Label: KETOROCAINE L KIT- ketorolac tromethamine, lidocaine hydrochloride, povidine iodine kit
- NDC Code(s): 55150-158-72, 67777-419-02, 72266-118-01, 76420-771-01
- Packager: Asclemed USA, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 18, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING
Ketorolac tromethamine, a nonsteroidal anti-inflammatory drug (NSAID), is indicated for the short-term (up to 5 days in adults) management of moderately severe acute pain that requires analgesia at the opioid level. Oral ketorolac tromethamine is indicated only as continuation treatment following intravenous or intramuscular dosing of ketorolac tromethamine, if necessary. The total combined duration of use of oral ketorolac tromethamine and ketorolac tromethamine injection should not exceed 5 days.
Ketorolac tromethamine is not indicated for use in pediatric patients and it is NOT indicated for minor or chronic painful conditions. Increasing the dose of ketorolac tromethamine beyond the label recommendations will not provide better efficacy but will increase the risk of developing serious adverse events.
GASTROINTESTINAL RISK
• Ketorolac tromethamine can cause peptic ulcers, gastrointestinal bleeding and/or perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Therefore, ketorolac tromethamine is CONTRAINDICATED in patients with active peptic ulcer disease, in patients with recent gastrointestinal bleeding or perforation, and in patients with a history of peptic ulcer disease or gastrointestinal bleeding. Elderly patients are at greater risk for serious gastrointestinal events (see WARNINGS).
CARDIOVASCULAR THROMBOTIC EVENTS
• Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (see WARNINGSand PRECAUTIONS).
• Ketorolac tromethamine is CONTRAINDICATED in the setting of coronary artery bypass graft (CABG) surgery (see CONTRAINDICATIONSand WARNINGS).
RENAL RISK
• Ketorolac tromethamine is CONTRAINDICATED in patients with advanced renal impairment and in patients at risk for renal failure due to volume depletion (see WARNINGS).
RISK OF BLEEDING
• Ketorolac tromethamine inhibits platelet function and is, therefore, CONTRAINDICATED in patients with suspected or confirmed cerebrovascular bleeding, patients with hemorrhagic diathesis, incomplete hemostasis and those at high risk of bleeding (see WARNINGS and PRECAUTIONS).
Ketorolac tromethamine is CONTRAINDICATED as prophylactic analgesic before any major surgery.
HYPERSENSITIVITY
• Hypersensitivity reactions, ranging from bronchospasm to anaphylactic shock, have occurred and appropriate counteractive measures must be available when administering the first dose of ketorolac tromethamine injection (see CONTRAINDICATIONSand WARNINGS). Ketorolac tromethamine is CONTRAINDICATED in patients with previously demonstrated hypersensitivity to ketorolac tromethamine or allergic manifestations to aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs).
INTRATHECAL OR EPIDURAL ADMINISTRATION
• Ketorolac tromethamine is CONTRAINDICATED for intrathecal or epidural administration due to its alcohol content.
RISK DURING LABOR AND DELIVERY
• The use of ketorolac tromethamine in labor and delivery is CONTRAINDICATED because it may adversely affect fetal circulation and inhibit uterine contractions.
CONCOMITANT USE WITH NSAIDs
• Ketorolac tromethamine is CONTRAINDICATED in patients currently receiving aspirin or NSAIDs because of the cumulative risk of inducing serious NSAID-related side effects.
SPECIAL POPULATIONS
• Dosage should be adjusted for patients 65 years or older, for patients under 50 kg (110 lbs.) of body weight (see DOSAGE AND ADMINISTRATION) and for patients with moderately elevated serum creatinine (see WARNINGS). Doses of ketorolac tromethamine injection are not to exceed 60 mg (total dose per day) in these patients.
DOSAGE AND ADMINISTRATION
Ketorolac Tromethamine Tablets
• Ketorolac tromethamine tablets are indicated only as continuation therapy to ketorolac tromethamine injection, and the combined duration of use of ketorolac tromethamine injection and ketorolac tromethamine tablets is not to exceed 5 (five) days, because of the increased risk of serious adverse events.
• The recommended total daily dose of ketorolac tromethamine tablets (maximum 40 mg) is significantly lower than for ketorolac tromethamine injection (maximum 120 mg) (see DOSAGE AND ADMINISTRATION). -
DESCRIPTION
Ketorolac Tromethamine Injection, USP is a member of the pyrrolo-pyrrole group of nonsteroidal anti-inflammatory drugs (NSAIDs). The chemical name for ketorolac tromethamine is (±)-5-benzoyl-2,3-dihydro-1 H-pyrrolizine-1-carboxylic acid, compound with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1), and the structural formula is presented in Figure 1.
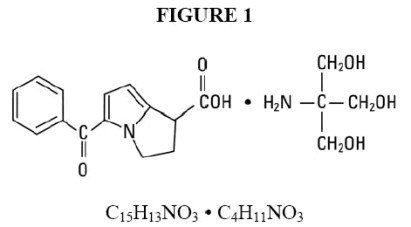
C 15H 13NO 3• C 4H 11NO 3
Ketorolac tromethamine is a racemic mixture of [-]S and [+]R ketorolac tromethamine. Ketorolac tromethamine may exist in three crystal forms. All forms are equally soluble in water. Ketorolac tromethamine has a pKa of 3.5 and an n-octanol/water partition coefficient of 0.26. The molecular weight of ketorolac tromethamine is 376.40.
Ketorolac Tromethamine Injection, USP is available for intravenous (IV) or intramuscular (IM) administration as: 15 mg in 1 mL (1.5%) and 30 mg in 1 mL (3%) in sterile solution; 60 mg in 2 mL (3%) of ketorolac tromethamine in sterile solution is available for intramuscular administration only. The solutions contain 10% (w/v) alcohol, USP, and 6.68 mg, 4.35 mg, and 8.70 mg, respectively, of sodium chloride in sterile water. The pH range is 6.9 to 7.9 and is adjusted with sodium hydroxide and/or hydrochloric acid. The sterile solutions are clear to slightly yellow in color. -
CLINICAL PHARMACOLOGY
Pharmacodynamics
Ketorolac tromethamine is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits analgesic activity in animal models. The mechanism of action of ketorolac, like that of other NSAIDs, is not completely understood but may be related to prostaglandin synthetase inhibition. The biological activity of ketorolac tromethamine is associated with the S-form.
Ketorolac tromethamine possesses no sedative or anxiolytic properties.
The peak analgesic effect of ketorolac tromethamine occurs within 2 to 3 hours and is not statistically significantly different over the recommended dosage range of ketorolac tromethamine. The greatest difference between large and small doses of ketorolac tromethamine by either route is in the duration of analgesia.
Pharmacokinetics
Ketorolac tromethamine is a racemic mixture of [-]S- and [+]R-enantiomeric forms, with the S-form having analgesic activity.
Comparison of Intravenous, Intramuscular and Oral Pharmacokinetics
The pharmacokinetics of ketorolac tromethamine, following intravenous, intramuscular and oral doses of ketorolac tromethamine are compared in Table 1. In adults, the extent of bioavailability following administration of the oral and intramuscular forms of ketorolac tromethamine was equal to that following an intravenous bolus.
Table 1: Table of Approximate Average Pharmacokinetic Parameters (Mean±SD) Following
Oral, Intramuscular and Intravenous Doses of Ketorolac Tromethamine
Pharmacokinetic Parameters
(units)
Oral †
Intramuscular*
Intravenous Bolus ‡
10 mg
15 mg
30 mg
60 mg
15 mg
30 mg
Bioavailability (extent)
100%
T max1(min)
44±34
33±21**
44±29
33±21**
1.1±0.7**
2.9±1.8
C max2(mcg/mL)
[Single-dose]
0.87±0.22
1.14±0.32**
2.42±0.68
4.55±1.27**
2.47±0.51**
4.65±0.96
C max(mcg/mL)
[steady state qid]
1.05±0.26**
1.56±0.44**
3.11±0.87**
N/A ††
3.09±1.17**
6.85±2.61
C min3(mcg/mL)
[steady state qid]
0.29±0.07**
0.47±0.13**
0.93±0.26**
N/A
0.61±0.21**
1.04±0.35
C avg4(mcg/mL)
[steady state qid]
0.59±0.2**
0.94±0.29**
1.88±0.59**
N/A
1.09±0.3**
2.17±0.59
V β5(L/kg)
——————0.175±0.039——————
0.210±0.044
% Dose metabolized=<50
% Dose excreted in feces=6
% Dose excreted in urine=91
% Plasma protein binding=99
1Time-to-peak plasma concentration
†Derived from oral pharmacokinetic studies in 77 normal fasted volunteers
2Peak plasma concentration
*Derived from intramuscular pharmacokinetic studies in 54 normal volunteers
3Trough plasma concentration
‡Derived from intravenous pharmacokinetic studies in 24 normal volunteers
4Average plasma concentration
††Not applicable because 60 mg is only recommended as a single dose
5Volume of distribution
**Mean value was simulated from observed plasma concentration data and standard deviation was simulated from percent coefficient of variation for observed C maxand T maxdata
Linear Kinetics
In adults, following administration of single oral, intramuscular or intravenous doses of ketorolac tromethamine in the recommended dosage ranges, the clearance of the racemate does not change. This implies that the pharmacokinetics of ketorolac tromethamine in adults, following single or multiple intramuscular, intravenous or recommended oral doses of ketorolac tromethamine, are linear. At the higher recommended doses, there is a proportional increase in the concentrations of free and bound racemate.
Distribution
The mean apparent volume (V β) of ketorolac tromethamine following complete distribution was approximately 13 liters. This parameter was determined from single-dose data. The ketorolac tromethamine racemate has been shown to be highly protein bound (99%). Nevertheless, plasma concentrations as high as 10 mcg/mL will only occupy approximately 5% of the albumin binding sites. Thus, the unbound fraction for each enantiomer will be constant over the therapeutic range. A decrease in serum albumin, however, will result in increased free drug concentrations.
Ketorolac tromethamine is excreted in human milk (see PRECAUTIONS – Nursing Mothers).
Metabolism
Ketorolac tromethamine is largely metabolized in the liver. The metabolic products are hydroxylated and conjugated forms of the parent drug. The products of metabolism, and some unchanged drug, are excreted in the urine.
Excretion
The principal route of elimination of ketorolac and its metabolites is renal. About 92% of a given dose is found in the urine, approximately 40% as metabolites and 60% as unchanged ketorolac. Approximately 6% of a dose is excreted in the feces. A single-dose study with 10 mg ketorolac tromethamine (n=9) demonstrated that the S-enantiomer is cleared approximately two times faster than the R-enantiomer and that the clearance was independent of the route of administration. This means that the ratio of S/R plasma concentrations decreases with time after each dose. There is little or no inversion of the R- to S- form in humans. The clearance of the racemate in normal subjects, elderly individuals and in hepatically and renally impaired patients is outlined in Table 2 (see CLINICAL PHARMACOLOGY – Kinetics in Special Populations).
The half-life of the ketorolac tromethamine S-enantiomer was approximately 2.5 hours (SD ± 0.4) compared with 5 hours (SD ± 1.7) for the R-enantiomer. In other studies, the half-life for the racemate has been reported to lie within the range of 5 to 6 hours.
Accumulation
Ketorolac tromethamine administered as an intravenous bolus, every 6 hours, for 5 days, to healthy subjects (n = 13), showed no significant difference in C maxon Day 1 and Day 5. Trough levels averaged 0.29 mcg/mL (SD ± 0.13) on Day 1 and 0.55 mcg/mL (SD ± 0.23) on Day 6. Steady state was approached after the fourth dose.
Accumulation of ketorolac tromethamine has not been studied in special populations (geriatric, pediatric, renal failure patients, or hepatic disease patients).
Kinetics in Special Populations
Geriatric Patients
Based on single-dose data only, the half-life of the ketorolac tromethamine racemate increased from 5 to 7 hours in the elderly (65 to 78 years) compared with young healthy volunteers (24 to 35 years) (see Table 2). There was little difference in the C max for the two groups (elderly, 2.52 mcg/mL ± 0.77; young, 2.99 mcg/mL ± 1.03) (see PRECAUTIONS – Geriatric Use).
Pediatric Patients
Limited information is available regarding the pharmacokinetics of dosing of ketorolac tromethamine in the pediatric population. Following a single intravenous bolus dose of 0.5 mg/kg in 10 children 4 to 8 years old, the half-life was 5.8 ± 1.6 hours, the average clearance was 0.042 ± 0.01 L/hr/kg, the volume of distribution during the terminal phase (V β) was 0.34 ± 0.12 L/kg and the volume of distribution at steady state (V ss) was 0.26 ± 0.08 L/kg. The volume of distribution and clearance of ketorolac in pediatric patients was higher than those observed in adult subjects (see Table 1). There are no pharmacokinetic data available for administration of ketorolac tromethamine by the intramuscular route in pediatric patients.
Renal Insufficiency
Based on single-dose data only, the mean half-life of ketorolac tromethamine in renally impaired patients is between 6 and 19 hours and is dependent on the extent of the impairment. There is poor correlation between creatinine clearance and total ketorolac tromethamine clearance in the elderly and populations with renal impairment (r = 0.5).
In patients with renal disease, the AUC ∞ of each enantiomer increased by approximately 100% compared with healthy volunteers. The volume of distribution doubles for the S-enantiomer and increases by 1/5th for the R-enantiomer. The increase in volume of distribution of ketorolac tromethamine implies an increase in unbound fraction.
The AUC ∞-ratio of the ketorolac tromethamine enantiomers in healthy subjects and patients remained similar, indicating there was no selective excretion of either enantiomer in patients compared to healthy subjects (see WARNINGS – Renal Effects).
Hepatic Insufficiency
There was no significant difference in estimates of half-life, AUC ∞ and C maxin 7 patients with liver disease compared to healthy volunteers (see PRECAUTIONS – Hepatic Effectsand Table 2).
Race
Pharmacokinetic differences due to race have not been identified.
Table 2: The Influence of Age, Liver and Kidney Function, on the Clearance and
Terminal Half-life of Ketorolac Tromethamine (Intramuscular 1and Oral 2) in Adult Populations
Total Clearance [in L/h/kg] 3
Terminal Half-life [in hours]
Type of Subjects
Intramuscular
Mean (range)
Oral
Mean (range)
Intramuscular
Mean (range)
Oral
Mean (range)
Normal Subjects
Intramuscular (n = 54)
mean age = 32, range = 18-60
Oral (n = 77)
mean age = 32, range = 20-60
0.023
(0.010-0.046)
0.025
(0.013-0.050)
5.3
(3.5-9.2)
5.3
(2.4-9)
Healthy Elderly Subjects
Intramuscular (n = 13), Oral (n = 12)
mean age = 72, range = 65-78
0.019
(0.013-0.034)
0.024
(0.018-0.034)
7
(4.7-8.6)
6.1
(4.3-7.6)
Patients with Hepatic Dysfunction
Intramuscular and Oral (n = 7)
mean age = 51, range = 43-64
0.029
(0.013-0.066)
0.033
(0.019-0.051)
5.4
(2.2-6.9)
4.5
(1.6-7.6)
Patients with Renal Impairment
Intramuscular (n = 25), Oral (n = 9)
serum creatinine = 1.9-5.0 mg/dL,
mean age (Intramuscular) = 54,
range = 35-71
mean age (Oral) = 57, range = 39-70
0.015
(0.005-0.043)
0.016
(0.007-0.052)
10.3
(5.9-19.2)
10.8
(3.4-18.9)
Renal Dialysis Patients
Intramuscular and Oral (n = 9)
mean age = 40, range = 27-63
0.016
(0.003-0.036)
-
13.6
(8.0-39.1)
-
1Estimated from 30 mg single intramuscular doses of ketorolac tromethamine
2Estimated from 10 mg single oral doses of ketorolac tromethamine
3Liters/hour/kilogram
Intravenous-Administration:In normal subjects (n=37), the total clearance of 30 mg intravenous-administered ketorolac tromethamine was 0.030 (0.017 to 0.051) L/h/kg. The terminal half-life was 5.6 (4.0 to 7.9) hours. (see Kinetics in Special Populationsfor use of intravenous dosing of ketorolac tromethamine in pediatric patients.)
CLINICAL STUDIES
Adult Patients
In a postoperative study, where all patients received morphine by a PCA device, patients treated with ketorolac tromethamine intravenous as fixed intermittent boluses (e.g., 30 mg initial dose followed by 15 mg every 3 hours), required significantly less morphine (26%) than the placebo group. Analgesia was significantly superior, at various postdosing pain assessment times, in the patients receiving ketorolac tromethamine intravenous plus PCA morphine as compared to patients receiving PCA-administered morphine alone.
-
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of ketorolac tromethamine and other treatment options before deciding to use ketorolac. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
Acute Pain in Adult Patients
Ketorolac tromethamine is indicated for the short-term (≤5 days) management of moderately severe acute pain that requires analgesia at the opioid level, usually in a postoperative setting. Therapy should always be initiated with intravenous or intramuscular dosing of ketorolac tromethamine, and oral ketorolac tromethamine is to be used only as continuation treatment, if necessary.
The total combined duration of use of ketorolac tromethamine injection and oral ketorolac tromethamine is not to exceed 5 days of use because of the potential of increasing the frequency and severity of adverse reactions associated with the recommended doses (see WARNINGS, PRECAUTIONS, DOSAGE AND ADMINISTRATION, and ADVERSE REACTIONS). Patients should be switched to alternative analgesics as soon as possible, but ketorolac tromethamine therapy is not to exceed 5 days. -
CONTRAINDICATIONS
(see also Boxed WARNING)
Ketorolac tromethamine is contraindicated in patients with previously demonstrated hypersensitivity to ketorolac tromethamine.
Ketorolac tromethamine is contraindicated in patients with active peptic ulcer disease, in patients with recent gastrointestinal bleeding or perforation and in patients with a history of peptic ulcer disease or gastrointestinal bleeding.
Ketorolac tromethamine should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS– Anaphylactoid Reactions, and PRECAUTIONS – Pre-existing Asthma).
Ketorolac tromethamine is contraindicated as prophylactic analgesic before any major surgery.
Ketorolac tromethamine is contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS).
Ketorolac tromethamine is contraindicated in patients with advanced renal impairment or in patients at risk for renal failure due to volume depletion (see WARNINGSfor correction of volume depletion).
Ketorolac tromethamine is contraindicated in labor and delivery because, through its prostaglandin synthesis inhibitory effect, it may adversely affect fetal circulation and inhibit uterine musculature, thus increasing the risk of uterine hemorrhage.
Ketorolac tromethamine inhibits platelet function and is, therefore, contraindicated in patients with suspected or confirmed cerebrovascular bleeding, hemorrhagic diathesis, incomplete hemostasis and those at high risk of bleeding (see WARNINGSand PRECAUTIONS).
Ketorolac tromethamine is contraindicated in patients currently receiving aspirin or NSAIDs because of the cumulative risks of inducing serious NSAID-related adverse events.
The concomitant use of ketorolac tromethamine and probenecid is contraindicated.
The concomitant use of ketorolac tromethamine and pentoxifylline is contraindicated.
Ketorolac tromethamine injection is contraindicated for neuraxial (epidural or intrathecal) administration due to its alcohol content. -
WARNINGS
(see also Boxed WARNING)
The total combined duration of use of oral ketorolac tromethamine and intravenous or intramuscular dosing of ketorolac tromethamine is not to exceed 5 days in adults. Ketorolac tromethamine is not indicated for use in pediatric patients.
The most serious risks associated with ketorolac tromethamine are:
- Gastrointestinal Effects - Risk of Ulceration, Bleeding and Perforation:Ketorolac tromethamine is contraindicated in patients with previously documented peptic ulcers and/or gastrointestinal (GI) bleeding. Ketorolac tromethamine can cause serious GI adverse events including bleeding, ulceration and perforation, of the stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with ketorolac tromethamine.
Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Minor upper gastrointestinal problems, such as dyspepsia, are common and may also occur at any time during NSAID therapy.
The incidence and severity of gastrointestinal complications increases with increasing dose of, and duration of treatment with ketorolac tromethamine. Do not use ketorolac tromethamine for more than five days.
However, even short-term therapy is not without risk. In addition to past history of ulcer disease, other factors that increase the risk for GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids, or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age, and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients and therefore, special care should be taken in treating this population.
To minimize the potential risk for an adverse GI event, the lowest effective dose should be used for the shortest possible duration.Patients and physicians should remain alert for signs and symptoms of GI ulceration and bleeding during NSAID therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. This should include discontinuation of ketorolac tromethamine until a serious GI adverse event is ruled out. For high risk patients, alternate therapies that do not involve NSAIDs should be considered.
NSAIDs should be given with care to patients with a history of inflammatory bowel disease (ulcerative colitis, Crohn’s disease) as their condition may be exacerbated.
Hemorrhage
Because prostaglandins play an important role in hemostasis and NSAIDs affect platelet aggregation as well, use of ketorolac tromethamine in patients who have coagulation disorders should be undertaken very cautiously, and those patients should be carefully monitored. Patients on therapeutic doses of anticoagulants (e.g., heparin or dicumarol derivatives) have an increased risk of bleeding complications if given ketorolac tromethamine concurrently; therefore, physicians should administer such concomitant therapy only extremely cautiously. The concurrent use of ketorolac tromethamine and therapy that affects hemostasis, including prophylactic low-dose heparin (2500 to 5000 units every 12 hours), warfarin and dextrans have not been studied extensively, but may also be associated with an increased risk of bleeding. Until data from such studies are available, physicians should carefully weigh the benefits against the risks, and use such concomitant therapy in these patients only extremely cautiously. Patients receiving therapy that affects hemostasis should be monitored closely.
In postmarketing experience, postoperative hematomas and other signs of wound bleeding have been reported in association with the peri-operative use of intravenous or intramuscular dosing of ketorolac tromethamine. Therefore, peri-operative use of ketorolac tromethamine should be avoided and postoperative use be undertaken with caution when hemostasis is critical (see PRECAUTIONS).
Renal Effects
Long-term administration of NSAIDs has resulted in renal papillary necrosis and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of a NSAID may cause a dose-dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics and ACE inhibitors, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
Ketorolac tromethamine and its metabolites are eliminated primarily by the kidneys, which, in patients with reduced creatinine clearance, will result in diminished clearance of the drug (see CLINICAL PHARMACOLOGY). Therefore, ketorolac tromethamine should be used with caution in patients with impaired renal function (see DOSAGE AND ADMINISTRATION) and such patients should be followed closely. With the use of ketorolac tromethamine, there have been reports of acute renal failure, interstitial nephritis and nephrotic syndrome.
Impaired Renal Function
Ketorolac tromethamine is contraindicated in patients with serum creatinine concentrations indicating advanced renal impairment (see CONTRAINDICATIONS). Ketorolac tromethamine should be used with caution in patients with impaired renal function or a history of kidney disease because it is a potent inhibitor of prostaglandin synthesis. Because patients with underlying renal insufficiency are at increased risk of developing acute renal decompensation or failure, the risks and benefits should be assessed prior to giving ketorolac tromethamine to these patients.
Anaphylactoid Reactions
As with other NSAIDs, anaphylactoid reactions may occur in patients without known prior exposure to ketorolac tromethamine. Ketorolac tromethamine should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs (see CONTRAINDICATIONSand PRECAUTIONS–Pre-existing Asthma). Emergency help should be sought in cases where an anaphylactoid reaction occurs.
Cardiovascular Effects
Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first few weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as ketorolac tromethamine, increases the risk of serious gastrointestinal (GI) events (see WARNINGS).
Status Post Coronary Artery Bypass Graft (CABG) Surgery
Two large, controlled clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10 to 14 days following CABG surgery found an increased incidence of myocardial infarction and stroke. NSAIDs are contraindicated in the setting of CABG surgery (see CONTRAINDICATIONS).
Post-MI Patients
Observational studies conducted in the Danish National Registry have demonstrated that patients treated with NSAIDs in the post-MI period were at increased risk of reinfarction, CV-related death, and all-cause mortality beginning in the first week of treatment. In this same cohort, the incidence of death in the first year post MI was 20 per 100 person years in NSAID-treated patients compared to 12 per 100 person years in non-NSAID exposed patients. Although the absolute rate of death declined somewhat after the first year post-MI, the increased relative risk of death in NSAID users persisted over at least the next four years follow-up.
Avoid the use of ketorolac tromethamine in patients with a recent MI unless the benefits are expected to outweigh the risk of recurrent CV thrombotic events. If ketorolac tromethamine is used in patients with a recent MI, monitor patients for signs of cardiac ischemia.
Hypertension
NSAIDs, including ketorolac tromethamine, can lead to onset of new hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Patients taking thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs. NSAIDs, including ketorolac tromethamine, should be used with caution in patients with hypertension. Blood pressure (BP) should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
Heart Failure and Edema
The Coxib and traditional NSAID Trialists’ Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of ketorolac tromethamine may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers (ARBs) (see PRECAUTIONS – Drug Interactions).
Avoid the use of ketorolac tromethamine in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If ketorolac tromethamine is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
Skin Reactions
NSAIDs, including ketorolac tromethamine, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Patients should be informed about the signs and symptoms of serious skin manifestations and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as ketorolac tromethamine. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue ketorolac tromethamine and evaluate the patient immediately.
Fetal Toxicity
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs, including ketorolac tromethamine, in pregnant women at about 30 weeks gestation and later. NSAIDs including ketorolac tromethamine, increase the risk of premature closure of the fetal ductus arteriosus at approximately this gestational age.
Oligohydramnios/Neonatal Renal Impairment:
Use of NSAIDs, including ketorolac tromethamine, at about 20 weeks gestation or later in pregnancy may cause fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. Oligohydramnios is often, but not always, reversible with treatment discontinuation. Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation. In some postmarketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If NSAID treatment is necessary between about 20 weeks and 30 weeks gestation, limit ketorolac tromethamine use to the lowest effective dose and shortest duration possible. Consider ultrasound monitoring of amniotic fluid if ketorolac tromethamine treatment extends beyond 48 hours. Discontinue ketorolac tromethamine if oligohydramnios occurs and follow up according to clinical practice (see PRECAUTIONS; Pregnancy).
-
PRECAUTIONS
General
Ketorolac tromethamine cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of ketorolac tromethamine in reducing inflammation may diminish the utility of this diagnostic sign in detecting complications of presumed noninfectious, painful conditions.
Hepatic EffectsKetorolac tromethamine should be used with caution in patients with impaired hepatic function or a history of liver disease. Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including ketorolac tromethamine. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately three or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes have been reported.
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with ketorolac tromethamine. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), ketorolac tromethamine should be discontinued.
Hematologic Effects
Anemia is sometimes seen in patients receiving NSAIDs, including ketorolac tromethamine. This may be due to fluid retention, occult or gross GI blood loss, or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including ketorolac tromethamine, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia. NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Patients receiving ketorolac tromethamine who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants, should be carefully monitored.
Pre-existing AsthmaPatients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other nonsteroidal anti-inflammatory drugs has been reported in such aspirin-sensitive patients, ketorolac tromethamine should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with pre-existing asthma.
Information for Patients
Ketorolac tromethamine is a potent NSAID and may cause serious side effects such as gastrointestinal bleeding or kidney failure, which may result in hospitalization and even fatal outcome.
Physicians, when prescribing ketorolac tromethamine, should inform their patients or their guardians of the potential risks of ketorolac tromethamine treatment (see Boxed WARNING, WARNINGS, PRECAUTIONS, and ADVERSE REACTIONSsections), instruct patients to seek medical advice if they develop treatment-related adverse events, and advise patients not to give oral ketorolac tromethamine to other family members and to discard any unused drug. Remember that the total combined duration of use of oral ketorolac tromethamine and intravenous or intramuscular dosing of ketorolac tromethamine is not to exceed 5 days in adults. Ketorolac tromethamine is not indicated for use in pediatric patients. Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
1. Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their health care provider immediately (see WARNINGS).
2. Ketorolac tromethamine, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see WARNINGS, Gastrointestinal Effects: Risk of Ulceration, Bleeding, and Perforation).
3. Serious Skin Reactions, including DRESS
Advise patients to stop taking ketorolac tromethamine immediately if they develop any type of rash or fever and to contact their healthcare provider as soon as possible (see WARNINGS).
4. Advise patients to be alert for the symptoms of congestive heart failure including shortness of breath, unexplained weight gain, or edema and to contact their healthcare provider if such symptoms occur (see WARNINGS).
5. Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
6. Patients should be informed of the signs of an anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see WARNINGS).
7. Fetal ToxicityInform pregnant women to avoid use of ketorolac tromethamine and other NSAIDs starting at 30 weeks gestation because of the risk of the premature closing of the fetal ductus arteriosus. If treatment with ketorolac tromethamine is needed for a pregnant woman between about 20 to 30 weeks gestation, advise her that she may need to be monitored for oligohydramnios, if treatment continues for longer than 48 hours (see WARNINGS; Fetal Toxicity,PRECAUTIONS; Pregnancy).
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs, should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash etc.) or if abnormal liver tests persist or worsen, ketorolac tromethamine should be discontinued.
Drug Interactions
Ketorolac is highly bound to human plasma protein (mean 99.2%). There is no evidence in animal or human studies that ketorolac tromethamine induces or inhibits hepatic enzymes capable of metabolizing itself or other drugs.
Warfarin, Digoxin, Salicylate, and Heparin
The in vitrobinding of warfarinto plasma proteins is only slightly reduced by ketorolac tromethamine (99.5% control vs 99.3%) when ketorolac plasma concentrations reach 5 to 10 mcg/mL. Ketorolac does not alter digoxinprotein binding. In vitrostudies indicate that, at therapeutic concentrations of salicylate(300 mcg/mL), the binding of ketorolac was reduced from approximately 99.2% to 97.5%, representing a potential two-fold increase in unbound ketorolac plasma levels. Therapeutic concentrations of digoxin, warfarin, ibuprofen, naproxen, piroxicam, acetaminophen, phenytoinand tolbutamidedid not alter ketorolac tromethamine protein binding.
In a study involving 12 adult volunteers, oral ketorolac tromethamine was coadministered with a single dose of 25 mg warfarin, causing no significant changes in pharmacokinetics or pharmacodynamics of warfarin. In another study, ketorolac tromethamine dosed intravenous or intramuscular was given with two doses of 5000 U of heparinto 11 healthy volunteers, resulting in a mean template bleeding time of 6 minutes (3.2 to 11.4 min) compared to a mean of 6.0 minutes (3.4 to 7.5 min) for heparin alone and 5.1 minutes (3.5 to 8.5 min) for placebo. Although these results do not indicate a significant interaction between ketorolac tromethamine and warfarin or heparin, the administration of ketorolac tromethamine to patients taking anticoagulants should be done extremely cautiously, and patients should be closely monitored (see WARNINGSand PRECAUTIONS – Hematologic Effects).
The effects of warfarin and NSAIDs, in general, on GI bleeding are synergistic, such that the users of both drugs together have a risk of serious GI bleeding higher than the users of either drug alone.
Aspirin
When ketorolac tromethamine is administered with aspirin, its protein binding is reduced, although the clearance of free ketorolac tromethamine is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of ketorolac tromethamine and aspirin is not generally recommended because of the potential of increased adverse effects.
Diuretics
Clinical studies, as well as postmarketing observations, have shown that ketorolac tromethamine can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see WARNINGS – Renal Effects), as well as to assure diuretic efficacy.
Probenecid
Concomitant administration of oral ketorolac tromethamine and probenecid resulted in decreased clearance and volume of distribution of ketorolac and significant increases in ketorolac plasma levels (total AUC increased approximately three-fold from 5.4 to 17.8 mcg/h/mL) and terminal half-life increased approximately two-fold from 6.6 to 15.1 hours. Therefore, concomitant use of ketorolac tromethamine and probenecid is contraindicated.
Lithium
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Methotrexate
NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
ACE Inhibitors/Angiotensin II Receptor Antagonists
Concomitant use of ACE inhibitors and/or angiotensin II receptor antagonistsmay increase the risk of renal impairment, particularly in volume-depleted patients.
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE inhibitors and/or angiotensin II receptor antagonists. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE inhibitors and/or angiotensin II receptor antagonists.
Antiepileptic Drugs
Sporadic cases of seizures have been reported during concomitant use of ketorolac tromethamine and antiepileptic drugs (phenytoin, carbamazepine).
Psychoactive Drugs
Hallucinations have been reported when ketorolac tromethamine was used in patients taking psychoactive drugs (fluoxetine, thiothixene, alprazolam).
Pentoxifylline
When ketorolac tromethamine is administered concurrently with pentoxifylline, there is an increased tendency to bleeding.
Nondepolarizing Muscle Relaxants
In postmarketing experience there have been reports of a possible interaction between ketorolac tromethamine intravenous/intramuscular and nondepolarizing muscle relaxantsthat resulted in apnea. The concurrent use of ketorolac tromethamine with muscle relaxants has not been formally studied.
Selective Serotonin Reuptake Inhibitors (SSRIs)
There is an increased risk of gastrointestinal bleeding when selective serotonin reuptake inhibitors(SSRIs) are combined with NSAIDs. Caution should be used when NSAIDs are administered concomitantly with SSRIs.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
An 18-month study in mice with oral doses of ketorolac tromethamine tablets at 2 mg/kg/day (0.9 times the human systemic exposure at the recommended intramuscular or intravenous dose of 30 mg qid, based on area-under-the-plasma-concentration curve [AUC]), and a 24-month study in rats at 5 mg/kg/day (0.5 times the human AUC) showed no evidence of tumorigenicity.
Ketorolac tromethamine was not mutagenic in the Ames test, unscheduled DNA synthesis and repair, and in forward mutation assays. Ketorolac tromethamine did not cause chromosome breakage in the in vivomouse micronucleus assay. At 1590 mcg/mL and at higher concentrations, ketorolac tromethamine increased the incidence of chromosomal aberrations in Chinese hamster ovarian cells.
Impairment of fertility did not occur in male or female rats at oral doses of 9 mg/kg (0.9 times the human AUC) and 16 mg/kg (1.6 times the human AUC) of ketorolac tromethamine, respectively.
Pregnancy
Risk Summary
Use of NSAIDs, including ketorolac tromethamine, can cause premature closure of the fetal ductus arteriosus and fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. Because of these risks, limit dose and duration of ketorolac tromethamine use between about 20 and 30 weeks of gestation, and avoid ketorolac tromethamine use at about 30 weeks of gestation and later in pregnancy (see WARNINGS; Fetal Toxicity).
Premature Closure of Fetal Ductus Arteriosus:
Use of NSAIDs, including ketorolac tromethamine, at about 30 weeks gestation or later in pregnancy increases the risk of premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment:
Use of NSAIDs at about 20 weeks gestation or later in pregnancy has been associated with cases of fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment.
Data from observational studies regarding other potential embryofetal risks of NSAID use in women in the first or second trimesters of pregnancy are inconclusive. Reproduction studies have been performed during organogenesis using daily oral doses of ketorolac tromethamine at 3.6 mg/kg (0.37 times the human AUC) in rabbits and at 10 mg/kg (1.0 times the human AUC) in rats. Results of these studies did not reveal evidence of teratogenicity to the fetus. However, animal reproduction studies are not always predictive of human response. Oral doses of ketorolac tromethamine at 1.5 mg/kg (0.14 times the human AUC), administered after gestation Day 17, caused dystocia and higher pup mortality in rats. Based on animal data, prostaglandins have been shown to have an important role in endometrial vascular permeability, blastocyst implantation, and decidualization. In animal studies, administration of prostaglandin synthesis inhibitors such as ketorolac tromethamine, resulted in increased pre- and post-implantation loss. Prostaglandins also have been shown to have an important role in fetal kidney development. In published animal studies, prostaglandin synthesis inhibitors have been reported to impair kidney development when administered at clinically relevant doses.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy, because NSAIDs, including ketorolac tromethamine, can cause premature closure of the fetal ductus arteriosus (see WARNINGS; Fetal Toxicity).
Oligohydramnios/Neonatal Renal Impairment:
If an NSAID is necessary at about 20 weeks gestation or later in pregnancy, limit the use to the lowest effective dose and shortest duration possible. If ketorolac tromethamine treatment extends beyond 48 hours, consider monitoring with ultrasound for oligohydramnios. If oligohydramnios occurs, discontinue ketorolac tromethamine and follow up according to clinical practice (see WARNINGS; Fetal Toxicity).
Data
Human Data
There are no adequate and well-controlled studies of ketorolac tromethamine in pregnant women. Ketorolac tromethamine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Premature Closure of Fetal Ductus Arteriosus
Published literature reports that the use of NSAIDs at about 30 weeks of gestation and later in pregnancy may cause premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment
Published studies and postmarketing reports describe maternal NSAID use at about 20 weeks gestation or later in pregnancy associated with fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. In many cases, but not all, the decrease in amniotic fluid was transient and reversible with cessation of the drug. There have been a limited number of case reports of maternal NSAID use and neonatal renal dysfunction without oligohydramnios, some of which were irreversible. Some cases of neonatal renal dysfunction required treatment with invasive procedures, such as exchange transfusion or dialysis.
Methodological limitations of these postmarketing studies and reports include lack of a control group; limited information regarding dose, duration, and timing of drug exposure; and concomitant use of other medications. These limitations preclude establishing a reliable estimate of the risk of adverse fetal and neonatal outcomes with maternal NSAID use. Because the published safety data on neonatal outcomes involved mostly preterm infants, the generalizability of certain reported risks to the full-term infant exposed to NSAIDs through maternal use is uncertain.
Labor and Delivery
The use of ketorolac tromethamine is contraindicated in labor and delivery because, through its prostaglandin synthesis inhibitory effect, it may adversely affect fetal circulation and inhibit uterine contractions, thus increasing the risk of uterine hemorrhage (see CONTRAINDICATIONS).
Effects on Fertility
The use of ketorolac tromethamine, as with any drug known to inhibit cyclooxygenase/ prostaglandin synthesis, may impair fertility and is not recommended in women attempting to conceive. In women who have difficulty conceiving or are undergoing investigation of infertility, withdrawal of ketorolac tromethamine should be considered.
Nursing Mothers
Limited data from one published study that included 10 breastfeeding women 2-6 days postpartum showed low levels of ketorolac in breast milk and were undetectable (less than 5 ng/mL) in 4 of the patients. After a single administration of 10 mg of ketorolac tromethamine, the maximum milk concentration observed was 7.3 ng/mL, and the maximum milk-to-plasma ratio was 0.037. After 1 day of dosing (10 mg every 6 hours), the maximum milk concentration was 7.9 ng/mL, and the maximum milk-to-plasma ratio was 0.025. Assuming a daily intake of 400 to 1,000 mL of human milk per day and a maternal body weight of 60 kg, the calculated maximum daily infant exposure was 0.00263 mg/kg/day, which is 0.4% of the maternal weight-adjusted dose.
Exercise caution when ketorolac is administered to a nursing woman. Available information has not shown any specific adverse events in nursing infants; however, instruct patients to contact their infant's healthcare provider if they note any adverse events.Pediatric Use
Ketorolac tromethamine is not indicated for use in pediatric patients. The safety and effectiveness of ketorolac tromethamine in pediatric patients below the age of 17 years have not been established.
Geriatric Use
(≥ 65 Years of Age)
Because ketorolac tromethamine may be cleared more slowly by the elderly (see CLINICAL PHARMACOLOGY) who are also more sensitive to the dose-related adverse effects of NSAIDs (see WARNINGS - Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation), extreme caution and reduced dosages (see DOSAGE AND ADMINISTRATION) and careful clinical monitoring must be used when treating the elderly with ketorolac tromethamine . -
ADVERSE REACTIONS
Adverse reaction rates increase with higher doses of ketorolac tromethamine. Practitioners should be alert for the severe complications of treatment with ketorolac tromethamine, such as G.I. ulceration, bleeding and perforation, postoperative bleeding, acute renal failure, anaphylactic and anaphylactoid reactions and liver failure (see Boxed WARNING, WARNINGS, PRECAUTIONS, and DOSAGE AND ADMINISTRATION). These NSAID-related complications can be serious in certain patients for whom ketorolac tromethamine is indicated, especially when the drug is used inappropriately.
In patients taking ketorolac tromethamine or other NSAIDs in clinical trials, the most frequently reported adverse experiences in approximately 1% to 10% of patients are:Gastrointestinal (GI) experiences including: abdominal pain constipation/diarrhea dyspepsia flatulence GI fullness GI ulcers (gastric/duodenal) gross bleeding/perforation heartburn nausea* stomatitis vomiting Other experiences: abnormal renal function anemia dizziness drowsiness edema elevated liver enzymes headaches* hypertension increased bleeding time injection site pain pruritus purpura rashes tinnitus sweating *Incidence greater than 10%
Additional adverse experiences reported occasionally (<1% in patients taking ketorolac tromethamine or other NSAIDs in clinical trials) include:
Body as a Whole:fever, infections, sepsis
Cardiovascular:congestive heart failure, palpitation, pallor, tachycardia, syncope
Dermatologic:alopecia, photosensitivity, urticaria
Gastrointestinal:anorexia, dry mouth, eructation, esophagitis, excessive thirst, gastritis, glossitis, hematemesis, hepatitis, increased appetite, jaundice, melena, rectal bleeding
Hemic and Lymphatic:ecchymosis, eosinophilia, epistaxis, leukopenia, thrombocytopenia
Metabolic and Nutritional:weight change
Nervous System:abnormal dreams, abnormal thinking, anxiety, asthenia, confusion, depression, euphoria, extrapyramidal symptoms, hallucinations, hyperkinesis, inability to concentrate, insomnia, nervousness, paresthesia, somnolence, stupor, tremors, vertigo, malaiseReproductive, female:infertility
Respiratory:asthma, cough, dyspnea, pulmonary edema, rhinitis
Special Senses:abnormal taste, abnormal vision, blurred vision, hearing loss
Urogenital:cystitis, dysuria, hematuria, increased urinary frequency, interstitial nephritis, oliguria/polyuria, proteinuria, renal failure, urinary retention
Other rarely observed reactions (reported from postmarketing experience in patients taking ketorolactromethamine or other NSAIDs) are:Body as a Whole:angioedema, death, hypersensitivity reactions such as anaphylaxis, anaphylactoid reaction, laryngeal edema, tongue edema (see WARNINGS), myalgia
Cardiovascular:arrhythmia, bradycardia, chest pain, flushing, hypotension, myocardial infarction, vasculitis
Dermatologic:exfoliative dermatitis, erythema multiforme, Lyell's syndrome, bullous reactions including Stevens-Johnson syndrome and toxic epidermal necrolysisGastrointestinal:acute pancreatitis, liver failure, ulcerative stomatitis, exacerbation of inflammatory bowel disease (ulcerative colitis, Crohn’s disease)
Hemic and Lymphatic:agranulocytosis, aplastic anemia, hemolytic anemia, lymphadenopathy, pancytopenia, post operative wound hemorrhage (rarely requiring blood transfusion — see Boxed WARNING, WARNINGS, and PRECAUTIONS)
Metabolic and Nutritional:hyperglycemia, hyperkalemia, hyponatremia
Nervous System:aseptic meningitis, convulsions, coma, psychosis
Respiratory:bronchospasm, respiratory depression, pneumonia
Special Senses:conjunctivitis
Urogenital:flank pain with or without hematuria and/or azotemia, hemolytic uremic syndrome
Postmarketing Surveillance StudyA large postmarketing observational, nonrandomized study, involving approximately 10,000 patients receiving ketorolac tromethamine, demonstrated that the risk of clinically serious gastrointestinal (GI) bleeding was dose-dependent (see Tables 3Aand 3B). This was particularly true in elderly patients who received an average daily dose greater than 60 mg/day of ketorolac tromethamine (see Table 3A).
Table 3:Incidence of Clinically Serious G.I. Bleeding as Related to Age, Total Daily Dose, and History of G.I. Perforation, Ulcer, Bleeding (PUB) after up to 5 Days of Treatment with Ketorolac Tromethamine Injection A. Adult Patients without History of PUB Age of Patients Total Daily Dose of Ketorolac Tromethamine Injection ≤60 mg >60 to 90 mg >90 to 120 mg >120 mg <65 years of age 0.4% 0.4% 0.9% 4.6% ≥65 years of age 1.2% 2.8% 2.2% 7.7% B. Adult Patients with History of PUB Age of Patients Total Daily Dose of Ketorolac Tromethamine Injection ≤60 mg >60 to 90 mg >90 to 120 mg >120 mg <65 years of age 2.1% 4.6% 7.8% 15.4% ≥65 years of age 4.7% 3.7% 2.8% 25.0% To report SUSPECTED ADVERSE REACTIONS, contact Fosun Pharma USA Inc. at 1-866-611-3762 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
Symptoms and Signs
Symptoms following acute NSAIDs overdoses are usually limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Hypertension, acute renal failure, respiratory depression and coma may occur, but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
Treatment
Patients should be managed by symptomatic and supportive care following a NSAIDs overdose. There are no specific antidotes. Forced diuresis, alkalization of urine, hemodialysis or hemoperfusion may not be useful due to high protein binding.
Single overdoses of ketorolac tromethamine have been variously associated with abdominal pain, nausea, vomiting, hyperventilation, peptic ulcers and/or erosive gastritis and renal dysfunction which have resolved after discontinuation of dosing.
-
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of ketorolac tromethamine and other treatment options before deciding to use ketorolac tromethamine. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals. In adults, the combined duration of use of intravenous or intramuscular dosing of ketorolac tromethamine and oral ketorolac tromethamine is not to exceed 5 days. In adults, the use of oral ketorolac tromethamine is only indicated as continuation therapy to intravenous or intramuscular dosing of ketorolac tromethamine. See package insert for ketorolac tromethamine tablets for transition from intravenous or intramuscular dosing of ketorolac tromethamine (single- or multiple-dose) to multiple-dose oral ketorolac tromethamine.
Note: Oral formulationshould notbe given as an initial dose.Use minimum effective dosefor the individual patient.
Total duration of treatment in adult patients:the combined duration of use of intravenous or intramuscular dosing of ketorolac tromethamine and oral ketorolac tromethamine is not to exceed 5 days.
KETOROLAC TROMETHAMINE INJECTION
Ketorolac tromethamine injection may be used as a single or multiple dose on a regular or “as needed” schedule for the management of moderately severe, acute pain that requires analgesia at the opioid level, usually in a postoperative setting. Hypovolemia should be corrected prior to the administration of ketorolac tromethamine (see WARNINGS - Renal Effects). Patients should be switched to alternative analgesics as soon as possible, but ketorolac tromethamine therapy is not to exceed 5 days.
When administering ketorolac tromethamine injection, the intravenous bolus must be given over no less than 15 seconds. The intramuscular administration should be given slowly and deeply into the muscle. The analgesic effect begins in ~30 minutes with maximum effect in 1 to 2 hours after dosing intravenous or intramuscular. Duration of analgesic effect is usually 4 to 6 hours.
Single-Dose Treatment: The following regimen should be limited to single administration use only
Intramuscular Dosing
• Patients <65 years of age: One dose of 60 mg.
• Patients ≥65 years of age, renally impaired and/or less than 50 kg (110 lbs) of body weight:One dose of 30 mg.
Intravenous Dosing
• Patients <65 years of age: One dose of 30 mg.
•Patients ≥65 years of age, renally impaired and/or less than 50 kg (110 lbs) of body weight:One dose of 15 mg.
Multiple-Dose Treatment (Intravenous or Intramuscular)
• Patients <65 years of age: The recommended dose is 30 mg ketorolac tromethamine injection every 6 hours. The maximum daily dose for these populations should
not exceed 120 mg.
• For patients ≥65 years of age, renally impaired patients (see WARNINGS), and patients less than 50 kg (110 lbs): The recommended dose is 15 mg ketorolac tromethamine injection every 6 hours. The maximum daily dose for these populations should not exceed 60 mg.
For breakthrough pain, do not increase the dose or the frequency of ketorolac tromethamine. Consideration should be given to supplementing these regimens with low doses of opioids "as needed" unless otherwise contraindicated.
Pharmaceutical Information for Ketorolac Tromethamine InjectionKetorolac tromethamine injection should not be mixed in a small volume (e.g., in a syringe) with morphine sulfate, meperidine hydrochloride, promethazine hydrochloride or hydroxyzine hydrochloride; this will result in precipitation of ketorolac from solution.
NOTE:Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
-
HOW SUPPLIED
Ketorolac Tromethamine Injection, USP is supplied as follows:
NDC Number Ketorolac Tromethamine Injection USP Package Factor 72266-234-25 15 mg/mL, 1 mL single-dose vial 25 vials per carton 72266-118-25 30 mg/mL, 1 mL single-dose vial 25 vials per carton 72266-119-25* 60 mg/2 mL, 2 mL single-dose vial 25 vials per carton *FOR IM USE ONLY
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]Protect from light. Retain in carton until time of use.
Distributed by:
Fosun Pharma USA Inc.
Princeton, NJ 08540
Made in India
Revised: 05/2023 -
Medication Guide for Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
What is the most important information I should know about medicines called Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAIDs can cause serious side effects, including:
• Increased risk of a heart attack or stroke that can lead to death.This risk may happen early in treatment and may increase:
o with increasing doses of NSAIDs
o with longer use of NSAIDsDo not take NSAIDs right before or after a heart surgery called a"coronary artery bypass graft (CABG)."
Avoid taking NSAIDs after a recent heart attack, unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take NSAIDs after a recent heart attack.• Increased risk of bleeding, ulcers, and tears (perforation) of the esophagus (tube leading from the mouth to the stomach), stomach and intestines:
•anytime during use
•without warning symptoms
•that may cause deathThe risk of getting an ulcer or bleeding increases with:
•past history of stomach ulcers, or stomach or intestinal bleeding with the use of NSAIDs
•taking medicines called "corticosteroids","anticoagulants", "SSRIs", or "SNRIs"
•increasing doses of NSAIDs
•longer use of NSAIDs
•smoking
•drinking alcohol
•older age
•poor health
•advanced liver disease
•bleeding problemsNSAIDs should only be used:
•exactly as prescribed
•at the lowest dose possible for your treatment
•for the shortest time neededWhat are NSAIDs?
NSAIDs are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as different types of arthritis, menstrual cramps, and other types of short-term pain.Who should not take NSAIDs?
Do not take NSAIDs:
• if you had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAIDs.
• right before or after heart bypass surgery.
Before taking NSAIDs, tell your healthcare provider about all of your medical conditions, including if you:
• have liver or kidney problems
• have high blood pressure
• have asthma
• are pregnant or plan to become pregnant. Taking NSAIDs at about 20 weeks of pregnancy or later may harm your unborn baby. If you need to take NSAIDs for more than 2 days when you are between 20 and 30 weeks of pregnancy, your healthcare provider may need to monitor the amount of fluid in your womb around your baby. You should not takeNSAIDs after about 30 weeks of pregnancy.• are breastfeeding or plan to breast feed.
Tell your healthcare provider about all of the medicines you take, including prescription or over-the-counter medicines, vitamins or herbalsupplements.NSAIDs and some other medicines can interact with each other and cause serious side effects. Do not start taking any new medicine without talking to your healthcare provider first.
What are the possible side effects of NSAIDs?
NSAIDs can cause serious side effects, including:
See "What is the most important information I should know about medicines called Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) ?"
• new or worse high blood pressure
• heart failure
• liver problems including liver failure
• kidney problems including kidney failure
• low red blood cells (anemia)
• life-threatening skin reactions
• life-threatening allergic reactions
• Other side effects of NSAIDs include:stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness.Get emergency help right away if you get any of the following symptoms:
• shortness of breath or trouble breathing
• chest pain
• weakness in one part or side of your body
• slurred speech
• swelling of the face or throatStop taking your NSAID and call your healthcare provider right away if you get any of the following symptoms:
• nausea
• more tired or weaker than usual
• diarrhea
• itching
• your skin or eyes look yellow
• indigestion or stomach pain
• flu-like symptoms
• vomit blood
• there is blood in your bowel movement or it is black and sticky like tar
• unusual weight gain
• skin rash or blisters with fever
• swelling of the arms and legs, hands and feetIf you take too much of your NSAID, call your healthcare provider or get medical help right away.
These are not all the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs.Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about NSAIDs
• Aspirin is an NSAID medicine but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
• Some NSAIDs are sold in lower doses without a prescription (over-the-counter). Talk to your healthcare provider before using over-the-counter NSAIDs for more than 10 days.General information about the safe and effective use of NSAIDs
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use NSAIDs for a condition for which it was not prescribed. Do not give NSAIDs to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information about NSAIDs, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about NSAIDs that is written for health professionals.
This Medication Guide has been approved by the U.S. Food and Drug Administration.Distributed by:
Fosun Pharma USA Inc.
Princeton, NJ 08540
Made in India
Revised: 05/2023 - SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Lidocaine hydrochloride injection, USP is sterile, nonpyrogenic, aqueous solution that contains a local anesthetic agent and is administered parenterally by injection. See INDICATIONS AND USAGEsection for specific uses.
Lidocaine hydrochloride injection, USP contains lidocaine hydrochloride, which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the molecular weight 270.8. Lidocaine hydrochloride (C 14H 22N 2O • HCl) has the following structural formula: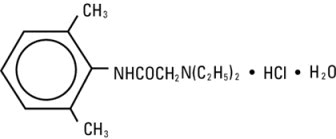
Lidocaine hydrochloride injection, USP is a sterile, nonpyrogenic, isotonic solution containing sodium chloride. The pH of the solution is adjusted to approximately 6.5 (5.0 to 7.0) with sodium hydroxide and/or hydrochloric acid.
-
CLINICAL PHARMACOLOGY
Mechanism of Action
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby effecting local anesthetic action.
Hemodynamics
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. With central neural blockade these changes may be attributable to block of autonomic fibers, a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system, and/or the beta-adrenergic receptor stimulating action of epinephrine when present. The net effect is normally a modest hypotension when the recommended dosages are not exceeded.
Pharmacokinetics and Metabolism
Information derived from diverse formulations, concentrations and usages reveals that lidocaine is completely absorbed following parenteral administration, its rate of absorption depending, for example, upon various factors such as the site of administration and the presence or absence of a vasoconstrictor agent. Except for intravascular administration, the highest blood levels are obtained following intercostal nerve block and the lowest after subcutaneous administration.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Lidocaine is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2,6-dimethylaniline.
The elimination half-life of lidocaine following an intravenous bolus injection is typically 1.5 to 2 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity. -
INDICATIONS AND USAGE
Lidocaine hydrochloride injection is indicated for production of local or regional anesthesia by infiltration techniques such as percutaneous injection and intravenous regional anesthesia by peripheral nerve block techniques such as brachial plexus and intercostal and by central neural techniques such as lumbar and caudal epidural blocks, when the accepted procedures for these techniques as described in standard textbooks are observed.
- CONTRAINDICATIONS
-
WARNINGS
LIDOCAINE HYDROCHLORIDE INJECTION FOR INFILTRATION AND NERVE BLOCK SHOULD BE EMPLOYED ONLY BY CLINICIANS WHO ARE WELL VERSED IN DIAGNOSIS AND MANAGEMENT OF DOSE-RELATED TOXICITY AND OTHER ACUTE EMERGENCIES THAT MIGHT ARISE FROM THE BLOCK TO BE EMPLOYED AND THEN ONLY AFTER ENSURING THE IMMEDIATEAVAILABILITY OF OXYGEN, OTHER RESUSCITATIVE DRUGS, CARDIOPULMONARY EQUIPMENT AND THE PERSONNEL NEEDED FOR PROPER MANAGEMENT OF TOXIC REACTIONS AND RELATED EMERGENCIES (see also ADVERSE REACTIONSand PRECAUTIONS). DELAY IN PROPER MANAGEMENT OF DOSE-RELATED TOXICITY, UNDERVENTILATION FROM ANY CAUSE AND/OR ALTERED SENSITIVITY MAY LEAD TO THE DEVELOPMENT OF ACIDOSIS, CARDIAC ARREST AND, POSSIBLY, DEATH.
Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue lidocaine hydrochloride and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
Intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures is an unapproved use, and there have been postmarketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are not associated with these findings. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2 ndmonth after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
To avoid intravascular injection, aspiration should be performed before the local anesthetic solution is injected. The needle must be repositioned until no return of blood can be elicited by aspiration. Note, however, that the absence of blood in the syringe does not guarantee that intravascular injection has been avoided.
Anaphylactic reactions may occur following administration of lidocaine hydrochloride (see ADVERSE REACTIONS).
In the case of severe reaction, discontinue the use of the drug. -
PRECAUTIONS
General
The safety and effectiveness of lidocaine hydrochloride depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Standard textbooks should be consulted for specific techniques and precautions for various regional anesthetic procedures.
Resuscitative equipment, oxygen, and other resuscitative drugs should be available for immediate use (see WARNINGSand ADVERSE REACTIONS). The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Syringe aspirations should also be performed before and during each supplemental injection when using indwelling catheter techniques. During the administration of epidural anesthesia, it is recommended that a test dose be administered initially and that the patient be monitored for central nervous system toxicity and cardiovascular toxicity, as well as for signs of unintended intrathecal administration, before proceeding. When clinical conditions permit, consideration should be given to employing local anesthetic solutions that contain epinephrine for the test dose because circulatory changes compatible with epinephrine may also serve as a warning sign of unintended intravascular injection. An intravascular injection is still possible even if aspirations for blood are negative. Repeated doses of lidocaine hydrochloride may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical condition. Lidocaine hydrochloride should also be used with caution in patients with severe shock or heart block.
Lumbar and caudal epidural anesthesia should be used with extreme caution in persons with the following conditions: existing neurological disease, spinal deformities, septicemia, and severe hypertension.
Local anesthetic solutions containing a vasoconstrictor should be used cautiously and in carefully circumscribed quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply. Patients with peripheral vascular disease and those with hypertensive vascular disease may exhibit exaggerated vasoconstrictor response. Ischemic injury or necrosis may result. Preparations containing a vasoconstrictor should be used with caution in patients during or following the administration of potent general anesthetic agents, since cardiac arrhythmias may occur under such conditions.
Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient’s state of consciousness should be accomplished after each local anesthetic injection. It should be kept in mind at such times that restlessness, anxiety, tinnitus, dizziness, blurred vision, tremors, depression or drowsiness may be early warning signs of central nervous system toxicity.
Since amide-type local anesthetics are metabolized by the liver, lidocaine hydrochloride injection should be used with caution in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations. Lidocaine hydrochloride injection should also be used with caution in patients with impaired cardiovascular function since they may be less able to compensate for functional changes associated with the prolongation of A-V conduction produced by these drugs.
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for the management of malignant hyperthermia should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Proper tourniquet technique, as described in publications and standard textbooks, is essential in the performance of intravenous regional anesthesia. Solutions containing epinephrine or other vasoconstrictors should not be used for this technique.
Lidocaine hydrochloride should be used with caution in persons with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine hydrochloride.Use in the Head and Neck Area
Small doses of local anesthetics injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. Patients receiving these blocks should have their circulation and respiration monitored and be constantly observed. Resuscitative equipment and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded (see DOSAGE AND ADMINISTRATION).
Information for Patients
When appropriate, patients should be informed in advance that they may experience temporary loss of sensation and motor activity, usually in the lower half of the body, following proper administration of epidural anesthesia.
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.Clinically Significant Drug Interactions
The administration of local anesthetic solutions containing epinephrine or norepinephrine to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension.
Phenothiazines and butyrophenones may reduce or reverse the pressor effect of epinephrine.
Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful patient monitoring is essential.
Concurrent administration of vasopressor drugs (for the treatment of hypotension related to obstetric blocks) and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents.Drug/Laboratory Test Interactions
The intramuscular injection of lidocaine hydrochloride may result in an increase in creatine phosphokinase levels. Thus, the use of this enzyme determination, without isoenzyme separation, as a diagnostic test for the presence of acute myocardial infarction may be compromised by the intramuscular injection of lidocaine hydrochloride.
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
Class
Examples
Nitrates/Nitrites
nitric oxide, nitroglycerin, nitroprusside, nitrous oxide
Local anesthetics
articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine
Antineoplastic agents
cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase
Antibiotics
dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides
Antimalarials
chloroquine, primaquine
Anticonvulsants
Phenobarbital, phenytoin, sodium valproate
Other drugs
acetaminophen, metoclopramide, quinine,
sulfasalazine
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies of lidocaine hydrochloride in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
Pregnancy
Teratogenic Effects
Reproduction studies have been performed in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by lidocaine hydrochloride. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering lidocaine hydrochloride to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
Labor and Delivery
Local anesthetics rapidly cross the placenta and when used for epidural, paracervical, pudendal or caudal block anesthesia, can cause varying degrees of maternal, fetal and neonatal toxicity see CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism). The potential for toxicity depends upon the procedure performed, the type and amount of drug used, and the technique of drug administration. Adverse reactions in the parturient, fetus and neonate involve alterations of the central nervous system, peripheral vascular tone and cardiac function.
Maternal hypotension has resulted from regional anesthesia. Local anesthetics produce vasodilation by blocking sympathetic nerves. Elevating the patient’s legs and positioning her on her left side will help prevent decreases in blood pressure.
The fetal heart rate also should be monitored continuously, and electronic fetal monitoring is highly advisable.
Epidural, spinal, paracervical, or pudendal anesthesia may alter the forces of parturition through changes in uterine contractility or maternal expulsive efforts. In one study, paracervical block anesthesia was associated with a decrease in the mean duration of first stage labor and facilitation of cervical dilation. However, spinal and epidural anesthesia have also been reported to prolong the second stage of labor by removing the parturient’s reflex urge to bear down or by interfering with motor function. The use of obstetrical anesthesia may increase the need for forceps assistance.
The use of some local anesthetic drug products during labor and delivery may be followed by diminished muscle strength and tone for the first day or two of life. The long-term significance of these observations is unknown. Fetal bradycardia may occur in 20 to 30 percent of patients receiving paracervical nerve block anesthesia with the amide-type local anesthetics and may be associated with fetal acidosis. Fetal heart rate should always be monitored during paracervical anesthesia. The physician should weigh the possible advantages against risks when considering a paracervical block in prematurity, toxemia of pregnancy, and fetal distress. Careful adherence to recommended dosage is of the utmost importance in obstetrical paracervical block. Failure to achieve adequate analgesia with recommended doses should arouse suspicion of intravascular or fetal intracranial injection. Cases compatible with unintended fetal intracranial injection of local anesthetic solution have been reported following intended paracervical or pudendal block or both. Babies so affected present with unexplained neonatal depression at birth, which correlates with high local anesthetic serum levels, and often manifest seizures within six hours. Prompt use of supportive measures combined with forced urinary excretion of the local anesthetic has been used successfully to manage this complication.
Case reports of maternal convulsions and cardiovascular collapse following use of some local anesthetics for paracervical block in early pregnancy (as anesthesia for elective abortion) suggest that systemic absorption under these circumstances may be rapid. The recommended maximum dose of each drug should not be exceeded. Injection should be made slowly and with frequent aspiration. Allow a 5-minute interval between sides.Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when lidocaine hydrochloride is administered to a nursing woman.
Pediatric Use
Dosages in children should be reduced, commensurate with age, body weight and physical condition, see DOSAGE AND ADMINISTRATION.
-
ADVERSE REACTIONS
Systemic
Adverse experiences following the administration of lidocaine hydrochloride are similar in nature to those observed with other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage, rapid absorption or inadvertent intravascular injection, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
Central Nervous System
CNS manifestations are excitatory and/or depressant and may be characterized by lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest.
Drowsiness following the administration of lidocaine hydrochloride is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption.
Cardiovascular System
Cardiovascular manifestations are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
Allergic
Allergic reactions are characterized by cutaneous lesions, urticaria, edema or anaphylactoid reactions. Allergic reactions may occur as a result of sensitivity to local anesthetic agents. Allergic reactions, including anaphylactic reactions, may occur as a result of sensitivity to lidocaine, but are infrequent. If allergic reactions do occur, they should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
There have been no reports of cross sensitivity between lidocaine hydrochloride and procainamide or between lidocaine hydrochloride and quinidine.Neurologic
The incidences of adverse reactions associated with the use of local anesthetics may be related to the total dose of local anesthetic administered and are also dependent upon the particular drug used, the route of administration and the physical status of the patient. In a prospective review of 10,440 patients who received lidocaine hydrochloride for spinal anesthesia, the incidences of adverse reactions were reported to be about 3 percent each for positional headaches, hypotension and backache; 2 percent for shivering; and less than 1 percent each for peripheral nerve symptoms, nausea, respiratory inadequacy and double vision. Many of these observations may be related to local anesthetic techniques, with or without a contribution from the local anesthetic.
In the practice of caudal or lumbar epidural block, occasional unintentional penetration of the subarachnoid space by the catheter may occur. Subsequent adverse effects may depend partially on the amount of drug administered subdurally. These may include spinal block of varying magnitude (including total spinal block), hypotension secondary to spinal block, loss of bladder and bowel control, and loss of perineal sensation and sexual function. Persistent motor, sensory and/or autonomic (sphincter control) deficit of some lower spinal segments with slow recovery (several months) or incomplete recovery have been reported in rare instances when caudal or lumbar epidural block has been attempted. Backache and headache have also been noted following use of these anesthetic procedures.
There have been reported cases of permanent injury to extraocular muscles requiring surgical repair following retrobulbar administration. -
OVERDOSAGE
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics or to unintended subarachnoid injection of local anesthetic solution (see ADVERSE REACTIONS, WARNINGS, and PRECAUTIONS).
Management of Local Anesthetic Emergencies
The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient’s state of consciousness after each local anesthetic injection. At the first sign of change, oxygen should be administered.
The first step in the management of convulsions, as well as underventilation or apnea due to unintended subarachnoid injection of drug solution, consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask. Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously. The clinician should be familiar, prior to the use of local anesthetics, with these anticonvulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor as directed by the clinical situation (e.g., ephedrine).
If not treated immediately, both convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias and cardiac arrest. Underventilation or apnea due to unintentional subarachnoid injection of local anesthetic solution may produce these same signs and also lead to cardiac arrest if ventilatory support is not instituted. If cardiac arrest should occur, standard cardiopulmonary resuscitative measures should be instituted.
Endotracheal intubation, employing drugs and techniques familiar to the clinician, may be indicated, after initial administration of oxygen by mask, if difficulty is encountered in the maintenance of a patent airway or if prolonged ventilatory support (assisted or controlled) is indicated.
Dialysis is of negligible value in the treatment of acute overdosage with lidocaine hydrochloride.
The oral LD 50of lidocaine hydrochloride in non-fasted female rats is 459 (346 to 773) mg/kg (as the salt) and 214 (159 to 324) mg/kg (as the salt) in fasted female rats. -
DOSAGE AND ADMINISTRATION
Table 1 (Recommended Dosages) summarizes the recommended volumes and concentrations of lidocaine hydrochloride injection for various types of anesthetic procedures. The dosages suggested in this table are for normal healthy adults and refer to the use of epinephrine-free solutions. When larger volumes are required, only solutions containing epinephrine should be used except in those cases where vasopressor drugs may be contraindicated.
There have been adverse event reports of chondrolysis in patients receiving intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures. Lidocaine hydrochloride injection is not approved for this use (see WARNINGSand DOSAGE AND ADMINISTRATION).
These recommended doses serve only as a guide to the amount of anesthetic required for most routine procedures. The actual volumes and concentrations to be used depend on a number of factors such as type and extent of surgical procedure, depth of anesthesia and degree of muscular relaxation required, duration of anesthesia required, and the physical condition of the patient. In all cases the lowest concentration and smallest dose that will produce the desired result should be given. Dosages should be reduced for children and for the elderly and debilitated patients and patients with cardiac and/or liver disease.
The onset of anesthesia, the duration of anesthesia and the degree of muscular relaxation are proportional to the volume and concentration (i.e., total dose) of local anesthetic used. Thus, an increase in volume and concentration of lidocaine hydrochloride injection will decrease the onset of anesthesia, prolong the duration of anesthesia, provide a greater degree of muscular relaxation and increase the segmental spread of anesthesia. However, increasing the volume and concentration of lidocaine hydrochloride injection may result in a more profound fall in blood pressure when used in epidural anesthesia. Although the incidence of side effects with lidocaine hydrochloride is quite low, caution should be exercised when employing large volumes and concentrations, since the incidence of side effects is directly proportional to the total dose of local anesthetic agent injected.Epidural Anesthesia
For epidural anesthesia, only the following dosage forms of lidocaine hydrochloride injection are recommended:
1% without epinephrine 5 mL ampules
2% without epinephrine 2 mL ampules
Although these solutions are intended specifically for epidural anesthesia, they may also be used for infiltration and peripheral nerve block, provided they are employed as single dose units. These solutions contain no bacteriostatic agent.
In epidural anesthesia, the dosage varies with the number of dermatomes to be anesthetized (generally 2 to 3 mL of the indicated concentration per dermatome).Caudal and Lumbar Epidural Block
As a precaution against the adverse experience sometimes observed following unintentional penetration of the subarachnoid space, a test dose such as 2 to 3 mL of 1.5% lidocaine hydrochloride should be administered at least 5 minutes prior to injecting the total volume required for a lumbar or caudal epidural block. The test dose should be repeated if the patient is moved in a manner that may have displaced the catheter. Epinephrine, if contained in the test dose (10 to 15 mcg have been suggested), may serve as a warning of unintentional intravascular injection. If injected into a blood vessel, this amount of epinephrine is likely to produce a transient “epinephrine response” within 45 seconds, consisting of an increase in heart rate and systolic blood pressure, circumoral pallor, palpitations and nervousness in the unsedated patient. The sedated patient may exhibit only a pulse rate increase of 20 or more beats per minute for 15 or more seconds. Patients on beta blockers may not manifest changes in heart rate, but blood pressure monitoring can detect an evanescent rise in systolic blood pressure. Adequate time should be allowed for onset of anesthesia after administration of each test dose. The rapid injection of a large volume of lidocaine hydrochloride injection through the catheter should be avoided, and, when feasible, fractional doses should be administered.
In the event of the known injection of a large volume of local anesthetic solution into the subarachnoid space, after suitable resuscitation and if the catheter is in place, consider attempting the recovery of drug by draining a moderate amount of cerebrospinal fluid (such as 10 mL) through the epidural catheter. -
MAXIMUM RECOMMENDED DOSAGES
Adults
For normal healthy adults, the maximum individual dose should not exceed 4.5 mg/kg (2 mg/lb) of body weight, and in general it is recom mended that the max i m um t otal dose does not exceed 300 mg. For continuous epidural or caudal anesthesia, the maximum recommended dosage should not be administered at intervals of less than 90 minutes. When continuous lumbar or caudal epidural anesthesia is used for non-obstetrical procedures, more drug may be administered if required to produce adequate anesthesia.
The maximum recommended dose per 90 minute period of lidocaine hydrochloride for paracervical block in obstetrical patients and non-obstetrical patients is 200 mg total. One half of the total dose is usually administered to each side. Inject slowly, five minutes between sides (see also discussion of paracervical block in PRECAUTIONS).
For intravenous regional anesthesia, the dose administered should not exceed 4 mg/kg in adults.Children
It is difficult to recommend a maximum dose of any drug for children, since this varies as a function of age and weight. For children over 3 years of age who have a normal lean body mass and normal body development, the maximum dose is determined by the child’s age and weight. For example, in a child of 5 years weighing 50 lbs the dose of lidocaine hydrochloride should not exceed 75 to 100 mg (1.5 to 2 mg/lb). The use of even more dilute solutions (i.e., 0.25 to 0.5%) and total dosages not to exceed 3 mg/kg (1.4 mg/lb) are recommended for induction of intravenous regional anesthesia in children.
In order to guard against systemic toxicity, the lowest effective concentration and lowest effective dose should be used at all times. In some cases it will be necessary to dilute available concentrations with 0.9% sodium chloride injection in order to obtain the required final concentration.
NOTE: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not use if solution is discolored or contains a precipitate.Table 1: Recommended Dosages Procedure Lidocaine Hydrochloride Injection
(without epinephrine)Conc (%) Vol (mL) Total Dose (mg) *Dose determined by number of dermatomes to be anesthetized (2 to 3 mL/dermatome).
Infiltration
Percutaneous
0.5 or 1
1 to 60
5 to 300
Intravenous regional
0.5
10 to 60
50 to 300
Peripheral Nerve Blocks, e.g.,
Brachial
1.5
15 to 20
225 to 300
Dental
2
1 to 5
20 to 100
Intercostal
1
3
30
Paravertebral
1
3 to 5
30 to 50
Pudendal (each side)
1
10
100
Paracervical
Obstetrical analgesia (each side)
1
10
100
Sympathetic Nerve Blocks, e.g.,
Cervical (stellate ganglion)
1
5
50
Lumbar
1
5 to 10
50 to 100
Central Neural Blocks
Epidural*
Thoracic
1
20 to 30
200 to 300
Lumbar
Analgesia
1
25 to 30
250 to 300
Anesthesia
1.5
15 to 20
225 to 300
2
10 to 15
200 to 300
Caudal
Obstetrical analgesia
1
20 to 30
200 to 300
Surgical anesthesia
1.5
15 to 20
225 to 300
THE ABOVE SUGGESTED CONCENTRATIONS AND VOLUMES SERVE ONLY AS A GUIDE. OTHER VOLUMES AND CONCENTRATIONS MAY BE USED PROVIDED THE TOTAL MAXIMUM RECOMMENDED DOSE IS NOT EXCEEDED.
- STERILIZATION, STORAGE AND TECHNICAL PROCEDURES
-
HOW SUPPLIED
Lidocaine Hydrochloride Injection USP, is supplied as follows:
Lidocaine Hydrochloride Injection USP, 1% (10 mg/mL)
2 mL Ampules in a Carton of 10 NDC 55150-158-72
Sterile, Nonpyrogenic
Discard unused portion.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Distributed by:
AuroMedics Pharma LLC
279 Princeton-Hightstown Rd.
E. Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
Revised: October 2021 - Povidone-Iodine Swabsticks
- ACTIVE INGREDIENT
- Purpose:
- Warnings:
- Do not use:
- Ask a doctor before use if you have:
- Stop Use:
- Keep Out Of Reach Of Children
- Directions Povidone iodine:
- Other information:
- INDICATIONS & USAGE
- Inactive Ingredients
- Isopropyl Alcohol 70% Prep Pads
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only
Flammable - keep away from fire or flame
When using this product do not
- get into eyes
- apply over large areas of the body
- in case of deep or puncture wounds, animal bites or serious burns consult a doctor
- Directions
- Other information
- Inactive ingredient
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL
NDC: 76420-771-01
RX Only
Ketorocaine-L™
Kit Contains
1 Ketorolac Tromethamine Inj., USP 30mg/mL (1mL)
1 Lidocaine HCl Injection, USP 1% Ampule (2mL)
1 Povidone-Iodine Swabsticks (3 Swabs)
2 Isopropyl Alcohol 70% Prep Pads
1 Pair Nitrile Powder Free Sterile Gloves (M)
1 Drape
1 Adhesive Bandage
5 Non Sterile 4x4 Gauze
Needles and Syringes Not Included
1 Dose
Single Use Only
Distributed by:
Enovachem™
PHARMACEUTICALS
Torrance, CA 90501

-
INGREDIENTS AND APPEARANCE
KETOROCAINE L KIT
ketorolac tromethamine, lidocaine hydrochloride, povidine iodine kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:76420-771 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76420-771-01 1 in 1 CARTON; Type 1: Convenience Kit of Co-Package 09/23/2021 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 1 mL Part 2 1 AMPULE 2 mL Part 3 1 PACKET 0.9 mL Part 4 2 POUCH 10 mL Part 1 of 4 KETOROLAC TROMETHAMINE
ketorolac tromethamine injection, solutionProduct Information Item Code (Source) NDC:72266-118 Route of Administration INTRAMUSCULAR, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength KETOROLAC TROMETHAMINE (UNII: 4EVE5946BQ) (KETOROLAC - UNII:YZI5105V0L) KETOROLAC TROMETHAMINE 30 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) 100 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 4.35 mg in 1 mL HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72266-118-01 1 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA204216 06/23/2023 Part 2 of 4 LIDOCAINE HYDROCHLORIDE
lidocaine hydrochloride injection, solutionProduct Information Item Code (Source) NDC:55150-158 Route of Administration INFILTRATION, PERINEURAL, INTRACAUDAL, EPIDURAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 7 mg in 1 mL WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55150-158-72 10 in 1 CARTON 1 2 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA203040 03/14/2013 Part 3 of 4 POVIDINE IODINE
povidine iodine swabProduct Information Item Code (Source) NDC:67777-419 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) CITRIC ACID ACETATE (UNII: DSO12WL7AU) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67777-419-02 0.9 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 09/13/2016 Part 4 of 4 ISOPROPYL ALCOHOL
isopropyl alcohol swabProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 5 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 01/01/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/15/2014 Labeler - Asclemed USA, Inc. (059888437)

