. Histomorphometry in baboons and rats showed that alendronate treatment reduces bone turnover (i.e., the number of sites at which bone is remodeled). In addition, bone formation exceeds bone resorption at these remodeling sites, leading to progressive gains in bone mass.
12.2 Pharmacodynamics
Alendronate is a bisphosphonate that binds to bone hydroxyapatite and specifically inhibits the activity of osteoclasts, the bone-resorbing cells. Alendronate reduces bone resorption with no direct effect on bone formation, although the latter process is ultimately reduced because bone resorption and formation are coupled during bone turnover.
Osteoporosis in Postmenopausal Women
Osteoporosis is characterized by low bone mass that leads to an increased risk of fracture. The diagnosis can be confirmed by the finding of low bone mass, evidence of fracture on x-ray, a history of osteoporotic fracture, or height loss or kyphosis, indicative of vertebral (spinal) fracture. Osteoporosis occurs in both males and females but is most common among women following the menopause, when bone turnover increases and the rate of bone resorption exceeds that of bone formation. These changes result in progressive bone loss and lead to osteoporosis in a significant proportion of women over age 50. Fractures, usually of the spine, hip, and wrist, are the common consequences. From age 50 to age 90, the risk of hip fracture in white women increases 50-fold and the risk of vertebral fracture 15- to 30-fold. It is estimated that approximately 40% of 50-year-old women will sustain one or more osteoporosis-related fractures of the spine, hip, or wrist during their remaining lifetimes. Hip fractures, in particular, are associated with substantial morbidity, disability, and mortality.
Daily oral doses of alendronate (5, 20, and 40 mg for six weeks) in postmenopausal women produced biochemical changes indicative of dose-dependent inhibition of bone resorption, including decreases in urinary calcium and urinary markers of bone collagen degradation (such as deoxypyridinoline and cross-linked N-telopeptides of type I collagen). These biochemical changes tended to return toward baseline values as early as 3 weeks following the discontinuation of therapy with alendronate and did not differ from placebo after 7 months.
Long-term treatment of osteoporosis with FOSAMAX 10 mg/day (for up to five years) reduced urinary excretion of markers of bone resorption, deoxypyridinoline and cross-linked N-telopeptides of type l collagen, by approximately 50% and 70%, respectively, to reach levels similar to those seen in healthy premenopausal women. Similar decreases were seen in patients in osteoporosis prevention studies who received FOSAMAX 5 mg/day. The decrease in the rate of bone resorption indicated by these markers was evident as early as one month and at three to six months reached a plateau that was maintained for the entire duration of treatment with FOSAMAX. In osteoporosis treatment studies FOSAMAX 10 mg/day decreased the markers of bone formation, osteocalcin and bone specific alkaline phosphatase by approximately 50%, and total serum alkaline phosphatase by approximately 25 to 30% to reach a plateau after 6 to 12 months. In osteoporosis prevention studies FOSAMAX 5 mg/day decreased osteocalcin and total serum alkaline phosphatase by approximately 40% and 15%, respectively. Similar reductions in the rate of bone turnover were observed in postmenopausal women during one-year studies with once weekly FOSAMAX 70 mg for the treatment of osteoporosis and once weekly FOSAMAX 35 mg for the prevention of osteoporosis. These data indicate that the rate of bone turnover reached a new steady-state, despite the progressive increase in the total amount of alendronate deposited within bone.
As a result of inhibition of bone resorption, asymptomatic reductions in serum calcium and phosphate concentrations were also observed following treatment with FOSAMAX. In the long-term studies, reductions from baseline in serum calcium (approximately 2%) and phosphate (approximately 4 to 6%) were evident the first month after the initiation of FOSAMAX 10 mg. No further decreases in serum calcium were observed for the five-year duration of treatment; however, serum phosphate returned toward prestudy levels during years three through five. Similar reductions were observed with FOSAMAX 5 mg/day. In one-year studies with once weekly FOSAMAX 35 and 70 mg, similar reductions were observed at 6 and 12 months. The reduction in serum phosphate may reflect not only the positive bone mineral balance due to FOSAMAX but also a decrease in renal phosphate reabsorption.
Osteoporosis in Men
Treatment of men with osteoporosis with FOSAMAX 10 mg/day for two years reduced urinary excretion of cross-linked N-telopeptides of type I collagen by approximately 60% and bone-specific alkaline phosphatase by approximately 40%. Similar reductions were observed in a one-year study in men with osteoporosis receiving once weekly FOSAMAX 70 mg.
Glucocorticoid-Induced Osteoporosis
Sustained use of glucocorticoids is commonly associated with development of osteoporosis and resulting fractures (especially vertebral, hip, and rib). It occurs both in males and females of all ages. Osteoporosis occurs as a result of inhibited bone formation and increased bone resorption resulting in net bone loss. Alendronate decreases bone resorption without directly inhibiting bone formation.
In clinical studies of up to two years' duration, FOSAMAX 5 and 10 mg/day reduced cross-linked N-telopeptides of type I collagen (a marker of bone resorption) by approximately 60% and reduced bone-specific alkaline phosphatase and total serum alkaline phosphatase (markers of bone formation) by approximately 15 to 30% and 8 to 18%, respectively. As a result of inhibition of bone resorption, FOSAMAX 5 and 10 mg/day induced asymptomatic decreases in serum calcium (approximately 1 to 2%) and serum phosphate (approximately 1 to 8%).
Paget's Disease of Bone
Paget's disease of bone is a chronic, focal skeletal disorder characterized by greatly increased and disorderly bone remodeling. Excessive osteoclastic bone resorption is followed by osteoblastic new bone formation, leading to the replacement of the normal bone architecture by disorganized, enlarged, and weakened bone structure.
Clinical manifestations of Paget's disease range from no symptoms to severe morbidity due to bone pain, bone deformity, pathological fractures, and neurological and other complications. Serum alkaline phosphatase, the most frequently used biochemical index of disease activity, provides an objective measure of disease severity and response to therapy.
FOSAMAX decreases the rate of bone resorption directly, which leads to an indirect decrease in bone formation. In clinical trials, FOSAMAX 40 mg once daily for six months produced significant decreases in serum alkaline phosphatase as well as in urinary markers of bone collagen degradation. As a result of the inhibition of bone resorption, FOSAMAX induced generally mild, transient, and asymptomatic decreases in serum calcium and phosphate.
12.3 Pharmacokinetics
Absorption
Relative to an intravenous reference dose, the mean oral bioavailability of alendronate in women was 0.64% for doses ranging from 5 to 70 mg when administered after an overnight fast and two hours before a standardized breakfast. Oral bioavailability of the 10 mg tablet in men (0.59%) was similar to that in women when administered after an overnight fast and 2 hours before breakfast.
FOSAMAX 70 mg oral solution and FOSAMAX 70 mg tablet are equally bioavailable.
A study examining the effect of timing of a meal on the bioavailability of alendronate was performed in 49 postmenopausal women. Bioavailability was decreased (by approximately 40%) when 10 mg alendronate was administered either 0.5 or 1 hour before a standardized breakfast, when compared to dosing 2 hours before eating. In studies of treatment and prevention of osteoporosis, alendronate was effective when administered at least 30 minutes before breakfast.
Bioavailability was negligible whether alendronate was administered with or up to two hours after a standardized breakfast. Concomitant administration of alendronate with coffee or orange juice reduced bioavailability by approximately 60%.
Distribution
Preclinical studies (in male rats) show that alendronate transiently distributes to soft tissues following 1 mg/kg intravenous administration but is then rapidly redistributed to bone or excreted in the urine. The mean steady-state volume of distribution, exclusive of bone, is at least 28 L in humans. Concentrations of drug in plasma following therapeutic oral doses are too low (less than 5 ng/mL) for analytical detection. Protein binding in human plasma is approximately 78%.
Excretion
Following a single intravenous dose of [14C]alendronate, approximately 50% of the radioactivity was excreted in the urine within 72 hours and little or no radioactivity was recovered in the feces. Following a single 10 mg intravenous dose, the renal clearance of alendronate was 71 mL/min (64, 78; 90% confidence interval [CI]), and systemic clearance did not exceed 200 mL/min. Plasma concentrations fell by more than 95% within 6 hours following intravenous administration. The terminal half-life in humans is estimated to exceed 10 years, probably reflecting release of alendronate from the skeleton. Based on the above, it is estimated that after 10 years of oral treatment with FOSAMAX (10 mg daily) the amount of alendronate released daily from the skeleton is approximately 25% of that absorbed from the gastrointestinal tract.
Specific Populations
Gender: Bioavailability and the fraction of an intravenous dose excreted in urine were similar in men and women.
Geriatric: Bioavailability and disposition (urinary excretion) were similar in elderly and younger patients. No dosage adjustment is necessary in elderly patients.
Race: Pharmacokinetic differences due to race have not been studied.
Renal Impairment: Preclinical studies show that, in rats with kidney failure, increasing amounts of drug are present in plasma, kidney, spleen, and tibia. In healthy controls, drug that is not deposited in bone is rapidly excreted in the urine. No evidence of saturation of bone uptake was found after 3 weeks dosing with cumulative intravenous doses of 35 mg/kg in young male rats. Although no formal renal impairment pharmacokinetic study has been conducted in patients, it is likely that, as in animals, elimination of alendronate via the kidney will be reduced in patients with impaired renal function. Therefore, somewhat greater accumulation of alendronate in bone might be expected in patients with impaired renal function.
No dosage adjustment is necessary for patients with creatinine clearance 35 to 60 mL/min. FOSAMAX is not recommended for patients with creatinine clearance less than 35 mL/min due to lack of experience with alendronate in renal failure.
Hepatic Impairment: As there is evidence that alendronate is not metabolized or excreted in the bile, no studies were conducted in patients with hepatic impairment. No dosage adjustment is necessary.
Drug Interactions
Intravenous ranitidine was shown to double the bioavailability of oral alendronate. The clinical significance of this increased bioavailability and whether similar increases will occur in patients given oral H2-antagonists is unknown.
In healthy subjects, oral prednisone (20 mg three times daily for five days) did not produce a clinically meaningful change in the oral bioavailability of alendronate (a mean increase ranging from 20 to 44%).
Products containing calcium and other multivalent cations are likely to interfere with absorption of alendronate.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Harderian gland (a retro-orbital gland not present in humans) adenomas were increased in high-dose female mice (p=0.003) in a 92-week oral carcinogenicity study at doses of alendronate of 1, 3, and 10 mg/kg/day (males) or 1, 2, and 5 mg/kg/day (females). These doses are equivalent to approximately 0.1 to 1 times the highest recommended clinical daily dose of 40 mg based on surface area, mg/m2. The relevance of this finding to humans is unknown.
Parafollicular cell (thyroid) adenomas were increased in high-dose male rats (p=0.003) in a 2-year oral carcinogenicity study at doses of 1 and 3.75 mg/kg body weight. These doses are equivalent to approximately 0.3 and 1 times the 40 mg clinical daily dose based on surface area, mg/m2. The relevance of this finding to humans is unknown.
Alendronate was not genotoxic in the in vitro microbial mutagenesis assay with and without metabolic activation, in an in vitro mammalian cell mutagenesis assay, in an in vitro alkaline elution assay in rat hepatocytes, and in an in vivo chromosomal aberration assay in mice. In an in vitro chromosomal aberration assay in Chinese hamster ovary cells, however, alendronate gave equivocal results.
Alendronate had no effect on fertility (male or female) in rats at oral doses up to 5 mg/kg/day (approximately 1 times the 40 mg clinical daily dose based on surface area, mg/m2).
13.2 Animal Toxicology and/or Pharmacology
The relative inhibitory activities on bone resorption and mineralization of alendronate and etidronate were compared in the Schenk assay, which is based on histological examination of the epiphyses of growing rats. In this assay, the lowest dose of alendronate that interfered with bone mineralization (leading to osteomalacia) was 6000-fold the antiresorptive dose. The corresponding ratio for etidronate was one to one. These data suggest that alendronate administered in therapeutic doses is highly unlikely to induce osteomalacia.
14 CLINICAL STUDIES
14.1 Treatment of Osteoporosis in Postmenopausal Women
Daily Dosing
The efficacy of FOSAMAX 10 mg daily was assessed in four clinical trials. Study 1, a three-year, multicenter, double-blind, placebo-controlled, US clinical study enrolled 478 patients with a BMD T-score at or below minus 2.5 with or without a prior vertebral fracture; Study 2, a three-year, multicenter, double-blind, placebo-controlled Multinational clinical study enrolled 516 patients with a BMD T-score at or below minus 2.5 with or without a prior vertebral fracture; Study 3, the Three-Year Study of the Fracture Intervention Trial (FIT) a study which enrolled 2027 postmenopausal patients with at least one baseline vertebral fracture; and Study 4, the Four-Year Study of FIT: a study which enrolled 4432 postmenopausal patients with low bone mass but without a baseline vertebral fracture.
Effect on Fracture Incidence
To assess the effects of FOSAMAX on the incidence of vertebral fractures (detected by digitized radiography; approximately one third of these were clinically symptomatic), the U.S. and Multinational studies were combined in an analysis that compared placebo to the pooled dosage groups of FOSAMAX (5 or 10 mg for three years or 20 mg for two years followed by 5 mg for one year). There was a statistically significant reduction in the proportion of patients treated with FOSAMAX experiencing one or more new vertebral fractures relative to those treated with placebo (3.2% vs. 6.2%; a 48% relative risk reduction). A reduction in the total number of new vertebral fractures (4.2 vs. 11.3 per 100 patients) was also observed. In the pooled analysis, patients who received FOSAMAX had a loss in stature that was statistically significantly less than was observed in those who received placebo (-3.0 mm vs. -4.6 mm).
The Fracture Intervention Trial (FIT) consisted of two studies in postmenopausal women: the Three-Year Study of patients who had at least one baseline radiographic vertebral fracture and the Four-Year Study of patients with low bone mass but without a baseline vertebral fracture. In both studies of FIT, 96% of randomized patients completed the studies (i.e., had a closeout visit at the scheduled end of the study); approximately 80% of patients were still taking study medication upon completion.
Fracture Intervention Trial: Three-Year Study (patients with at least one baseline radiographic vertebral fracture)
This randomized, double-blind, placebo-controlled, 2027-patient study (FOSAMAX, n=1022; placebo, n=1005) demonstrated that treatment with FOSAMAX resulted in statistically significant reductions in fracture incidence at three years as shown in Table 6.
| Percent of Patients | ||||
|---|---|---|---|---|
| FOSAMAX (n=1022) | Placebo (n=1005) | Absolute Reduction in Fracture Incidence | Relative Reduction in Fracture Risk % |
|
| Patients with: | ||||
| Vertebral fractures (diagnosed by X-ray)* | ||||
| ≥1 new vertebral fracture | 7.9 | 15.0 | 7.1 | 47† |
| ≥2 new vertebral fractures | 0.5 | 4.9 | 4.4 | 90† |
| Clinical (symptomatic) fractures | ||||
| Any clinical (symptomatic) fracture | 13.8 | 18.1 | 4.3 | 26‡ |
| ≥1 clinical (symptomatic) vertebral fracture | 2.3 | 5.0 | 2.7 | 54§ |
| Hip fracture | 1.1 | 2.2 | 1.1 | 51¶ |
| Wrist (forearm) fracture | 2.2 | 4.1 | 1.9 | 48¶ |
Furthermore, in this population of patients with baseline vertebral fracture, treatment with FOSAMAX significantly reduced the incidence of hospitalizations (25.0% vs. 30.7%).
In the Three-Year Study of FIT, fractures of the hip occurred in 22 (2.2%) of 1005 patients on placebo and 11 (1.1%) of 1022 patients on FOSAMAX, p=0.047. Figure 1 displays the cumulative incidence of hip fractures in this study.
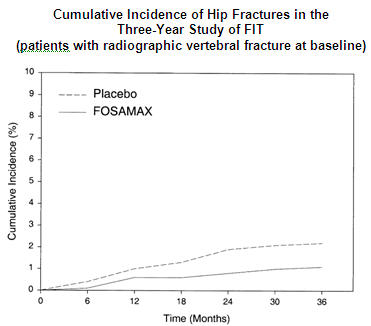
Fracture Intervention Trial: Four-Year Study (patients with low bone mass but without a baseline radiographic vertebral fracture)
This randomized, double-blind, placebo-controlled, 4432-patient study (FOSAMAX, n=2214; placebo, n=2218) further investigated the reduction in fracture incidence due to FOSAMAX. The intent of the study was to recruit women with osteoporosis, defined as a baseline femoral neck BMD at least two standard deviations below the mean for young adult women. However, due to subsequent revisions to the normative values for femoral neck BMD, 31% of patients were found not to meet this entry criterion and thus this study included both osteoporotic and non-osteoporotic women. The results are shown in Table 7 for the patients with osteoporosis.
| Percent of Patients | ||||
|---|---|---|---|---|
| FOSAMAX (n=1545) | Placebo (n=1521) | Absolute Reduction in Fracture Incidence | Relative Reduction in Fracture Risk (%) |
|
| Patients with: | ||||
| Vertebral fractures (diagnosed by X-ray)† | ||||
| ≥1 new vertebral fracture | 2.5 | 4.8 | 2.3 | 48‡ |
| ≥2 new vertebral fractures | 0.1 | 0.6 | 0.5 | 78§ |
| Clinical (symptomatic) fractures | ||||
| Any clinical (symptomatic) fracture | 12.9 | 16.2 | 3.3 | 22¶ |
| ≥1 clinical (symptomatic) vertebral fracture | 1.0 | 1.6 | 0.6 | 41 (NS)# |
| Hip fracture | 1.0 | 1.4 | 0.4 | 29 (NS)# |
| Wrist (forearm) fracture | 3.9 | 3.8 | -0.1 | NS# |
Fracture Results Across Studies
In the Three-Year Study of FIT, FOSAMAX reduced the percentage of women experiencing at least one new radiographic vertebral fracture from 15.0% to 7.9% (47% relative risk reduction, p<0.001); in the Four-Year Study of FIT, the percentage was reduced from 3.8% to 2.1% (44% relative risk reduction, p=0.001); and in the combined U.S./Multinational studies, from 6.2% to 3.2% (48% relative risk reduction, p=0.034).
FOSAMAX reduced the percentage of women experiencing multiple (two or more) new vertebral fractures from 4.2% to 0.6% (87% relative risk reduction, p<0.001) in the combined U.S./Multinational studies and from 4.9% to 0.5% (90% relative risk reduction, p<0.001) in the Three-Year Study of FIT. In the Four-Year Study of FIT, FOSAMAX reduced the percentage of osteoporotic women experiencing multiple vertebral fractures from 0.6% to 0.1% (78% relative risk reduction, p=0.035).
Thus, FOSAMAX reduced the incidence of radiographic vertebral fractures in osteoporotic women whether or not they had a previous radiographic vertebral fracture.
Effect on Bone Mineral Density
The bone mineral density efficacy of FOSAMAX 10 mg once daily in postmenopausal women, 44 to 84 years of age, with osteoporosis (lumbar spine bone mineral density [BMD] of at least 2 standard deviations below the premenopausal mean) was demonstrated in four double-blind, placebo-controlled clinical studies of two or three years' duration.
Figure 2 shows the mean increases in BMD of the lumbar spine, femoral neck, and trochanter in patients receiving FOSAMAX 10 mg/day relative to placebo-treated patients at three years for each of these studies.
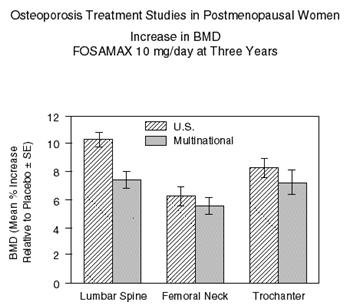
At three years significant increases in BMD, relative both to baseline and placebo, were seen at each measurement site in each study in patients who received FOSAMAX 10 mg/day. Total body BMD also increased significantly in each study, suggesting that the increases in bone mass of the spine and hip did not occur at the expense of other skeletal sites. Increases in BMD were evident as early as three months and continued throughout the three years of treatment. (See Figure 3 for lumbar spine results.) In the two-year extension of these studies, treatment of 147 patients with FOSAMAX 10 mg/day resulted in continued increases in BMD at the lumbar spine and trochanter (absolute additional increases between years 3 and 5: lumbar spine, 0.94%; trochanter, 0.88%). BMD at the femoral neck, forearm and total body were maintained. FOSAMAX was similarly effective regardless of age, race, baseline rate of bone turnover, and baseline BMD in the range studied (at least 2 standard deviations below the premenopausal mean).
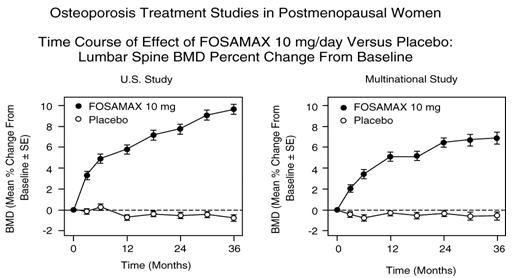
In patients with postmenopausal osteoporosis treated with FOSAMAX 10 mg/day for one or two years, the effects of treatment withdrawal were assessed. Following discontinuation, there were no further increases in bone mass and the rates of bone loss were similar to those of the placebo groups.
Bone Histology
Bone histology in 270 postmenopausal patients with osteoporosis treated with FOSAMAX at doses ranging from 1 to 20 mg/day for one, two, or three years revealed normal mineralization and structure, as well as the expected decrease in bone turnover relative to placebo. These data, together with the normal bone histology and increased bone strength observed in rats and baboons exposed to long-term alendronate treatment, support the conclusion that bone formed during therapy with FOSAMAX is of normal quality.
Effect on Height
FOSAMAX, over a three- or four-year period, was associated with statistically significant reductions in loss of height vs. placebo in patients with and without baseline radiographic vertebral fractures. At the end of the FIT studies, the between-treatment group differences were 3.2 mm in the Three-Year Study and 1.3 mm in the Four-Year Study.
Weekly Dosing
The therapeutic equivalence of once-weekly FOSAMAX 70 mg (n=519) and FOSAMAX 10 mg daily (n=370) was demonstrated in a one-year, double-blind, multicenter study of postmenopausal women with osteoporosis. In the primary analysis of completers, the mean increases from baseline in lumbar spine BMD at one year were 5.1% (4.8, 5.4%; 95% CI) in the 70-mg once-weekly group (n=440) and 5.4% (5.0, 5.8%; 95% CI) in the 10-mg daily group (n=330). The two treatment groups were also similar with regard to BMD increases at other skeletal sites. The results of the intention-to-treat analysis were consistent with the primary analysis of completers.
Concomitant Use with Estrogen/Hormone Replacement Therapy (HRT)
The effects on BMD of treatment with FOSAMAX 10 mg once daily and conjugated estrogen (0.625 mg/day) either alone or in combination were assessed in a two-year, double-blind, placebo-controlled study of hysterectomized postmenopausal osteoporotic women (n=425). At two years, the increases in lumbar spine BMD from baseline were significantly greater with the combination (8.3%) than with either estrogen or FOSAMAX alone (both 6.0%).
The effects on BMD when FOSAMAX was added to stable doses (for at least one year) of HRT (estrogen ± progestin) were assessed in a one-year, double-blind, placebo-controlled study in postmenopausal osteoporotic women (n=428). The addition of FOSAMAX 10 mg once daily to HRT produced, at one year, significantly greater increases in lumbar spine BMD (3.7%) vs. HRT alone (1.1%).
In these studies, significant increases or favorable trends in BMD for combined therapy compared with HRT alone were seen at the total hip, femoral neck, and trochanter. No significant effect was seen for total body BMD.
Histomorphometric studies of transiliac biopsies in 92 subjects showed normal bone architecture. Compared to placebo there was a 98% suppression of bone turnover (as assessed by mineralizing surface) after 18 months of combined treatment with FOSAMAX and HRT, 94% on FOSAMAX alone, and 78% on HRT alone. The long-term effects of combined FOSAMAX and HRT on fracture occurrence and fracture healing have not been studied.
14.2 Prevention of Osteoporosis in Postmenopausal Women
Daily Dosing
Prevention of bone loss was demonstrated in two double-blind, placebo-controlled studies of postmenopausal women 40-60 years of age. One thousand six hundred nine patients (FOSAMAX 5 mg/day; n=498) who were at least six months postmenopausal were entered into a two-year study without regard to their baseline BMD. In the other study, 447 patients (FOSAMAX 5 mg/day; n=88), who were between six months and three years postmenopause, were treated for up to three years. In the placebo-treated patients BMD losses of approximately 1% per year were seen at the spine, hip (femoral neck and trochanter) and total body. In contrast, FOSAMAX 5 mg/day prevented bone loss in the majority of patients and induced significant increases in mean bone mass at each of these sites (see Figure 4). In addition, FOSAMAX 5 mg/day reduced the rate of bone loss at the forearm by approximately half relative to placebo. FOSAMAX 5 mg/day was similarly effective in this population regardless of age, time since menopause, race and baseline rate of bone turnover.
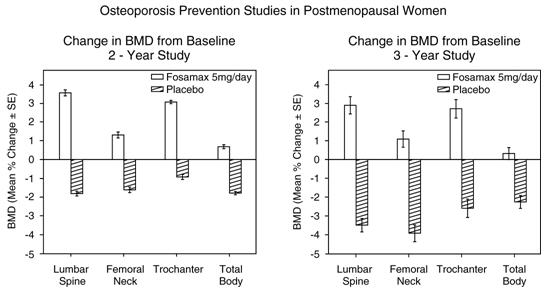
Weekly Dosing
The therapeutic equivalence of once weekly FOSAMAX 35 mg (n=362) and FOSAMAX 5 mg daily (n=361) was demonstrated in a one-year, double-blind, multicenter study of postmenopausal women without osteoporosis. In the primary analysis of completers, the mean increases from baseline in lumbar spine BMD at one year were 2.9% (2.6, 3.2%; 95% CI) in the 35-mg once-weekly group (n=307) and 3.2% (2.9, 3.5%; 95% CI) in the 5-mg daily group (n=298). The two treatment groups were also similar with regard to BMD increases at other skeletal sites. The results of the intention-to-treat analysis were consistent with the primary analysis of completers.
14.3 Treatment to Increase Bone Mass in Men with Osteoporosis
The efficacy of FOSAMAX in men with hypogonadal or idiopathic osteoporosis was demonstrated in two clinical studies.
Daily Dosing
A two-year, double-blind, placebo-controlled, multicenter study of FOSAMAX 10 mg once daily enrolled a total of 241 men between the ages of 31 and 87 (mean, 63). All patients in the trial had either a BMD T-score less than or equal to -2 at the femoral neck and less than or equal to -1 at the lumbar spine, or a baseline osteoporotic fracture and a BMD T-score less than or equal to -1 at the femoral neck. At two years, the mean increases relative to placebo in BMD in men receiving FOSAMAX 10 mg/day were significant at the following sites: lumbar spine, 5.3%; femoral neck, 2.6%; trochanter, 3.1%; and total body, 1.6%. Treatment with FOSAMAX also reduced height loss (FOSAMAX, -0.6 mm vs. placebo, -2.4 mm).
Weekly Dosing
A one-year, double-blind, placebo-controlled, multicenter study of once weekly FOSAMAX 70 mg enrolled a total of 167 men between the ages of 38 and 91 (mean, 66). Patients in the study had either a BMD T-score less than or equal to -2 at the femoral neck and less than or equal to -1 at the lumbar spine, or a BMD T-score less than or equal to -2 at the lumbar spine and less than or equal to -1 at the femoral neck, or a baseline osteoporotic fracture and a BMD T-score less than or equal to -1 at the femoral neck. At one year, the mean increases relative to placebo in BMD in men receiving FOSAMAX 70 mg once weekly were significant at the following sites: lumbar spine, 2.8%; femoral neck, 1.9%; trochanter, 2.0%; and total body, 1.2%. These increases in BMD were similar to those seen at one year in the 10 mg once-daily study.
In both studies, BMD responses were similar regardless of age (greater than or equal to 65 years vs. less than 65 years), gonadal function (baseline testosterone less than 9 ng/dL vs. greater than or equal to 9 ng/dL), or baseline BMD (femoral neck and lumbar spine T-score less than or equal to -2.5 vs. greater than -2.5).
14.4 Treatment of Glucocorticoid-Induced Osteoporosis
The efficacy of FOSAMAX 5 and 10 mg once daily in men and women receiving glucocorticoids (at least 7.5 mg/day of prednisone or equivalent) was demonstrated in two, one-year, double-blind, randomized, placebo-controlled, multicenter studies of virtually identical design, one performed in the United States and the other in 15 different countries (Multinational [which also included FOSAMAX 2.5 mg/day]). These studies enrolled 232 and 328 patients, respectively, between the ages of 17 and 83 with a variety of glucocorticoid-requiring diseases. Patients received supplemental calcium and vitamin D. Figure 5 shows the mean increases relative to placebo in BMD of the lumbar spine, femoral neck, and trochanter in patients receiving FOSAMAX 5 mg/day for each study.
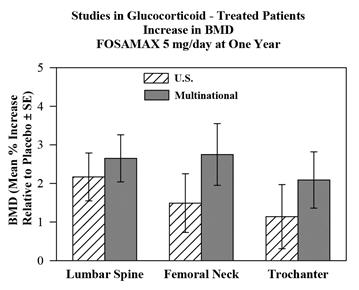
After one year, significant increases relative to placebo in BMD were seen in the combined studies at each of these sites in patients who received FOSAMAX 5 mg/day. In the placebo-treated patients, a significant decrease in BMD occurred at the femoral neck (-1.2%), and smaller decreases were seen at the lumbar spine and trochanter. Total body BMD was maintained with FOSAMAX 5 mg/day. The increases in BMD with FOSAMAX 10 mg/day were similar to those with FOSAMAX 5 mg/day in all patients except for postmenopausal women not receiving estrogen therapy. In these women, the increases (relative to placebo) with FOSAMAX 10 mg/day were greater than those with FOSAMAX 5 mg/day at the lumbar spine (4.1% vs. 1.6%) and trochanter (2.8% vs. 1.7%), but not at other sites. FOSAMAX was effective regardless of dose or duration of glucocorticoid use. In addition, FOSAMAX was similarly effective regardless of age (less than 65 vs. greater than or equal to 65 years), race (Caucasian vs. other races), gender, underlying disease, baseline BMD, baseline bone turnover, and use with a variety of common medications.
Bone histology was normal in the 49 patients biopsied at the end of one year who received FOSAMAX at doses of up to 10 mg/day.
Of the original 560 patients in these studies, 208 patients who remained on at least 7.5 mg/day of prednisone or equivalent continued into a one-year double-blind extension. After two years of treatment, spine BMD increased by 3.7% and 5.0% relative to placebo with FOSAMAX 5 and 10 mg/day, respectively. Significant increases in BMD (relative to placebo) were also observed at the femoral neck, trochanter, and total body.
After one year, 2.3% of patients treated with FOSAMAX 5 or 10 mg/day (pooled) vs. 3.7% of those treated with placebo experienced a new vertebral fracture (not significant). However, in the population studied for two years, treatment with FOSAMAX (pooled dosage groups: 5 or 10 mg for two years or 2.5 mg for one year followed by 10 mg for one year) significantly reduced the incidence of patients with a new vertebral fracture (FOSAMAX 0.7% vs. placebo 6.8%).
14.5 Treatment of Paget's Disease of Bone
The efficacy of FOSAMAX 40 mg once daily for six months was demonstrated in two double-blind clinical studies of male and female patients with moderate to severe Paget's disease (alkaline phosphatase at least twice the upper limit of normal): a placebo-controlled, multinational study and a U.S. comparative study with etidronate disodium 400 mg/day. Figure 6 shows the mean percent changes from baseline in serum alkaline phosphatase for up to six months of randomized treatment.
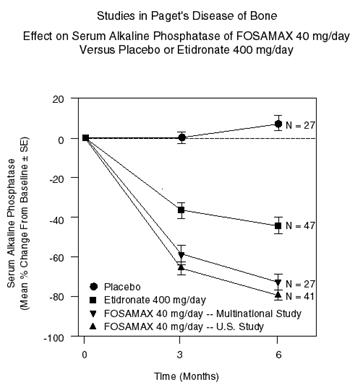
At six months the suppression in alkaline phosphatase in patients treated with FOSAMAX was significantly greater than that achieved with etidronate and contrasted with the complete lack of response in placebo-treated patients. Response (defined as either normalization of serum alkaline phosphatase or decrease from baseline greater than or equal to 60%) occurred in approximately 85% of patients treated with FOSAMAX in the combined studies vs. 30% in the etidronate group and 0% in the placebo group. FOSAMAX was similarly effective regardless of age, gender, race, prior use of other bisphosphonates, or baseline alkaline phosphatase within the range studied (at least twice the upper limit of normal).
Bone histology was evaluated in 33 patients with Paget's disease treated with FOSAMAX 40 mg/day for 6 months. As in patients treated for osteoporosis [see Clinical Studies (14.1)], FOSAMAX did not impair mineralization, and the expected decrease in the rate of bone turnover was observed. Normal lamellar bone was produced during treatment with FOSAMAX, even where preexisting bone was woven and disorganized. Overall, bone histology data support the conclusion that bone formed during treatment with FOSAMAX is of normal quality.
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Instruct patients to read the Medication Guide before starting therapy with FOSAMAX and to reread it each time the prescription is renewed.
17.1 Osteoporosis Recommendations, Including Calcium and Vitamin D Supplementation
Instruct patients to take supplemental calcium and vitamin D, if daily dietary intake is inadequate. Weight-bearing exercise should be considered along with the modification of certain behavioral factors, such as cigarette smoking and/or excessive alcohol consumption, if these factors exist.
17.2 Dosing Instructions
Instruct patients that the expected benefits of FOSAMAX may only be obtained when it is taken with plain water the first thing upon arising for the day at least 30 minutes before the first food, beverage, or medication of the day. Even dosing with orange juice or coffee has been shown to markedly reduce the absorption of FOSAMAX [see Clinical Pharmacology (12.3)].
Instruct patients not to chew or suck on the tablet because of a potential for oropharyngeal ulceration.
Instruct patients to swallow each tablet of FOSAMAX with a full glass of water (6-8 ounces) to facilitate delivery to the stomach and thus reduce the potential for esophageal irritation. Instruct patients to drink at least 2 ounces (a quarter of a cup) of water after taking FOSAMAX oral solution, to facilitate gastric emptying.
Instruct patients not to lie down for at least 30 minutes and until after their first food of the day.
Instruct patients not to take FOSAMAX at bedtime or before arising for the day. Patients should be informed that failure to follow these instructions may increase their risk of esophageal problems.
Instruct patients that if they develop symptoms of esophageal disease (such as difficulty or pain upon swallowing, retrosternal pain or new or worsening heartburn) they should stop taking FOSAMAX and consult their physician.
If patients miss a dose of once weekly FOSAMAX, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day.
Distributed by: Merck Sharp & Dohme Corp., a subsidiary of
MERCK & CO., INC., Whitehouse Station, NJ 08889, USA
For patent information: www.merck.com/product/patent/home.html
Copyright © 1995-2019 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.
All rights reserved.
uspi-mk0217-mf-1908r026
MEDICATION GUIDE
FOSAMAX® (FOSS-ah-max)
(alendronate sodium)
Tablets
Read the Medication Guide that comes with FOSAMAX® before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. Talk to your doctor if you have any questions about FOSAMAX.
What is the most important information I should know about FOSAMAX?
FOSAMAX can cause serious side effects including:
- 1. Esophagus problems
- 2. Low calcium levels in your blood (hypocalcemia)
- 3. Bone, joint, or muscle pain
- 4. Severe jaw bone problems (osteonecrosis)
- 5. Unusual thigh bone fractures
- 1.
-
Esophagus problems.
Some people who take FOSAMAX may develop problems in the esophagus (the tube that connects the mouth and the stomach). These problems include irritation, inflammation, or ulcers of the esophagus which may sometimes bleed.- It is important that you take FOSAMAX exactly as prescribed to help lower your chance of getting esophagus problems. (See the section "How should I take FOSAMAX?")
- Stop taking FOSAMAX and call your doctor right away if you get chest pain, new or worsening heartburn, or have trouble or pain when you swallow.
- 2.
-
Low calcium levels in your blood (hypocalcemia).
FOSAMAX may lower the calcium levels in your blood. If you have low blood calcium before you start taking FOSAMAX, it may get worse during treatment. Your low blood calcium must be treated before you take FOSAMAX. Most people with low blood calcium levels do not have symptoms, but some people may have symptoms. Call your doctor right away if you have symptoms of low blood calcium such as:- Spasms, twitches, or cramps in your muscles
- Numbness or tingling in your fingers, toes, or around your mouth
- 3.
-
Bone, joint, or muscle pain.
Some people who take FOSAMAX develop severe bone, joint, or muscle pain. - 4.
-
Severe jaw bone problems (osteonecrosis).
Severe jaw bone problems may happen when you take FOSAMAX. Your doctor should examine your mouth before you start FOSAMAX. Your doctor may tell you to see your dentist before you start FOSAMAX. It is important for you to practice good mouth care during treatment with FOSAMAX. - 5.
-
Unusual thigh bone fractures.
Some people have developed unusual fractures in their thigh bone. Symptoms of a fracture may include new or unusual pain in your hip, groin, or thigh.
Call your doctor right away if you have any of these side effects.
What is FOSAMAX?
FOSAMAX is a prescription medicine used to:
- Treat or prevent osteoporosis in women after menopause. It helps reduce the chance of having a hip or spinal fracture (break).
- Increase bone mass in men with osteoporosis.
- Treat osteoporosis in either men or women who are taking corticosteroid medicines.
- Treat certain men and women who have Paget's disease of the bone.
It is not known how long FOSAMAX works for the treatment and prevention of osteoporosis. You should see your doctor regularly to determine if FOSAMAX is still right for you.
FOSAMAX is not for use in children.
Who should not take FOSAMAX?
Do not take FOSAMAX if you:
- Have certain problems with your esophagus, the tube that connects your mouth with your stomach
- Cannot stand or sit upright for at least 30 minutes
- Have low levels of calcium in your blood
- Are allergic to FOSAMAX or any of its ingredients. A list of ingredients is at the end of this leaflet.
What should I tell my doctor before taking FOSAMAX?
Before you start FOSAMAX, be sure to talk to your doctor if you:
- Have problems with swallowing
- Have stomach or digestive problems
- Have low blood calcium
- Plan to have dental surgery or teeth removed
- Have kidney problems
- Have been told you have trouble absorbing minerals in your stomach or intestines (malabsorption syndrome)
- Are pregnant, trying to become pregnant or suspect that you are pregnant. If you become pregnant while taking FOSAMAX, stop taking it and contact your doctor. It is not known if FOSAMAX can harm your unborn baby.
- Are breast-feeding or plan to breast-feed. It is not known if FOSAMAX passes into your milk and may harm your baby.
Especially tell your doctor if you take:
- antacids
- aspirin
- Nonsteroidal Anti-Inflammatory (NSAID) medicines
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Certain medicines may affect how FOSAMAX works.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist each time you get a new medicine.
- Take FOSAMAX exactly as your doctor tells you.
- FOSAMAX works only if taken on an empty stomach.
- Take FOSAMAX, after you get up for the day and before taking your first food, drink, or other medicine.
- Take FOSAMAX while you are sitting or standing.
- Do not chew or suck on a tablet of FOSAMAX.
- Swallow FOSAMAX tablet with a full glass (6-8 oz) of plain water only.
-
Do not take FOSAMAX with mineral water, coffee, tea, soda, or juice.
- If you take Alendronate Daily:
- Take 1 alendronate tablet one time a day, every day after you get up for the day and before taking your first food, drink, or other medicine.
- If you take Once Weekly FOSAMAX:
- Choose the day of the week that best fits your schedule.
- Take 1 dose of FOSAMAX every week on your chosen day after you get up for the day and before taking your first food, drink, or other medicine.
- If you take Alendronate Daily:
After swallowing FOSAMAX tablet, wait at least 30 minutes:
- Before you lie down. You may sit, stand or walk, and do normal activities like reading.
- Before you take your first food or drink except for plain water.
- Before you take other medicines, including antacids, calcium, and other supplements and vitamins.
Do not lie down for at least 30 minutes after you take FOSAMAX and after you eat your first food of the day.
If you miss a dose of FOSAMAX, do not take it later in the day. Take your missed dose on the next morning after you remember and then return to your normal schedule. Do not take 2 doses on the same day.
If you take too much FOSAMAX, call your doctor. Do not try to vomit. Do not lie down.
What are the possible side effects of FOSAMAX?
FOSAMAX may cause serious side effects.
The most common side effects of FOSAMAX are:
- Stomach area (abdominal) pain
- Heartburn
- Constipation
- Diarrhea
- Upset stomach
- Pain in your bones, joints, or muscles
- Nausea
You may get allergic reactions, such as hives or swelling of your face, lips, tongue, or throat.
Worsening of asthma has been reported.
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of FOSAMAX. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I store FOSAMAX?
- Store FOSAMAX at room temperature, 59°F to 86°F (15°C to 30°C).
- Keep FOSAMAX in a tightly closed container.
Keep FOSAMAX and all medicines out of the reach of children.
General information about the safe and effective use of FOSAMAX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use FOSAMAX for a condition for which it was not prescribed. Do not give FOSAMAX to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about FOSAMAX. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about FOSAMAX that is written for health professionals. For more information, go to: www.FOSAMAX.com or call 1-800-622-4477 (toll-free).
What are the ingredients in FOSAMAX?
Tablets:
Active ingredient: alendronate sodium
Inactive ingredients: microcrystalline cellulose, anhydrous lactose, croscarmellose sodium, magnesium stearate.
Distributed by: Merck Sharp & Dohme Corp., a subsidiary of
MERCK & CO., INC., Whitehouse Station, NJ 08889, USA
For patent information: www.merck.com/product/patent/home.html
Copyright © 2010-2019 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.
All rights reserved.
Revised: 08/2019
usmg-mk0217-mf-1908r019
This Medication Guide has been approved by the U.S. Food and Drug Administration.
PRINCIPAL DISPLAY PANEL - 4 Tablet Blister Pack
ONCE WEEKLY
FOSAMAX® 70 mg
(Alendronate SodiumTablets)
Dispense the enclosed
Medication Guide
to each patient.
Each tablet contains
91.37 mg
alendronate sodium
(70 mg free acid
equivalent)
4 Tablets
Dist. by:
Merck Sharp & Dohme Corp.,
a subsidiary of
MERCK & CO., INC.
Whitehouse Station, NJ 08889, USA
Manuf. by:
Aesica Pharmaceuticals GmbH
Galileistraβe 6, 08056 Zwickau, Germany
Alendronate Sodium(active ingred.) Made in Ireland.
Formulated in Germany.
Copyright © 2015 Merck Sharp & Dohme Corp.,
a subsidiary of Merck & Co., Inc.
All rights reserved.
Rx only
USUAL ADULT
DOSAGE:
ONE 70 mg TABLET
ONCE WEEKLY
See accompanying
circular for complete
dosage information.

| FOSAMAX
alendronate sodium tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Merck Sharp & Dohme Corp. (001317601) |