Label: ATOPALM MUSCLE AND JOINT- menthol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 51141-0240-4 - Packager: NeoPharm Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 15, 2012
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- SPL UNCLASSIFIED SECTION
-
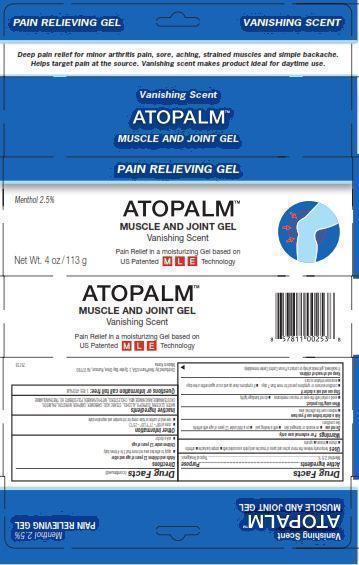
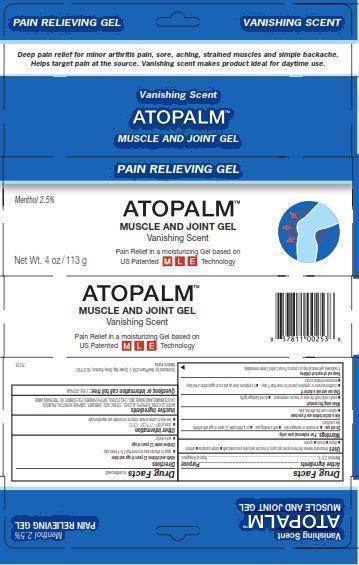
PRINCIPAL DISPLAY PANEL
Pain Relieving Gel VANISHING SCENT
Deep pain relief for minor arthritis pain, sore, aching, strained muscles and simple backache.
Helps target pain at the source. Vanishing scent makes product ideal for daytime use.
Vanishing Scent
ATOPALM (TM)
MUSCLE AND JOINT GEL
Menthol 2.5%
Pain Relief in moisturizing Gel based on US Patented MLE Technology
Net Wt. 4 oz/113 g

-
INGREDIENTS AND APPEARANCE
ATOPALM MUSCLE AND JOINT
menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51141-0240 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 2.5 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) ISOPROPYL ALCOHOL (UNII: ND2M416302) STEARIC ACID (UNII: 4ELV7Z65AP) MYRISTOYL/PALMITOYL OXOSTEARAMIDE/ARACHAMIDE MEA (UNII: 1211AIM8G7) STEARIC ACID (UNII: 4ELV7Z65AP) CHOLESTEROL (UNII: 97C5T2UQ7J) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 60 (UNII: CAL22UVI4M) TROLAMINE (UNII: 9O3K93S3TK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51141-0240-4 1 in 1 CARTON 1 113 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 08/14/2012 Labeler - NeoPharm Co., Ltd. (965502912) Establishment Name Address ID/FEI Business Operations NeoPharm Co., Ltd. 631101883 manufacture(51141-0240)