Label: TORBUGESIC- butorphanol tartrate solution
- NDC Code(s): 54771-2033-1, 54771-2033-2
- Packager: Zoetis Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: CIV
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated March 20, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- CAUTION
-
DESCRIPTION
TORBUGESIC (butorphanol tartrate injection) is a totally synthetic, centrally acting, narcotic agonist-antagonist analgesic with potent antitussive activity. It is a member of the phenanthrene series. The chemical name is Morphinan-3, 14-diol, 17-(cyclobutylmethyl)-, (-)-, (S- (R*, R*))-2,3-dihydroxybutanedioate (1:1) (salt). It is a white, crystalline, water soluble substance having a molecular weight of 477.55; its molecular formula is C21H29NO2•C4H6O6.
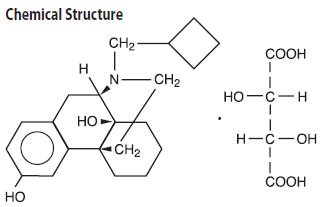
Chemical Structure
Each mL of TORBUGESIC contains 10 mg butorphanol base (as butorphanol tartrate), 3.3 mg citric acid, USP, 6.4 mg sodium citrate, 4.7 mg sodium chloride, and 0.1 mg benzethonium chloride, q.s. with water for injection.
-
CLINICAL PHARMACOLOGY
Comparative Pharmacology
In animals, butorphanol has been demonstrated to be 4 to 30 times more potent than morphine and pentazocine (Talwin®-V) respectively.1
In humans, butorphanol has been shown to have 5 to 7 times the analgesic activity of morphine and 20 times that of pentazocine.2,3
Butorphanol has 15 to 20 times the oral antitussive activity of codeine or dextromethorphan in dogs and guinea pigs.4
As an antagonist, butorphanol is approximately equivalent to nalorphine and 30 times more potent than pentazocine.1
Cardiopulmonary depressant effects are minimal after treatment with butorphanol as demonstrated in dogs5, humans6,7 and horses.8
Unlike classical narcotic agonist analgesics which are associated with decreases in blood pressure, reduction in heart rate and concomitant release of histamine, butorphanol does not cause histamine release.1
Furthermore, the cardiopulmonary effects of butorphanol are not distinctly dosage-related but rather reach a ceiling effect beyond which further
dosage increases result in relatively lesser effects.Reproduction: Studies performed in mice and rabbits revealed no evidence of impaired fertility or harm to the fetus due to butorphanol tartrate. In the female rat, parenteral administration was associated with increased nervousness and decreased care for the newborn, resulting in a decreased survival rate of the newborn. This nervousness was seen only in the rat species.
Equine Pharmacology
Following intravenous injection in horses, butorphanol is largely eliminated from the blood within 3 to 4 hours. The drug is extensively metabolized in the liver and excreted in the urine. In ponies, butorphanol given intramuscularly at a dosage of 0.22 mg/kg, was shown to alleviate experimentally induced visceral pain for about 4 hours.9
In horses, intravenous dosages of butorphanol ranging from 0.05 to 0.4 mg/kg were shown to be effective in alleviating visceral and superficial pain for at least 4 hours, as illustrated in the following figure:
Analgesic Effects of Butorphanol Given at
Various Dosages in Horses with Abdominal Pain*Pain threshold in butorphanol-treated colicky horses relative to placebo controls. 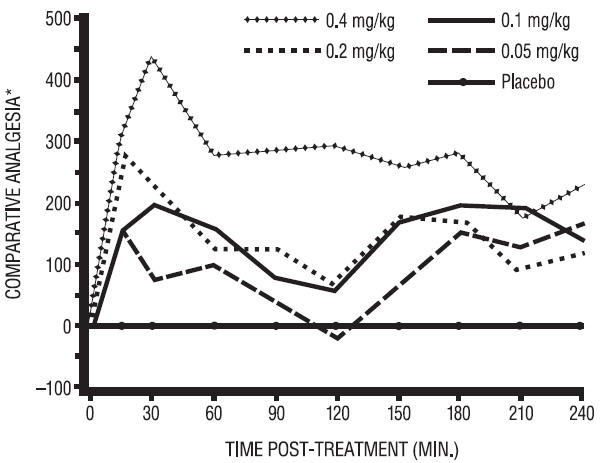
A definite dosage-response relationship was detected in that butorphanol dosage of 0.1 mg/kg was more effective than 0.05 mg/kg but not different from 0.2 mg/kg in alleviating deep abdominal pain.
Acute Equine Studies
Rapid intravenous administration of butorphanol at a dosage of 2 mg/kg (20 times the recommended dosage) to a previously unmedicated horse resulted in a brief episode of inability to stand, muscle fasciculation, a convulsive seizure of 6 seconds duration and recovery within three minutes. The same dosage administered after 10 successive daily 1 mg/kg dosages of butorphanol resulted only in transient sedative effects. During the 10-day course of administration at 1 mg/kg (10 times the recommended use level) in two horses, the only detectable drug effects were transient behavioral changes typical of narcotic agonist activity. These included muscle fasciculation about the head and neck, dysphoria, lateral nystagmus, ataxia and salivation.
Repeated administration of butorphanol at 1 mg/kg (10 times the recommended dose) every four hours for 48 hours caused constipation in one of two horses.
Subacute Equine Studies
Horses were found to tolerate butorphanol given intravenously at dosages of 0.1, 0.3 and 0.5 mg/kg every 4 hours for 48 hours followed by once daily injections for a total of 21 days. The only detectable drug effects were slight transient ataxia observed occasionally in the high dosage group. No clinical, laboratory, or gross or histopathologic evidence of any butorphanol-related toxicity was encountered in the horses.
-
INDICATIONS
TORBUGESIC (butorphanol tartrate injection) is indicated for the relief of pain associated with colic in adult horses and yearlings. Clinical studies in the horse have shown that TORBUGESIC alleviates abdominal pain associated with torsion, impaction, intussusception, spasmodic and tympanic colic and postpartum pain.
- WARNINGS
- CAUTION
- ADVERSE REACTIONS
-
DOSAGE
The recommended dosage in the horse is 0.1 mg of butorphanol per kilogram of body weight (0.05 mg/lb) by intravenous injection. This is equivalent to 5 mL of TORBUGESIC for each 1000 lbs body weight.
The dose may be repeated within 3 to 4 hours but treatment should not exceed 48 hours.
Use contents within 4 months of first puncture. Pre-clinical model studies and clinical field trials in horses demonstrate that the analgesic effects of TORBUGESIC are seen within 15 minutes following injection and persist for about 4 hours.
-
HOW SUPPLIED
50 mL vials TORBUGESIC (butorphanol tartrate injection) Veterinary Injection, 10 mg base activity per mL.
10 mL vials TORBUGESIC (butorphanol tartrate injection) Veterinary Injection, 10 mg base activity per mL.
Store at controlled room temperature 20°-25°C (68°-77°F) with excursions between 15°-30°C (59°-86°F).
-
REFERENCES
1. Pircio, A.W. et al: “The Pharmacology of Butorphanol,” Arch. Int. Pharmacodyn. Ther. 220 (2): 231–257, 1976.
2. Dobkin, A.B. et al: “Butorphanol and Pentazocinein Patients with Severe Postoperative Pain,” Clin. Pharmacol. Ther. 18: 547–553, 1975.
3. Gilbert, M.S. et al: “Intramuscular Butorphanol and Meperidine in Postoperative Pain,” Clin. Pharmacol. Ther. 20: 359 –364, 1976.
4. Cavanagh, R.L. et al: “Antitussive Properties of Butorphanol,” Arch. Int. Pharmacodyn. Ther. 220 (2):258–268, 1976.
5. Schurig, J.E. et al: “Eff ect of Butorphanol and Morphine on Pulmonary Mechanics, Arterial Blood Pressure, and Venous Plasma Histamine in the Anesthetized Dog,” Arch. Int. Pharmacodyn. Ther. 233: 296–304, 1978.
6. Nagashmina, H. et al: “Respiratory and Circulatory Effects of Intravenous Butorphanol and Morphine,” Clin. Pharmacol. Ther. 19:735–745, 1976.
7. Popio, K.A. et al: “Hemodynamic and Respiratory Effects of Morphine and Butorphanol,” Clin. Pharmacol. Ther. 23: 281–287, 1978.
8. Robertson, J.T. and Muir, W.W.: “Cardiopulmonary Effects of Butorphanol Tartrate in Horses,” Am. J. Vet. Res. 42: 41–44, 1981.
9. Kalpravidh, M. et al: “Effects of Butorphanol, Flunixin, Levorphanol, Morphine, Pentazocine zine in Ponies,” Am. J. Vet. Res. 45:217–223, 1984. - SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 10 mL Label
- PRINCIPAL DISPLAY PANEL - 50 mL Label
-
INGREDIENTS AND APPEARANCE
TORBUGESIC
butorphanol tartrate solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:54771-2033 Route of Administration INTRAVENOUS DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BUTORPHANOL TARTRATE (UNII: 2L7I72RUHN) (BUTORPHANOL - UNII:QV897JC36D) BUTORPHANOL TARTRATE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 3.3 mg in 1 mL SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM CHLORIDE (UNII: 451W47IQ8X) BENZETHONIUM CHLORIDE (UNII: PH41D05744) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54771-2033-2 1 in 1 CARTON 1 50 mL in 1 VIAL 2 NDC:54771-2033-1 10 mL in 1 VIAL Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA135780 06/11/1985 Labeler - Zoetis Inc. (828851555)




