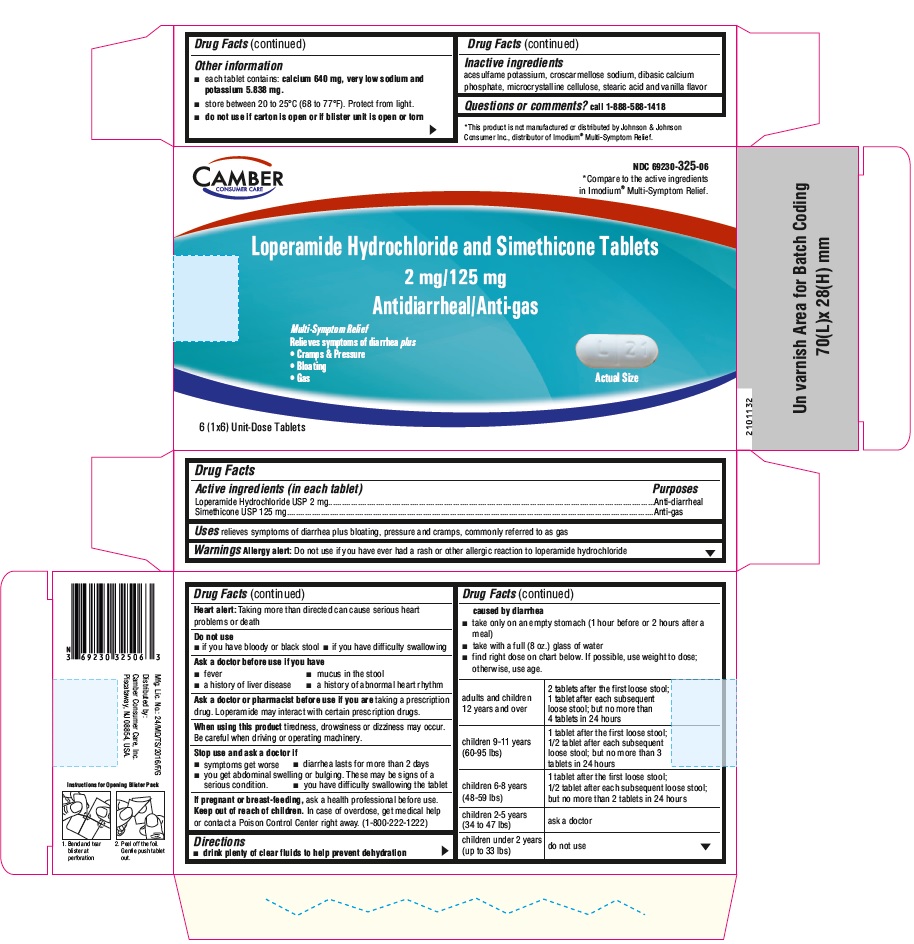
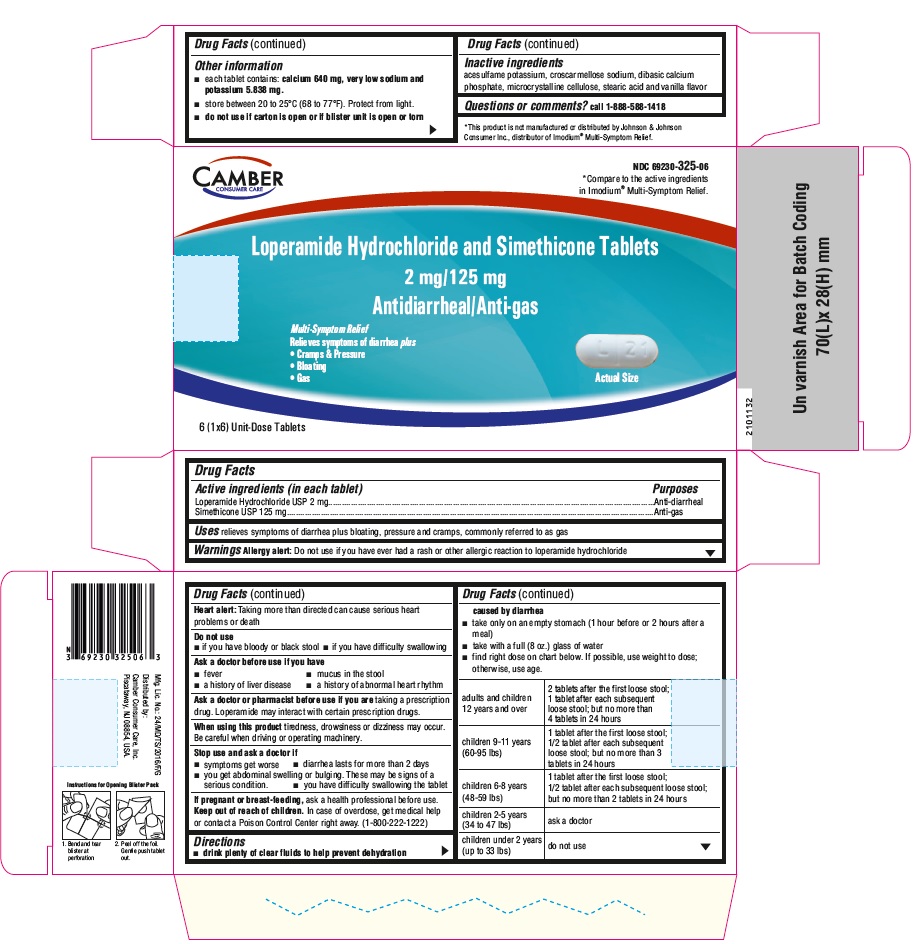
Label: LOPERAMIDE HYDROCHLORIDE AND SIMETHICONE- loperamide hydrochloride and simethicone tablet
-
NDC Code(s):
69230-325-04,
69230-325-06,
69230-325-08,
69230-325-12, view more69230-325-18, 69230-325-24
- Packager: Camber Consumer Care Inc
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated September 12, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT(S)
- PURPOSES
- USE(S)
- WARNINGS
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- ASK A DOCTOR OR PHARMACIST BEFORE USE IF
- WHEN USING THIS PRODUCT
- STOP USE AND ASK A DOCTOR IF
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
• drink plenty of clear fluids to help prevent dehydration caused by diarrhea
• take only on an empty stomach (1 hour before or 2 hours after a meal)
• take with a full (8 oz.) glass of water
• find right dose on chart below. If possible, use weight to dose; otherwise, use age.
adults and children 12 years and over
2 tablets after the first loose stool; 1 tablet after each subsequent loose stool; but no more than 4 tablets in 24 hours
children 9-11 years
(60-95 lbs)1 tablet after the first loose stool; 1/2 tablet after each subsequent loose stool; but no more than 3 tablets in 24 hours
children 6-8 years
(48-59 lbs)
1 tablet after the first loose stool; 1/2 tablet after each subsequent loose stool; but no more than 2 tablets in 24 hourschildren 2-5 years
(34 to 47 lbs)
ask a doctor
children under 2 years
(up to 33 lbs)
do not use
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LOPERAMIDE HYDROCHLORIDE AND SIMETHICONE
loperamide hydrochloride and simethicone tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69230-325 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 2 mg DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 125 mg Inactive Ingredients Ingredient Name Strength ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score 2 pieces Shape CAPSULE Size 17mm Flavor VANILLA Imprint Code H;L21 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69230-325-04 1 in 1 CARTON 06/17/2021 1 4 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:69230-325-08 2 in 1 CARTON 06/17/2021 2 4 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:69230-325-06 1 in 1 CARTON 06/17/2021 3 6 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC:69230-325-12 2 in 1 CARTON 06/17/2021 4 6 in 1 BLISTER PACK; Type 0: Not a Combination Product 5 NDC:69230-325-18 3 in 1 CARTON 06/17/2021 5 6 in 1 BLISTER PACK; Type 0: Not a Combination Product 6 NDC:69230-325-24 4 in 1 CARTON 06/17/2021 6 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211438 06/17/2021 Labeler - Camber Consumer Care Inc (079539968) Establishment Name Address ID/FEI Business Operations Annora Pharma Private Limited 650980746 manufacture(69230-325)