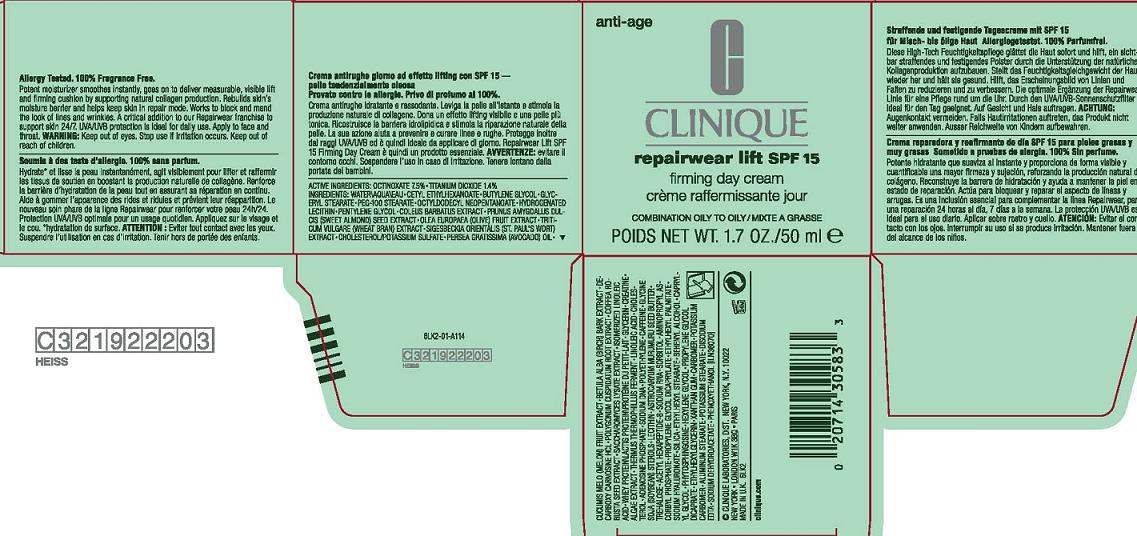
Label: REPAIRWEAR LIFT SPF 15 FIRMING DAY COMBO TO OILY SKIN- octinoxate, titanium dioxide cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 49527-009-01, 49527-009-02 - Packager: CLINIQUE LABORATORIES INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 17, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
ingredients: water [] cetyl ethylhexanoate [] butylene glycol [] glyceryl stearate [] peg-100 stearate [] octyldodecyl neopentanoate [] hydrogenated lecithin [] pentylene glycol [] coleus barbatus extract [] prunus amygdalus dulcis (sweet almond) seed extract [] olea europaea (olive) fruit extract [] triticum vulgare (wheat bran) extract [] sigesbeckia orientalis extract [] cholesterol/potassium sulfate [] persea gratissima (avocado) oil [] cucumis melo (melon) fruit extract [] betula alba (birch) bark extract [] decarboxy carnosine hcl [] polygonum cuspidatum root extract [] coffea robusta seed extract [] saccharomyces lysate extract [] isomerized linoleic acid [] whey protein [] glycerin [] creatine [] algae extract [] thermus thermophillus ferment [] linoleic acid [] cholesterol [] adenosine phosphate [] sodium dna [] polyethylene [] caffeine [] glycine soja (soybean) sterols [] lecithin [] astrocaryum murumuru seed butter [] trehalose [] acetyl hexapeptide-8 [] sodium rna [] sorbitol [] aminopropyl ascorbyl phosphate [] propylene glycol dicaprylate [] ethylhexyl palmitate [] sodium hyaluronate [] silica [] ethylhexyl stearate [] behenyl alcohol [] caprylyl glycol [] phytosphingosine [] hexylene glycol [] propylene glycol dicaprate [] ethylhexylglycerin [] xanthan gum [] carbomer [] potassium carbomer [] aluminum stearate [] potassium stearate [] disodium edta [] sodium dehydroacetate [] phenoxyethanol
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
REPAIRWEAR LIFT SPF 15 FIRMING DAY COMBO TO OILY SKIN
octinoxate, titanium dioxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-009 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 mL in 100 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 1.4 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PROPYLENE GLYCOL MONOSTEARATE (UNII: F76354LMGR) GLYCERIN (UNII: PDC6A3C0OX) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) PENTYLENE GLYCOL (UNII: 50C1307PZG) OLIVE OIL (UNII: 6UYK2W1W1E) POTASSIUM SULFATE (UNII: 1K573LC5TV) CHOLESTEROL (UNII: 97C5T2UQ7J) AVOCADO OIL (UNII: 6VNO72PFC1) CUCUMBER JUICE (UNII: 61845009SP) POLYGONUM CUSPIDATUM ROOT (UNII: 7TRV45YZF7) SACCHAROMYCES LYSATE (UNII: R85W246Z1C) CREATINE (UNII: MU72812GK0) ADENOSINE PHOSPHATE (UNII: 415SHH325A) CAFFEINE (UNII: 3G6A5W338E) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) TREHALOSE (UNII: B8WCK70T7I) SORBITOL (UNII: 506T60A25R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CAPRYLYL GLYCOL (UNII: 00YIU5438U) HEXYLENE GLYCOL (UNII: KEH0A3F75J) XANTHAN GUM (UNII: TTV12P4NEE) CARBOMER HOMOPOLYMER TYPE C (UNII: 4Q93RCW27E) EDETATE DISODIUM (UNII: 7FLD91C86K) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-009-01 1 in 1 CARTON 1 NDC:49527-009-02 50 mL in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 12/01/2010 Labeler - CLINIQUE LABORATORIES INC (173047747) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD 205952385 manufacture Establishment Name Address ID/FEI Business Operations ESTEE LAUDER N.V. 370151326 manufacture Establishment Name Address ID/FEI Business Operations Len-Ron Manufacturing Division of Aramis Inc. 809771152 manufacture Establishment Name Address ID/FEI Business Operations Aramis Inc. 042918826 manufacture Establishment Name Address ID/FEI Business Operations Northtec Bristol 949264774 manufacture, relabel, repack Establishment Name Address ID/FEI Business Operations Northtec Keystone 618107429 manufacture, relabel, repack Establishment Name Address ID/FEI Business Operations Estee Lauder Pennsylvania Distribution Center 2 828534516 manufacture, relabel, repack Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics, Ltd. 255175580 manufacture Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics, Ltd 253616536 manufacture Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Distribution Center 208579636 repack, relabel Establishment Name Address ID/FEI Business Operations Estee Lauder Kabushiki Kaisha 712808195 relabel, repack Establishment Name Address ID/FEI Business Operations Whitman Laboratories Ltd. 216866277 manufacture Establishment Name Address ID/FEI Business Operations Aveda Corporation 071352058 manufacture